- Healthcare Services
- Digital Twin in Healthcare Market
Digital Twin in Healthcare Market Size, Share, and Growth Forecast, 2026 - 2033
Digital Twin in Healthcare Market by Component (Software, Services), Application (Personalized Medicine, Others), End-user (Medical Device Companies, Pharma & Biopharma Companies, Others), and Regional Analysis for 2026 – 2033
Digital Twin in Healthcare Market Size and Trends Analysis
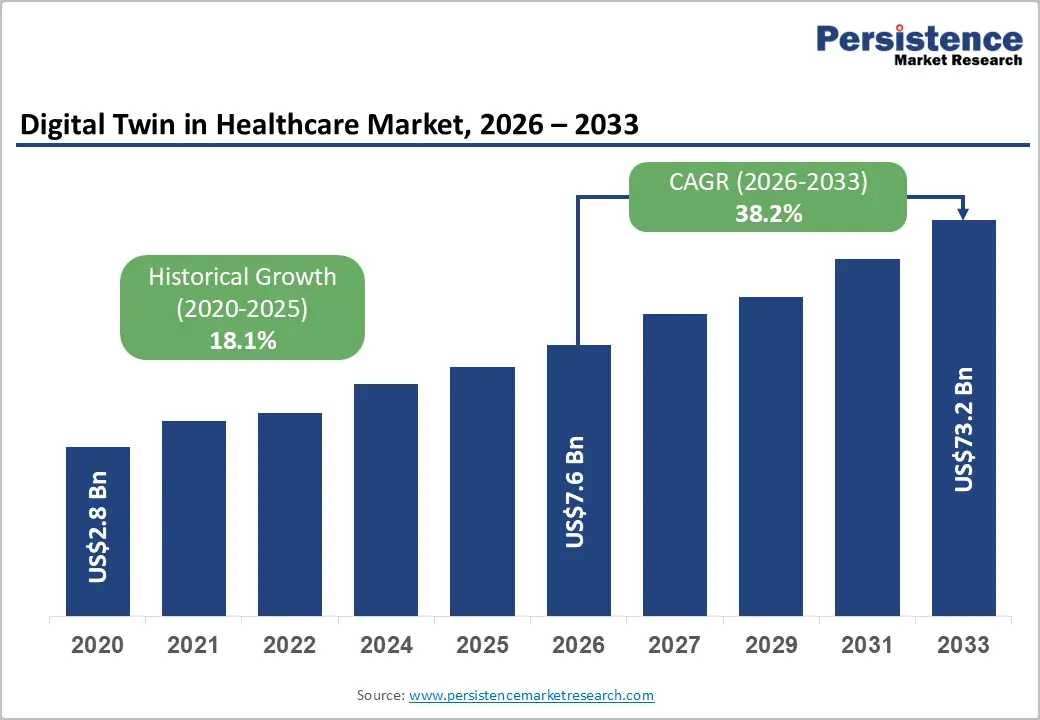
The global digital twin in healthcare market size is likely to be valued at US$7.6 billion in 2026, and is expected to reach US$73.2 billion by 2033, growing at a CAGR of 38.2% during the forecast period from 2026 to 2033, driven by the increasing prevalence of personalized medicine initiatives, rapid integration of AI/ML with patient-specific data models, growing demand for virtual simulation in surgical planning and drug discovery, and expanding adoption across hospitals, pharma companies, and medical device manufacturers.
Increasing recognition of digital twins in healthcare as critical for precision therapeutics, virtual clinical trials, and operational efficiency in emerging value-based care and digital health ecosystems remains a major driver of market growth.
Key Industry Highlights:
- Leading Region: North America, anticipated to account for a 42% market share in 2026, driven by early adoption of AI-driven twins, a strong funding ecosystem, and high demand in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by rapid digital health transformation, increasing pharma R&D outsourcing, and growing precision medicine initiatives in China and India.
- Dominant Component: Software, to hold approximately 65% of the market share, as it remains the core enabler of twin modelling and simulation.
- Leading Application: Personalized Medicine, contributing nearly 38% of the market revenue, due to the highest clinical and commercial impact.
