- Medical Devices
- Orthopedic Trauma Devices Market
Orthopedic Trauma Devices Market Size, Share, and Growth Forecast for 2025 - 2032
Orthopedic Trauma Devices Market by Product (Internal Fixators, External Fixators), End User (Hospitals, Orthopedic and Trauma Centers, Ambulatory Surgical Centers), and Regional Analysis from 2025 to 2032
Orthopedic Trauma Devices Market Size and Share Analysis
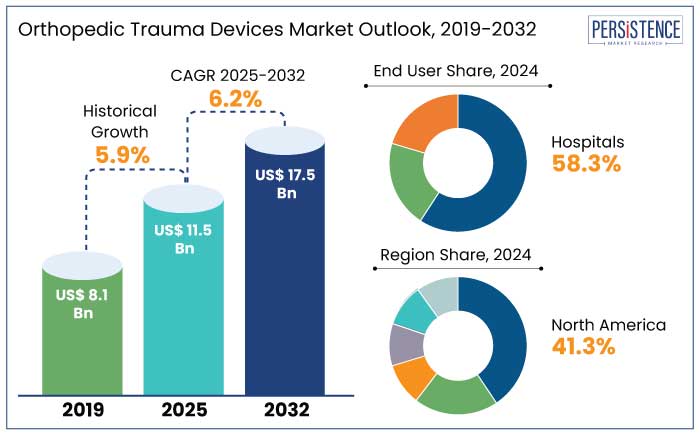
The global orthopedic trauma devices market is estimated to reach a size of US$ 11.5 Bn in 2025. It is predicted to rise at a CAGR of 6.2% through the assessment period to reach a value of US$ 17.5 Bn by 2032.
Increased incidence of fractures, sports-related injuries, and degenerative bone diseases is expected to propel the demand for orthopedic trauma devices.
- According to the WHO, approximately 344 Mn individuals worldwide suffer from osteoarthritis and rheumatoid arthritis, with 73% aged over 55, underscoring the growing demand for effective orthopedic solutions.
Asia Pacific, particularly countries like India and China, is witnessing a surge in sports injuries due to increasing participation in athletic activities, contributing to market expansion. In March 2024, Johnson & Johnson launched its advanced 3D-printed titanium implants for orthopedic trauma, enhancing patient-specific solutions and recovery outcomes.
Advancements in implant materials like bio-ceramics and biocompatible polymers address challenges such as stiffness and iatrogenic injuries. 3D printing technology is transforming therapy by allowing for the development of bespoke implants and anatomical models, improving precision in musculoskeletal care.
Key Highlights of the Market
- The rise in road accidents and sports injuries necessitates advanced trauma treatment solutions and surgical interventions.
- The increasing popularity of minimally invasive surgical procedures is driving a surge in the demand for advanced trauma fixation devices.
- The use of advanced implant materials and designs is revolutionizing surgical outcomes and patient recovery.
- The incidence of osteoporosis and related fractures is on the rise, necessitating the use of specialized trauma treatment devices.
- In 2025, hospitals are estimated to attain a market share of 58.3% due to rising prevalence of injuries and trauma.
- Based on product, the internal fixators are projected to account for a market share of 59.7% in 2025 due to its efficiency.
- North America market share is predicted to reach 41.3% in 2025 due to advancements in MIS devices.
- Asia Pacific is anticipated to hold a market share of 27.4% in 2025 with growing geriatric population.
|
Market Attributes |
Key Insights |
|
Orthopedic Trauma Devices Market Size (2025E) |
US$ 11.5 Bn |
|
Projected Market Value (2032F) |
US$ 17.5 Bn |
|
Global Market Growth Rate (CAGR 2025 to 2032) |
6.2% |
|
Historical Market Growth Rate (CAGR 2019 to 2023) |
5.9% |
Rising Prevalence of Orthopedic Surgeries Offers Vision in North America
North America is projected to hold a 41.3% market share in 2025 with a forecast CAGR of 5.2% during the forecast period, reflecting robust demand for advanced trauma care solutions. The demand for orthopedic trauma devices in North America is poised for significant growth, driven by the rising prevalence of arthritis, back pain, and neck pain.
