- Pharmaceuticals
- Specialty Pharmaceuticals Market
Specialty Pharmaceuticals Market Size, Share, and Growth Forecast, 2025 - 2032
Specialty pharmaceuticals market by Product Type (Biologics, Orphan Drugs, Gene Therapies, Precision Medicines), Therapeutic Area (Oncology, Autoimmune Diseases, Central Nervous System Disorders, Infectious Diseases, Haemophilia), Route of Administration (Injectable, Oral, Intravenous, Transdermal), End-use (Hospitals, Specialty Clinics, Homecare Settings, Pharmacies), and Regional Analysis for 2025 – 2032
Specialty Pharmaceuticals Market Size and Trends Analysis
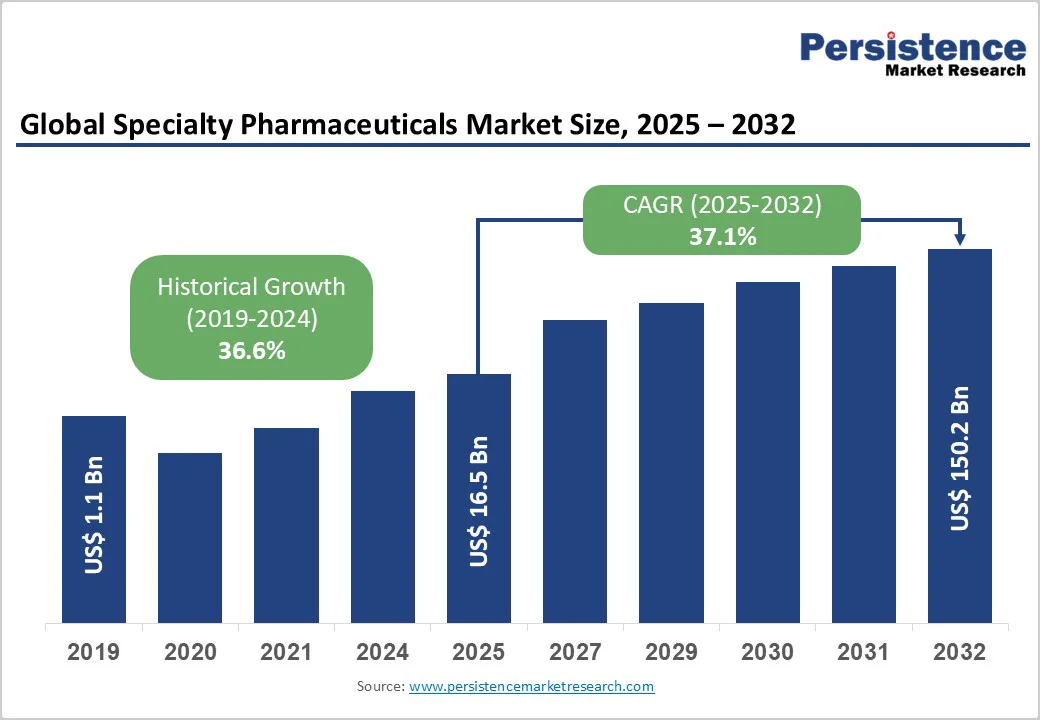
The global specialty pharmaceuticals market size is likely to be valued US$ 16.5 billion in 2025, forecast to reach US$ 150.2 billion by 2032 achieving at a CAGR of 37.1% during the forecast period from 2025 to 2032. The market is experiencing robust growth driven by increasing prevalence of chronic and rare diseases, advancements in biologics and gene therapies, and rising demand for personalized medicine. The growing acceptance of specialty pharmaceuticals as critical solutions for rare and chronic conditions, supported by regulatory incentives, is a key growth factor.