- May 2025: Denmark-based healthcare technology company Teton launched a real-time 3D hospital room digital twin system powered by Gefion, Denmark’s national supercomputer, to help nurses monitor patients, detect risks, and improve workflow efficiency.
| Report Attribute | Details |
|---|---|
|
Digital Twin in Healthcare Market Size (2026E) |
US$7.6 Bn |
|
Market Value Forecast (2033F) |
US$73.2 Bn |
|
Projected Growth CAGR (2026-2033) |
38.2% |
|
Historical Market Growth (2020-2025) |
18.1% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Growth Analysis – Precision Medicine and Virtual Clinical Trials Acceleration
Precision medicine and virtual clinical trials are transforming the way therapies are developed and delivered in modern healthcare. Precision medicine focuses on tailoring treatments to individual patient characteristics such as genetic profile, lifestyle, and disease biomarkers. By analyzing large datasets from genomics, medical imaging, and patient records, researchers can identify specific patient subgroups that are more likely to respond to a particular therapy. This targeted approach improves treatment effectiveness, reduces adverse effects, and increases the probability of success during drug development.
Virtual clinical trials further accelerate this process by using digital technologies to conduct portions of trials remotely. Instead of requiring patients to visit research centers frequently, participants can share health data through wearable devices, mobile applications, and telemedicine platforms. This approach allows researchers to collect real-time data, monitor patient outcomes continuously, and recruit participants from broader geographic locations. As a result, virtual trials reduce logistical challenges, shorten recruitment timelines, and improve patient participation rates.
Surging AI/ML Integration and Real-World Evidence Generation
The growing integration of artificial intelligence (AI) and machine learning (ML) in healthcare and life sciences is significantly improving data analysis, clinical decision-making, and evidence generation. AI and ML algorithms can process large volumes of structured and unstructured healthcare data, including electronic health records, medical imaging, genomic information, and patient monitoring data. By identifying complex patterns and correlations, these technologies help researchers and clinicians better understand disease progression, predict treatment outcomes, and optimize patient care strategies.
One of the key outcomes of AI and ML adoption is the increased generation of real-world evidence (RWE). Real-world evidence refers to clinical insights derived from real patient data collected outside traditional clinical trials, such as hospital records, insurance claims, wearable devices, and patient registries. AI-driven analytics platforms enable faster processing of this data, allowing healthcare stakeholders to evaluate treatment effectiveness, safety profiles, and long-term patient outcomes in real-world settings. The combination of AI/ML technologies and real-world data supports more informed decision-making across drug development, regulatory evaluation, and clinical practice.
Barrier Analysis – Data Privacy, Interoperability, and High Implementation Costs
Healthcare systems generate large volumes of sensitive patient data, including medical records, genetic information, and diagnostic reports. Ensuring the security and confidentiality of this data is critical, as breaches can lead to legal, ethical, and financial consequences for healthcare organizations. Strict regulatory requirements also make data management and sharing more complex. Interoperability is another major challenge because healthcare data is often stored across multiple platforms, systems, and institutions that may not communicate effectively with each other.
This lack of standardization can limit seamless data exchange, slowing clinical decision-making and research collaboration. Implementing advanced digital health platforms, artificial intelligence tools, and integrated data systems requires substantial financial investment. Costs related to infrastructure, software, data storage, cybersecurity, and staff training can be significant, making adoption difficult for smaller healthcare providers and research organizations.
Lack of Standardized Validation and Regulatory Frameworks
Rapid expansion of digital health and AI-driven technologies has created challenges related to consistent validation standards and regulatory oversight. Government agencies are still developing frameworks to evaluate the safety and performance of these technologies across their lifecycle. According to the U.S. Food and Drug Administration (FDA), it reported that nearly hundreds of AI-enabled medical devices had been authorized for market by 2024, reflecting rapid technological adoption while regulatory policies continue to evolve.
Gaps in standardized validation remain significant. An analysis of FDA-approved AI medical devices found that 97% relied primarily on retrospective evaluations, and only 37% of approval documents reported sample size information, limiting transparency and consistent performance assessment. These limitations highlight the need for harmonized global regulatory frameworks and standardized validation protocols to ensure reliability, safety, and fairness of emerging healthcare technologies.
