- Biotechnology
- Breast Recurrence Score Test Market
Breast Recurrence Score Test Market Size, Share, and Growth Forecast, 2026 - 2033
Breast Recurrence Score Test Market by Product (Test Kits, Reagents & Consumables), Indication (Stage 1 Breast Cancer, Stage 2 Breast Cancer, Estrogen-Receptor-Positive Cancer, Lymph-Node-Negative Cancer), and Regional Analysis for 2026 - 2033
Breast Recurrence Score Test Market Size and Trends Analysis
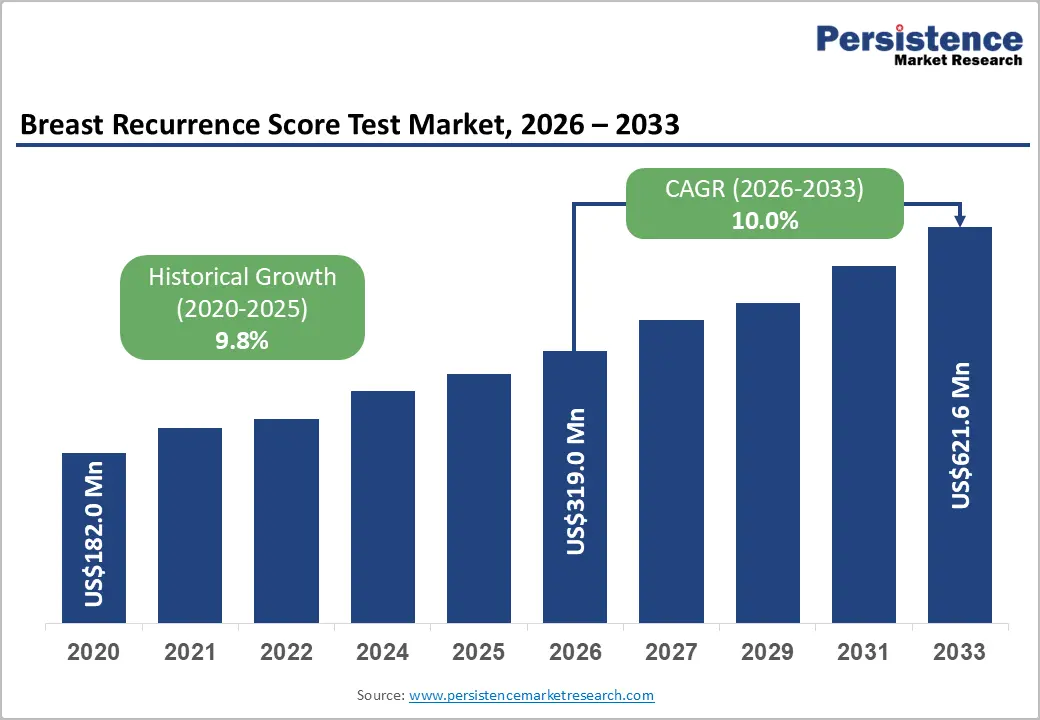
The global breast recurrence score test market size is likely to be valued at US$319.0 million in 2026 and is expected to reach US$621.6 million by 2033, growing at a CAGR of 10.0% during the forecast period from 2026 to 2033, driven by the rising burden of breast cancer and increasing adoption of precision oncology and genomic diagnostics.
Breast recurrence score assays are widely used to evaluate recurrence risk and guide chemotherapy decisions in patients with early-stage hormone receptor-positive breast cancer. According to the World Health Organization (WHO), breast cancer remained the most commonly diagnosed cancer among women globally, with around 2.3 million new cases and 670,000 deaths reported in 2022. Recent clinical guideline support from organizations such as NCCN and ASCO, along with growing awareness of personalized treatment planning and reduction of unnecessary chemotherapy use, continues to accelerate demand for recurrence score testing.