Key Industry Highlights:
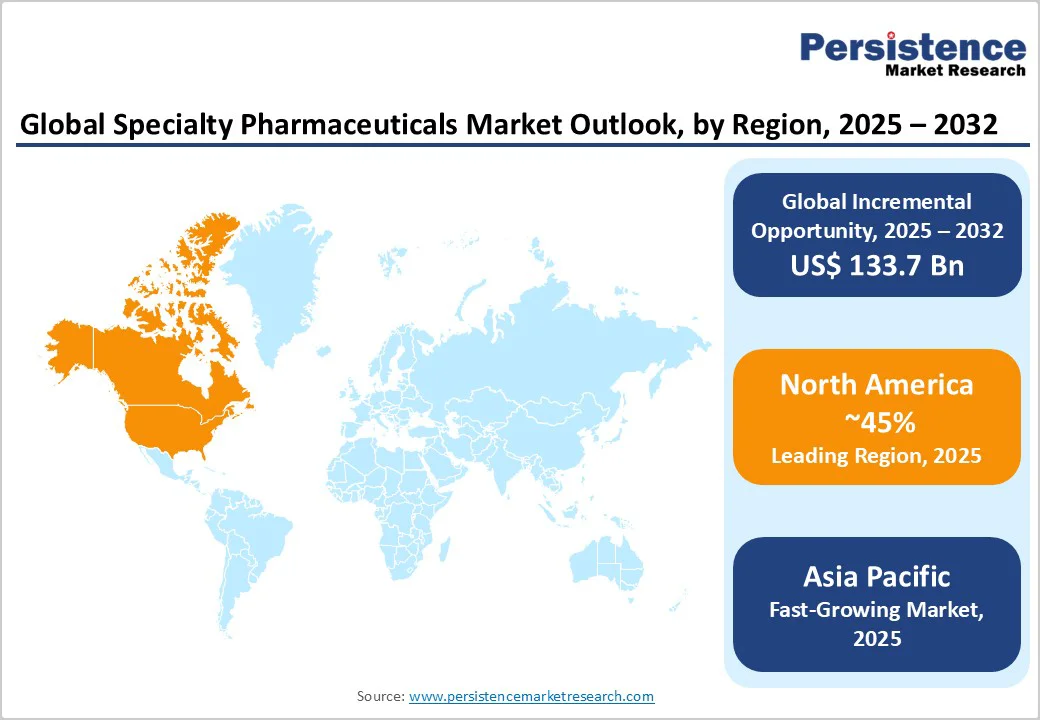
- Leading Region: North America, commanding a 45% market share in 2025, driven by advanced healthcare infrastructure and R&D investments in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by increasing healthcare access and rising chronic disease prevalence in China and India.
- Dominant Product Type: Biologics, holding approximately 50% share, due to their efficacy in treating complex diseases.
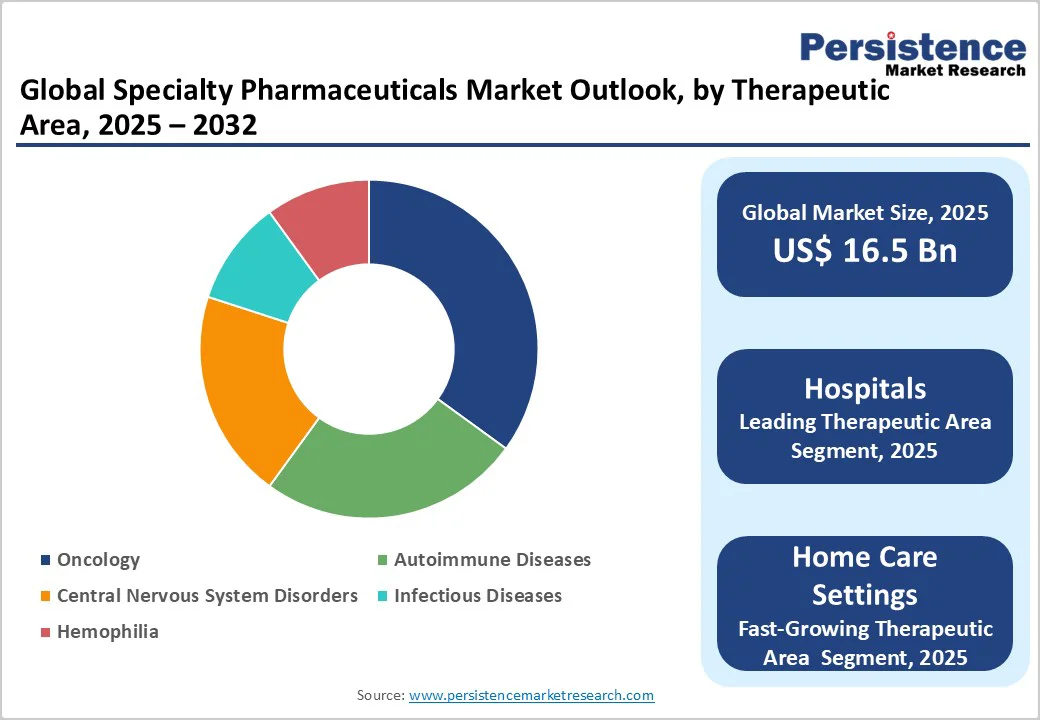
- Leading Therapeutic Area: Oncology, accounting for over 40% of market revenue, driven by rising cancer cases globally.