- The U.S. dominates the region, with an estimated 91.2 Mn individuals affected by arthritis and 54.4 Mn having a medical diagnosis, fueling the demand for fracture fixation tools.
Advancements in minimally invasive spine surgeries, such as the development of specialized orthopedic devices, are enhancing surgical precision and recovery times. In January 2024, Medtronic launched a new MIS spinal fixation system in the U.S., designed to reduce operative time and improve patient outcomes.
The demand for orthopedic procedures is expanding due to the growing geriatric population, increased awareness, and favorable government reimbursement policies.
Advancement in Medical Technology in Asia Pacific Presents Growth Avenues
The orthopedic trauma device market in Asia Pacific is expected to a market share of 27.4% in 2025, due to an aging population and advancements in medical technology, with a projected CAGR of 7.5% through 2032.
- Japan leads the market, with 29.1% of its population aged 65 or older, fueling the demand for knee replacement surgeries and advanced orthopedic solutions.
- In March 2019, Medtronic introduced the Demineralized Bone Matrix (DBM) Grafton bone grafting device in Japan, targeting spine and orthopedic surgeries.
Aging population, industrialization, and urbanization in China are contributing to an increase in traumatic fractures, especially among young and middle-aged patients. Health insurance policies in Asia Pacific countries are improving access to advanced trauma care, driving market growth due to technological innovations.
Surgeons Opt for Internal Fixators Owing to Its Efficiency
Internal fixators, such as rods, screws, metal plates, and wires, are expected to maintain dominance in the global orthopedic trauma devices market, accounting for 59.7% of market share in 2025. These devices ensure proper healing, reduce hospital stays, and expedite patient recovery.
- According to the American Academy of Orthopaedic Surgeons, over 6.3 Mn fractures occur annually in the U.S., highlighting the need for effective internal fixation devices.
- In February 2024, DePuy Synthes, a subsidiary of Johnson & Johnson, introduced its Variable Angle Locking Compression Plate System, offering enhanced versatility for complex fractures.
Advanced materials like titanium and bio-absorbable polymers are transforming orthopedic care, enhancing the durability, compatibility, and ease of use of internal fixators for better patient outcomes.
Hospitals Cultivates the Demand for Devices Owing to Prevalence of Injuries and Trauma
Hospitals are projected to maintain a 58.3% share of the global market in 2025. The increasing global prevalence of injuries and trauma, estimated by the WHO to be over 50 Mn non-fatal injuries annually, is propelling the hospitals segment.
Advancements in technology and minimally invasive procedures have made hospitals the preferred location for orthopedic surgeries. Hospitals benefit from reimbursement policies that ease the financial burden on patients, further driving procedure volumes.
- For instance, in February 2024, the U.S. Centers for Medicare & Medicaid Services expanded its reimbursement list to include advanced trauma fixation techniques, enhancing patient accessibility.
The presence of trauma centers and highly skilled medical professionals ensures timely and effective treatment, cementing hospitals' critical role in the orthopedic devices market. The integration of digital imaging and robotic-assisted surgery is enhancing the efficiency and outcomes of orthopedic care.
Market Introduction and Trend Analysis
The global orthopedic trauma devices market is propelling due to the rising incidence of road accidents and sports-related injuries. According to the WHO, road traffic injuries claim 1.3 Mn lives annually and cause up to 50 Mn non-fatal injuries, creating demand for trauma devices.
The aging population, especially in Japan and Italy, has increased osteoporosis and fracture cases, thereby boosting the demand. Technological developments have improved surgical results and durability, such as titanium-based implants and biocompatible materials. For example,
- In March 2024, Stryker introduced a new line of titanium-coated plates designed for faster healing and reduced infection rates.
The adoption of 3D printing in customized orthopedic implants is also gaining traction, offering patient-specific solutions. Improved healthcare infrastructure in emerging regions like India and Brazil and expanded insurance coverage in developed regions are facilitating access to advanced trauma care.