Key Industry Highlights:
- Leading Region: North America is anticipated to be the leading region, accounting for 30% market share in 2026, driven by strong adoption of precision oncology, favorable reimbursement policies, and an established genomic testing infrastructure.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region, supported by expanding cancer diagnostics infrastructure, rising breast cancer awareness, and increasing adoption of genomic testing.
- Leading Product Type: Test kits are projected to represent the leading product type in 2026, accounting for 70% of the revenue share, due to widespread clinical adoption and standardized genomic analysis capabilities.
- Leading Indication: Stage 2 breast cancer is anticipated to be the leading indication, accounting for over 43% of the revenue share in 2026, supported by the higher need for recurrence risk evaluation and treatment planning.
- Key Opportunity: The key market opportunity in the Breast Recurrence Score Test Market lies in expanding AI-driven genomic and liquid biopsy-based precision oncology solutions to enable more accurate, minimally invasive, and personalized breast cancer treatment decision-making worldwide.
DRO Analysis
Driver - Increasing Breast Cancer Incidence and Emphasis on Personalized Medicine
Healthcare providers are increasingly adopting precision oncology approaches to optimize treatment decisions and improve patient outcomes. Recurrence score assays help clinicians identify patients who may safely avoid chemotherapy while ensuring high-risk patients receive timely intervention.
Growing awareness of personalized medicine, combined with expanded breast cancer screening programs and supportive clinical guidelines from oncology organizations, has accelerated market adoption. Hospitals and cancer centers are integrating genomic testing into routine care pathways to reduce overtreatment, improve survival rates, and support evidence-based treatment planning worldwide.
The growing preference for individualized cancer treatment strategies is a major factor strengthening the breast cancer recurrence score test market. Physicians increasingly rely on molecular profiling tools to assess recurrence risk and determine chemotherapy benefits for hormone receptor-positive breast cancer patients. Advancements in genomic sequencing technologies and biomarker analysis have improved testing accuracy and clinical confidence.
Restraint - Complexity in Result Interpretation and Patient-Specific Factors
Breast recurrence score tests generate detailed molecular data that require specialized oncology expertise for accurate treatment decisions. Variations in patient age, tumor biology, genetic mutations, and comorbid conditions can influence interpretation and reduce consistency across clinical settings. Some physicians may hesitate to rely solely on recurrence scores when determining chemotherapy necessity, particularly in borderline-risk cases.
Patient-specific biological variability also restricts the universal applicability of breast recurrence score testing. Differences in ethnicity, tumor heterogeneity, hormonal status, and genetic expression patterns can affect test performance and predictive accuracy. In certain clinical situations, recurrence scores may not fully capture disease aggressiveness or long-term relapse risk, leading to uncertainty in treatment planning. High dependence on centralized laboratories and standardized tissue handling procedures may create inconsistencies in sample quality and reporting timelines.
Opportunity - Technological Convergence with AI, Liquid Biopsy, and Multi-Omics
AI-powered analytics can improve recurrence prediction accuracy by identifying complex genomic patterns and supporting faster clinical decision-making. Liquid biopsy advancements enable minimally invasive cancer monitoring through blood-based biomarker detection, improving patient convenience and expanding testing accessibility.
Multi-omics approaches combining genomics, proteomics, transcriptomics, and metabolomics provide deeper insights into tumor behavior and personalized treatment response. These innovations are expected to enhance precision oncology capabilities, strengthen recurrence risk assessment, and support the development of next-generation breast cancer diagnostic solutions.
Technological convergence is also encouraging strategic collaborations between diagnostic companies, research institutes, and oncology centers to develop advanced predictive testing platforms. Integration of AI algorithms with genomic databases can help clinicians generate more accurate individualized treatment recommendations while reducing interpretation complexity. Emerging digital pathology tools and cloud-based data platforms are improving scalability and enabling real-time clinical analysis across healthcare networks.
Category-wise Analysis
Product Type Insights
Test kits are expected to lead accounting for 70% of revenue in 2026, driven by their critical role in standardized genomic profiling and recurrence risk assessment for breast cancer patients. Continuous technological advancements in molecular diagnostics and automation improve testing efficiency and diagnostic accuracy. A notable example includes Exact Sciences, whose Oncotype DX Breast Recurrence Score test has become one of the most widely adopted genomic assays for evaluating recurrence risk.