- Key Market Driver: Aging populations, lifestyle changes, and better diagnostics are increasing the incidence of chronic and rare conditions, driving demand for innovative treatments and personalized therapies.
- Growth Opportunity: Expansion of gene therapies for rare diseases, supported by regulatory fast-tracking.
- Market Trend: Adoption of AI-driven drug development for personalized therapies.
|
Key Insights |
Details |
|
Specialty Pharmaceuticals Market Size (2025E) |
US$ 16.5 Bn |
|
Market Value Forecast (2032F) |
US$ 150.2 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
37.1% |
|
Historical Market Growth (CAGR 2019 to 2024) |
36.6% |
Market Dynamics
Driver - Rising prevalence of chronic and rare diseases
The rising prevalence of chronic and rare diseases is a major driver shaping healthcare, pharmaceuticals, and biotechnology markets worldwide. Chronic diseases, such as diabetes, cardiovascular disorders, and respiratory conditions, are increasing due to aging populations, urbanization, sedentary lifestyles, unhealthy diets, and environmental factors. These conditions require long-term management, frequent monitoring, and continuous medication, creating sustained demand for innovative treatments, advanced therapeutics, and patient care solutions.
Rare diseases, though affecting smaller populations individually, collectively impact millions globally. Advances in diagnostics and genetic research have led to higher detection rates of these conditions, while increased awareness among patients and healthcare providers has further highlighted unmet medical needs. The complexity of these diseases often necessitates personalized treatments, including biologics, gene therapies, and targeted therapies, which are reshaping the pharmaceutical and biotech landscapes.
Restraint - High development costs and complex reimbursement policies
High development costs and complex reimbursement policies pose significant challenges for many industries, particularly in healthcare, pharmaceuticals, and advanced medical technologies. Developing innovative products, such as biologics, gene therapies, or specialized medical devices, requires substantial investment in research and development (R&D), clinical trials, regulatory compliance, and manufacturing infrastructure. These processes are not only capital-intensive but also time-consuming, often taking several years before a product reaches the market.
In addition to the financial burden, complex reimbursement policies add another layer of difficulty. Companies must navigate a patchwork of insurance guidelines, government regulations, and payer criteria to ensure their products are covered and adequately reimbursed. Variability in reimbursement rates across regions or patient populations can significantly impact profitability and market adoption. Unclear or restrictive policies may delay patient access, reducing the incentive for investment in high-cost innovations.
Opportunity - Expansion in gene therapies and precision medicine
The specialty pharmaceuticals market is experiencing significant expansion driven by advances in gene therapies and precision medicine, which are transforming treatment approaches for complex and rare diseases. Gene therapies, designed to target the underlying genetic causes of disorders, offer potential curative solutions rather than symptom management. Recent breakthroughs in viral and non-viral vector technologies, CRISPR-based gene editing, and cell-based therapies have accelerated development pipelines, attracting substantial investment from both pharmaceutical companies and venture capital firms. This has led to a growing number of approved and late-stage gene therapy products addressing conditions such as hemophilia, spinal muscular atrophy, and inherited retinal disorders.