Historical Growth and Course Ahead
The global orthopedic trauma maneuvers market recorded a CAGR of 5.9% in the historical period from 2019 to 2023. Implants for bone fixation are often used in orthopedics, and the materials used have changed from being inert to matching the form of bone. For external fixation, bioactive, biocompatible, and absorbable materials are utilized to preserve fracture length, alignment, and rotation all of which are critical for trauma centers.
External fixation is crucial for fracture treatment due to its rapid application, reduced blood loss, and minimally invasive nature. It minimizes injection risk and wound complications by allowing time for soft tissue swelling in severe injuries.
An increase in small fractures, major injuries, accidents, deaths, trauma from traffic accidents, falls, osteoarthritis, and orthopedic sports injuries is expected to boost the need for external fixation worldwide. Demand for orthopedic pain devices is estimated to record a considerable CAGR of 6.2% during the forecast period between 2024 and 2032.
Market Growth Drivers
Surge in Adoption of Minimally Invasive Surgical Procedures Drive the Demand
The increasing use of minimally invasive surgical procedures is driving a surge in the demand for advanced trauma fixation devices. According to a 2023 report by the National Center for Biotechnology Information, MIS procedures in orthopedics have increased by 22% in the past five years. This shift has spurred innovation in trauma fixation devices designed for precise placement through smaller incisions.
- For example, in April 2023, Stryker launched the VariAx 2 Mini Fragment System, an advanced plate and screw system tailored for MIS in small bone fractures.
- Zimmer Biomet introduced the Periarticular Locking Plate System in January 2023, optimized for minimally invasive techniques in complex fractures.
The trend towards MIS is transforming orthopedic trauma care, promoting the use of advanced, biocompatible fixation technologies for cost efficiency and improved patient outcomes.
Rising Road Accidents and Sports Injuries Require Advanced Trauma Treatments
The increasing incidence of road accidents and sports injuries is driving the demand for advanced trauma treatment solutions and surgical interventions. The World Health Organization reports that road traffic accidents cause around 1.3 Mn deaths annually and cause injuries to 20 to 50 Mn people globally.
A 2023 report by the American Academy of Orthopaedic Surgeons (AAOS) indicates a 15% rise in ligament and fracture injuries among athletes, underscoring the need for effective trauma management.
- Stryker's T2 Alpha Tibial Nailing System, introduced in March 2023, is a technological advancement in orthopedic trauma devices, enhancing surgical precision and recovery times for complex tibial fractures.
- Zimmer Biomet introduced the Zyston Strut Open Titanium System in April 2023 for spinal trauma, enhancing strength and flexibility to improve patient outcomes in trauma care.
Market Restraining Factors
Limitations of Fixators Hinder the Adoption
For long-bone fractures, external fixation or joint replacements may be a better option than internal fixation, depending on the location. These techniques do have certain drawbacks, though, such as the possibility of infection and the size of the functional output. Additionally, joint contractures, a longer bone-healing period, and an elevated risk of infection at the sites where pins and wires enter the body might result from external fixators.
Orthopedic implants must be made of carefully selected materials to prevent bone fractures, abnormalities, or insufficient healing. The three biomaterials utilized to treat bone deformities and fractures are ceramics, metals, and polymers; each has certain benefits and drawbacks for its intended usage.
Key Market Opportunities
Adoption of 3D Printing Reshapes the Orthopedic Applications
3D printing technology is transforming the demand for orthopedic trauma devices by producing bespoke implants based on the anatomy of each patient to improve surgical results by guaranteeing a perfect fit and a quicker recovery. According to a 2023 report by the American Academy of Orthopaedic Surgeons, 3D printing applications in orthopedics grew by 18% over the prior year.
- Additive Orthopaedics launched the Patient-Specific Talus Spacer in May 2023, the first FDA-approved 3D-printed implant for complex ankle injuries.
- Johnson & Johnson introduced the TRUMATCH Graft Cage System in February 2023, enabling surgeons to create personalized spinal trauma solutions, reducing production lead times and costs.