Reagents & consumables are likely to represent the fastest-growing segment, supported by increasing genomic testing volumes and expanding laboratory infrastructure worldwide. The growing number of recurrence score assays performed in centralized diagnostic laboratories has significantly increased demand for specialized reagents, assay chemicals, buffers, cartridges, and consumable components required for molecular testing workflows. For example, Thermo Fisher Scientific, which provides a broad portfolio of molecular diagnostic reagents and consumables supporting genomic profiling and oncology testing applications.
Indication Insights
Stage 2 breast cancer is projected to lead the market, capturing around 43% of the revenue share in 2026, supported by the increased need for accurate recurrence risk evaluation and informed chemotherapy decisions among patients with moderate disease progression. Physicians frequently utilize recurrence score testing in Stage 2 breast cancer cases to determine whether chemotherapy can provide a significant clinical benefit while minimizing unnecessary treatment exposure. For instance, Agendia, whose genomic testing solutions are widely used to support treatment decision-making and recurrence risk assessment for early-stage and Stage 2 breast cancer patients in clinical oncology settings.
Estrogen-receptor-positive cancer is likely to be the fastest-growing indication due to increasing clinical reliance on genomic profiling for hormone receptor-positive breast cancer management. Recurrence score testing plays an essential role in identifying patients who may safely avoid chemotherapy while benefiting from endocrine therapy-based treatment approaches. For example, Hologic, whose molecular diagnostic solutions support personalized oncology care by enabling physicians to assess tumor biology.

Regional Insights
North America Breast Recurrence Score Test Market Trends
North America is anticipated to be the leading region, accounting for a market share of 30% in 2026, supported by advanced oncology infrastructure, strong reimbursement coverage, and high adoption of precision medicine technologies. The region benefits from widespread clinical acceptance of genomic recurrence assays for early-stage breast cancer treatment planning. A notable example includes Guardant Health, which continues expanding its oncology diagnostics portfolio and liquid biopsy technologies to improve personalized cancer management.
U.S. Breast Recurrence Score Test Market Trends
The U.S. dominates the regional market, driven by high breast cancer screening rates and rapid integration of genomic diagnostics into oncology treatment pathways. Strong reimbursement support for recurrence score testing and increasing physician confidence in personalized medicine continue driving demand. Leading cancer research institutions are expanding genomic profiling programs to optimize chemotherapy decisions and improve patient outcomes.
Canada Breast Recurrence Score Test Market Trends
Canada is a significant market for breast cancer recurrence score tests, supported by expanding awareness of personalized oncology treatment and increasing adoption of genomic diagnostics in cancer care programs. Government-supported healthcare infrastructure and growing investments in molecular pathology services are improving access to recurrence score testing. Canadian oncology centers are increasingly integrating genomic assays into breast cancer treatment decision-making processes.
Europe Breast Recurrence Score Test Market Trends
Europe is likely to be a significant market for breast recurrence score tests in 2026, due to strong healthcare infrastructure, rising breast cancer awareness, and increasing adoption of precision oncology solutions. The region is experiencing growing demand for genomic profiling tools that support personalized treatment planning and reduce unnecessary chemotherapy utilization. For instance, QIAGEN continues to strengthen its molecular diagnostics and oncology testing capabilities.
U.K. Breast Recurrence Score Test Market Trends
The U.K. is a significant market for breast cancer recurrence score tests, supported by increasing integration of genomic testing into national cancer care pathways and growing awareness regarding personalized medicine. Healthcare organizations are emphasizing early recurrence risk assessment and optimized chemotherapy selection for breast cancer patients. Rising adoption of molecular diagnostic technologies across hospitals and pathology laboratories is strengthening market growth.