Opportunity Analysis – Growth in Oncology and Surgical Digital Twins
The increasing burden of cancer and the need for more precise surgical planning are driving the adoption of digital twin technologies in oncology and surgical care. According to the World Health Organization (WHO) and its cancer research agency, IARC, around 20 million new cancer cases were reported globally in 2022, and this number is projected to rise to over 35 million cases by 2050, representing a 77% increase in global cancer incidence.
This growing disease burden is encouraging healthcare systems to adopt advanced digital technologies that can simulate patient-specific disease progression and treatment responses. Digital twins create virtual models of patients using clinical data, imaging, and genomic information, allowing oncologists to test multiple treatment strategies before applying them in real clinical settings. Such simulations help optimize chemotherapy, radiotherapy, or immunotherapy plans while minimizing damage to healthy tissues. In surgical care, digital twin models are increasingly used to replicate patient anatomy and simulate complex procedures before surgery. These systems enable surgeons to practice procedures, predict potential complications, and improve surgical accuracy.
Expansion in Academic-Industry Collaborations
Growing partnerships between universities and industry are playing a major role in accelerating innovation, technology transfer, and applied research across healthcare, biotechnology, and advanced manufacturing sectors. Academic institutions provide strong scientific expertise and research infrastructure, while industry partners contribute funding, commercialization capabilities, and real-world problem-solving perspectives. This collaboration helps translate laboratory discoveries into practical products such as medical technologies, therapeutics, and digital health solutions.
Government data highlights the increasing importance of these partnerships. According to the Office of the Principal Scientific Adviser, about 48% of research organizations reported collaborations with domestic industry, demonstrating strong engagement between academic institutions and industrial partners in national research programs. Around 14,000 collaborative research projects were undertaken in 2022–23, reflecting the expanding scale of joint innovation activities.
Category-wise Analysis
Component Insights
The software segment is anticipated to dominate the market, accounting for 65% of the market share in 2026. Its dominance is driven by growing use of advanced simulation platforms, data analytics tools, and artificial intelligence algorithms that enable the creation and management of digital replicas of patients, medical devices, and healthcare systems. Software solutions allow healthcare providers and researchers to model disease progression, predict treatment outcomes, and optimize clinical workflows using real-time data. An example of digital twin technology in healthcare is the digital twin platform developed by Siemens Healthineers.
The company implemented a digital twin software solution for the radiology department of Mater Private Hospital in Dublin. The platform created a virtual model of the department using operational and patient-flow data to simulate workflows, equipment usage, and staffing requirements.
The services segment represents the fastest-growing component, due to the increasing need for specialized support in implementing and managing digital twin technologies. Healthcare organizations often require consulting, system integration, customization, and maintenance services to successfully deploy digital twin platforms. These services help hospitals and research institutions integrate digital twin solutions with existing healthcare IT systems, medical devices, and data sources. IQVIA is a global healthcare analytics and consulting company.
The company offers consulting, system integration, and implementation services that help healthcare and life-science organizations deploy digital twin technologies.