Advancements in Implant Materials and Designs Enhance Surgical Outcomes
The use of advanced implant materials and designs is reforming orthopedic trauma care, improving surgical outcomes, and expediting patient recovery. Modern implants, crafted from advanced materials like titanium alloys and bio-ceramics, provide superior strength, biocompatibility, and reduced infection risk.
- A 2023 study by the Journal of Orthopedic Science highlighted that titanium implants reduce postoperative complications by 18% compared to traditional stainless steel alternatives.
Implants made with 3D printing may be precisely customized to meet the anatomy of the patient, improving fit and restorative functionality.
- For instance, in February 2023, Johnson & Johnson launched its ATTUNE Cementless Knee System with advanced material and design features, enhancing long-term implant stability.
- Medtronic's Adaptix Interbody System, introduced in April 2023, uses titanium lattice technology for improved bone fusion, reducing recovery times and enhancing the quality of life for orthopedic trauma patients globally.
Competitive Landscape for the Orthopedic Trauma Devices Market
Businesses must innovate and set themselves apart in the highly competitive orthopedic trauma device market by utilizing cutting-edge materials to enhance performance and durability. To increase their market presence and reach more people, they are also developing strategic alliances and growing their distribution networks.
Start-ups in the sector are focusing on innovative developments, such as creating personalized implant materials and 3D printing technologies for personalized medical solutions. The trend towards minimally invasive surgical techniques is a significant shift, as these methods can improve patient outcomes and speed up recovery times.
An inventive pioneer in the sector is Spentys, a Belgian-based company that specializes in orthopedic immobilization devices. They provide each patient with customized orthoses, combining cost-effectiveness with individualized attention to improve patient comfort and the effectiveness of orthopedic therapies.
Recent Industry Developments
- In September 2024, Orthofix Medical launched the Galaxy Fixation Gemini system, a stable external fixation for treating fractures from trauma. It eliminates tray sterilization and space constraints, making it ideal for trauma bays and intensive care units.
- In September 2024, Arthrex launched OrthoPedia Patient, an online learning tool for orthopedic conditions and treatments. The platform, developed by clinical specialists and peer-reviewed, encourages self-directed learning and is regularly updated to reflect the latest research and treatment standards.
- In September 2024, Temple OrthoBiologics, a new venture from Temple Therapeutics, launched a breakthrough in orthopedic sports medicine: TX-33. This breakthrough uses advanced biomaterials, targeted delivery, and a compound for muscle recovery and repair.
- In August 2024, Stryker introduced the Pangea Plating System, FDA-cleared for bone fractures, osteotomies, and arthrodesis. It enhances plate fit and screw placement, elevating the plating market with anatomically contoured implants.
- In August 2024, DePuy Synthes introduced TriLeap, a low-profile titanium plate system for foot and ankle surgeons. The FDA cleared it in 2023, and it addresses big toe and first toe bone conditions.
- In February 2023, CurvaFix, a medical device developer from Washington, launched the 7.5mm CurvaFix IM Implant for small-boned patients, simplifying surgery and providing strong, stable fixation.
Companies Covered in Orthopedic Trauma Devices Market
- Johnson & Johnson Services, Inc.
- Zimmer Biomet Holdings, Inc.
- Stryker Corporation
- Smith & Nephew
- B. Braun Melsungen AG
- Wright Medical Group N.V.
- Orthofix International N.V.
- Integra LifeSciences
- Citieffe S.R.L.
- Acumed
Frequently Asked Questions
The market is set to reach US$ 17.5 Bn by 2032.
Orthopedic trauma injuries are prevalent in falls, car crashes, bicycle accidents, gunshot wounds, and motorized scooter accidents.
The market is estimated to be valued at US$ 11.5 Bn in 2025.
Johnson & Johnson Services, Inc., Zimmer Biomet Holdings, Inc., Stryker Corporation, and Smith & Nephew are a few key players.
Advancements in implant materials and designs provides a key opportunity for the market players.