Germany Breast Recurrence Score Test Market Trends
Germany dominates the regional market due to its advanced healthcare infrastructure and strong focus on precision oncology. Increasing adoption of genomic testing in oncology clinics and cancer research centers is supporting market expansion. German healthcare providers are emphasizing evidence-based treatment planning and recurrence risk evaluation to improve breast cancer outcomes.
Asia Pacific Breast Recurrence Score Test Market Trends
The Asia Pacific region is likely to be the fastest-growing region, driven by the increasing breast cancer incidence, improving healthcare infrastructure, and rising adoption of genomic diagnostics. Growing awareness regarding personalized cancer treatment and advancements in molecular testing technologies are accelerating demand across hospitals and oncology centers. For example, Illumina, which continues expanding its genomic sequencing and oncology diagnostics capabilities to support precision medicine development and advanced cancer testing solutions.
China Breast Recurrence Score Test Market Trends
China dominates the regional market, supported by increasing breast cancer prevalence and the rapid expansion of precision oncology programs. Government initiatives supporting molecular diagnostics and cancer screening are strengthening genomic testing adoption across major healthcare institutions. Chinese biotechnology companies and hospitals are increasingly investing in next-generation sequencing and biomarker research technologies.
India Breast Recurrence Score Test Market Trends
India is a significant market for the breast cancer recurrence score test. Due to growing breast cancer awareness and increasing demand for affordable genomic diagnostic solutions. Expansion of private oncology hospitals and molecular pathology laboratories is improving access to recurrence score testing across urban healthcare centers. Physicians are increasingly adopting personalized treatment approaches to reduce overtreatment and optimize chemotherapy decisions.

Competitive Landscape
The global breast recurrence score test market exhibits a moderately fragmented structure, driven by the presence of established molecular diagnostics companies, precision oncology innovators, and emerging genomic testing providers. The market is witnessing increasing competition due to rising demand for personalized breast cancer treatment and recurrence risk assessment solutions.
With key leaders including Exact Sciences, Agendia, NanoString Technologies, and Myriad Genetics, the market continues to evolve through continuous innovation, international expansion, and strategic oncology partnerships. These players compete through product innovation, clinical validation studies, strategic collaborations, regulatory approvals, and expansion of precision oncology capabilities.
Key Industry Developments:
- In February 2026, Exact Sciences announced that its Oncotype DX Breast Recurrence Score test surpassed usage in over 2 million breast cancer patients worldwide, highlighting its strong position as a global standard in precision oncology for early-stage HR-positive, HER2-negative breast cancer. The milestone reflects more than two decades of clinical validation and widespread adoption across oncology care systems, where the test is used to guide chemotherapy decisions and reduce overtreatment.
- In May 2026, Caris Life Sciences launched Caris MI Clarity, an AI-powered prognostic test designed to predict both early and late distant recurrence risk in HR-positive/HER2-negative early-stage breast cancer patients.
Companies Covered in Breast Recurrence Score Test Market
- Exact Sciences (Genomic Health Inc.)
- NanoString Technologies, Inc.
- Myriad Genetics, Inc.
- Foundation Medicine, Inc.
- Thermo Fisher Scientific
- Illumina, Inc.
- Hologic Inc. (Biotheranostics)
- Roche Diagnostics
- Agendia
- Guardant Health
- Biocartis
- Qiagen
- Abbott
- Epic Sciences
Frequently Asked Questions
The global breast recurrence score test market is projected to reach US$319.0 million in 2026.
The breast recurrence score test market is driven by the rising incidence of breast cancer, growing adoption of personalized oncology, and increasing clinical use of genomic testing to guide chemotherapy decisions.
The breast recurrence score test market is expected to grow at a CAGR of 10.0% from 2026 to 2033.
Key opportunities lie in the integration of AI, liquid biopsy, and multi-omics technologies to enhance recurrence prediction accuracy and expand personalized breast cancer treatment applications.
Exact Sciences, NanoString Technologies, Myriad Genetics, Foundation Medicine, and Thermo Fisher Scientific are the leading players.