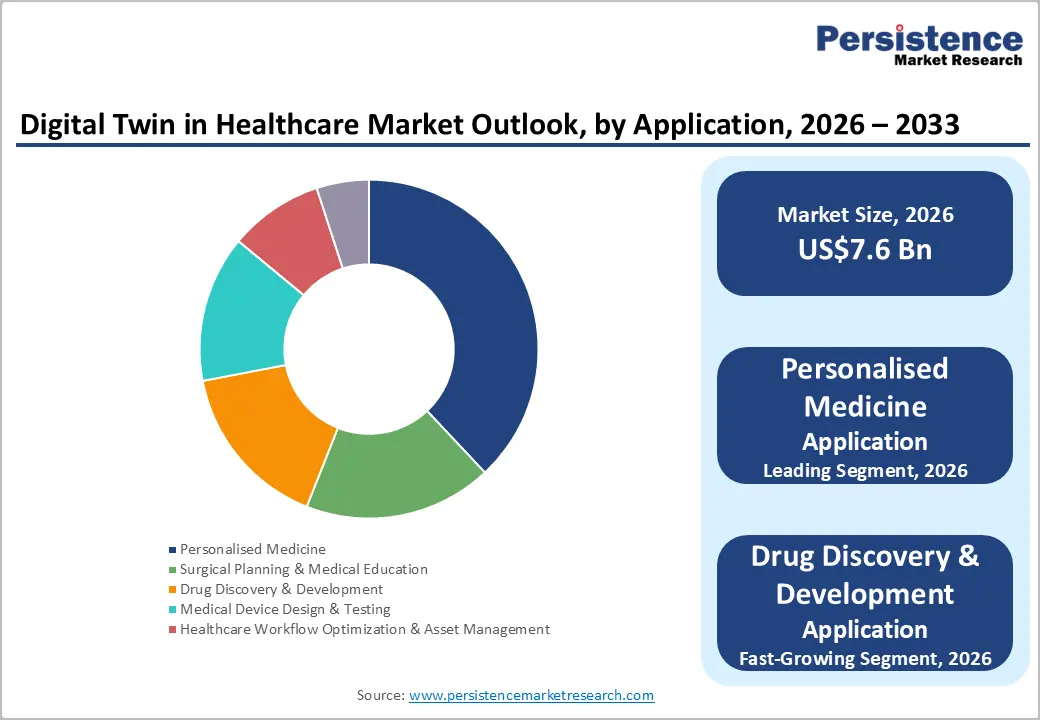
Application Insights
Personalized medicine is expected to dominate the market, contributing nearly 38% of revenue in 2026, propelled by increasing use of patient-specific data such as genetic information, medical history, lifestyle factors, and real-time health monitoring to create accurate digital replicas of individuals. These digital models allow healthcare professionals to simulate disease progression and evaluate potential treatment responses before applying them in real life. By enabling more targeted and effective therapies, personalized medicine helps reduce treatment risks and improve clinical outcomes.
SimBioSys is a healthcare technology company that developed the TumorScope platform. TumorScope creates a digital twin of a patient’s tumor using medical imaging and pathology data to simulate how the cancer may respond to different therapies. This virtual model allows oncologists to test multiple treatment options digitally and identify the therapy most likely to be effective for a specific patient.
Drug discovery & development represents the fastest-growing application, supported by the increasing need for faster and more efficient drug development processes. Digital twin technology allows researchers to create virtual models of biological systems, organs, or patient populations to simulate how potential drugs may interact within the body. These simulations help scientists evaluate drug safety, efficacy, and dosage before moving into costly laboratory or clinical studies.
Digital twin technology in drug discovery and development is the initiative by GlaxoSmithKline (GSK). The company collaborated with Siemens and Atos to create a digital twin of its vaccine manufacturing and research processes. This virtual model allows scientists to simulate experiments, analyze production conditions, and test different variables digitally before performing physical laboratory trials.
Regional Insights
North America Digital Twin in Healthcare Market Trends
North America is projected to lead the market, accounting for nearly 42% of revenue in 2026, driven by its advanced AI ecosystem, high R&D investment, and strong public awareness of precision medicine benefits. The region’s established biotechnology ecosystem, rising demand for biologics, and growing outsourcing by pharmaceutical and biotech companies are driving growth. The U.S. plays a major role, supported by advanced research infrastructure, clear regulatory frameworks, and numerous biotech startups that depend on contract manufacturers for fermentation-based production.
Innovation in digital twin technology, including real-time modeling, improved cloud delivery, and targeted oncology applications, is attracting significant public and private investment. Digital twin systems used for clinical decisions or treatment planning are classified under Software as a Medical Device (SaMD), meaning they must prove safety, effectiveness, and risk control, often with clinical evidence or real-world validation. The FDA’s Digital Health and AI Glossary defines digital twins in this context, emphasizing their role in personalized care and in silico trials.
Europe Digital Twin in Healthcare Market Trends
Market growth in Europe is driven by rising awareness of digital twin benefits, strong regulations, and government-led precision medicine programs. Countries like Germany, France, the U.K., and Switzerland have established healthcare systems that support routine use of digital twins and encourage innovative software approaches. These high-fidelity models are especially useful for personalized medicine, research, and providers who prioritize compliance, improving prediction accuracy, and treatment outcomes.
Technological advancements, such as better integration of multiple data types, targeted application delivery, and improved service quality, are further increasing the market potential. European authorities are actively supporting research and trials for both routine and specialized uses, boosting confidence in the technology. A key regulatory milestone came into force on 1 August 2024: the EU’s AI Act. This regulation applies to high-risk AI systems, including digital twins in healthcare, and requires them to meet strict standards for transparency, safety, quality of data, risk control, and human oversight before deployment. This ensures that digital twin tools are reliable, safe, and effective while protecting patient outcomes and supporting evidence-based care.
Asia Pacific Digital Twin in Healthcare Market Trends
Asia Pacific is expected to be the fastest-growing market, driven by rising precision medicine awareness, increasing government initiatives, and expanding application programs across the region. Countries such as China, India, Japan, and South Korea are actively promoting twin campaigns to address oncology growth and emerging R&D needs. Digital twins in healthcare are particularly attractive in these regions due to their scalable administration, ease of adoption, and suitability for large-scale pharma & biopharma companies and personalized medicine drives in both urban and semi-urban populations.
Technological advancements are supporting the development of stable, effective, and easy-to-deploy digital twins, which can withstand challenging data environments and minimize validation dependence. These innovations are critical for reaching domestic providers and improving overall precision coverage. Growing demand for software, personalized medicine, and pharma & biopharma companies' applications is contributing to market expansion. Public-private partnerships, increased digital health expenditure, and rising investment in twin research and deployment capacity are further accelerating growth. The convenience of twin delivery, combined with improved prediction and reduced risk of treatment failure, positions it as a preferred choice.

Competitive Landscape
The global digital twin in healthcare market is characterized by competition between established healthcare technology companies and emerging simulation-focused innovators. Major technology and medical imaging companies such as Siemens Healthineers and GE HealthCare play a significant role in advancing digital twin platforms through strong hospital partnerships, advanced imaging technologies, and integrated healthcare software ecosystems. Their platforms enable the creation of virtual patient models that support clinical decision-making, treatment simulation, and personalized healthcare planning.
In regions such as North America and Europe, digital twin adoption is supported by advanced healthcare infrastructure and strong research collaboration networks. Companies in Asia Pacific are increasingly developing cost-effective digital twin solutions to improve accessibility and adoption across healthcare systems. Software platforms remain central to this market, enabling predictive analytics, simulation-based testing, and integration with hospital data systems. Strategic partnerships, technology collaborations, and service-based implementation support are helping organizations expand digital twin ecosystems and accelerate real-world deployment in clinical environments.
Key Industry Developments
- In November 2025, Siemens Healthineers launched an AI-enabled radiology services suite to help healthcare providers address challenges ranging from image interpretation to complex healthcare environment planning. The platform covered the entire imaging workflow, from scheduling and image generation to reporting. It integrated existing solutions with new offerings such as AI-Enablement Services, which generated customized summaries of clinically relevant findings for radiologists.
- In March 2025, NVIDIA and GE HealthCare announced a collaboration to develop autonomous diagnostic imaging technologies using physical AI. The partnership focused on advancing autonomous X-ray and ultrasound systems by leveraging the NVIDIA Isaac for Healthcare simulation platform. GE HealthCare used the platform’s pretrained models and physics-based simulations to train, test, and validate imaging systems in virtual environments before real-world deployment.
Companies Covered in Digital Twin in Healthcare Market
- Siemens Healthineers AG
- GE HealthCare.
- Philips N.V.
- IBM Watson Health, Microsoft
- Medtronic
- Oracle
- Honeywell,
- GE Digital
- ANSYS
Frequently Asked Questions
The global digital twin in healthcare market is projected to reach US$7.6 billion in 2026.
Precision medicine and virtual clinical trials acceleration are key drivers.
The digital twin in healthcare market is poised to witness a CAGR of 38.2% from 2026 to 2033.
Oncology and surgical digital twins and expansion in Asia Pacific and academic-industry collaborations are key opportunities.
Siemens Healthineers AG, GE HealthCare, Philips N.V., IBM Watson Health, and Microsoft are the key players.