Precision medicine complements this growth by enabling highly individualized treatment strategies based on a patient’s genetic, molecular, and biomarker profiles. Tailoring therapies to specific patient subgroups enhances efficacy, minimizes adverse effects, and improves overall outcomes. Integration of precision diagnostics, genomic sequencing, and data analytics further facilitates the identification of suitable candidates for advanced therapies.
Category-wise Analysis
Product Type Insights
Biologics dominate the market, accounting for 50% of the share in 2025, due to their high efficacy in managing complex conditions like cancer and autoimmune diseases. These therapies offer targeted, personalized treatment options with improved outcomes. Continuous innovation in monoclonal antibodies, recombinant proteins, and immune-modulating biologics further reinforces their leading position in the market.
Gene Therapies are the fastest-growing segment, driven by breakthroughs targeting rare and genetic disorders. These therapies offer the potential for curative treatments by addressing the underlying genetic causes rather than just symptoms. Advances in vector technology, precision delivery, and regulatory support accelerate adoption, making gene therapies a transformative and rapidly expanding area within the specialty pharmaceuticals market.
Therapeutic Area Insights
Oncology leads with over 40% share, driven by the rising prevalence of cancer worldwide. The growth is supported by advanced targeted therapies, immunotherapies, and personalized treatment approaches that improve patient outcomes. Increased awareness, early diagnosis, and expanded access to innovative cancer treatments further reinforce oncology’s leading position in the specialty pharmaceuticals sector.
Autoimmune Diseases are the fastest-growing, fueled by rising diagnosis rates and growing awareness among patients and healthcare providers. Innovations in biologics, including targeted antibodies and immune-modulating therapies, offer more effective and personalized treatment options. This combination of increased prevalence and advanced therapies drives higher demand and rapid market expansion in this segment.
Route of Administration Insights
Intravenous holds nearly 45% share, benefiting from its ability to deliver biologics and gene therapies with precision and controlled dosing. IV administration ensures rapid therapeutic action, accurate bioavailability, and suitability for complex treatments, making it the preferred choice in hospitals and specialized clinics for managing chronic, severe, and rare medical conditions.
Oral is the fastest-growing, driven by patient preference for non-invasive, convenient treatment options. Oral therapies enhance adherence by reducing hospital visits and injection-related discomfort, while advancements in formulation technology improve bioavailability and efficacy. This shift toward patient-centric care supports broader adoption of specialty drugs in both homecare and outpatient settings.
End-use Insights
Hospitals lead with over 40% share, due to their capacity to deliver complex therapies for chronic and severe conditions. Equipped with advanced treatment facilities, specialized healthcare professionals, and robust monitoring systems, hospitals provide the infrastructure necessary for administering biologics, gene therapies, and high-cost specialty drugs, ensuring effective patient care and optimized treatment outcomes.
Homecare Settings are the fastest-growing, driven by increasing adoption of home infusion therapies. Patients with chronic or rare conditions prefer receiving treatments at home for convenience, comfort, and reduced hospital visits. Advances in portable infusion devices, telehealth monitoring, and patient support programs further support this shift, enabling safe, effective, and patient-centric care outside traditional clinical settings.
Regional Insights
North America Specialty Pharmaceuticals Market Trends
North America is accounting for 45% in 2025, driven by U.S. R&D investments and FDA approvals for biologics. The region benefits from a well-established regulatory environment, with the U.S. Food and Drug Administration (FDA) facilitating approvals for innovative therapies, particularly biologics, gene therapies, and precision medicines. These approvals enable faster patient access to high-value treatments for rare, chronic, and complex diseases.
Strong healthcare infrastructure, advanced diagnostic capabilities, and high per capita healthcare spending further support market growth. The focus on personalized medicine and targeted therapies encourages the development of treatments tailored to specific patient populations, increasing the demand for specialty pharmaceuticals. Interestingly, the U.K. market, while geographically part of Europe, reflects similar dynamics, with rising adoption driven by the National Health Service’s support for rare disease treatments and precision medicine programs.
Europe Specialty Pharmaceuticals Market Trends
Europe holds about 30% market share, led by driven by countries such as Germany and France, which benefit from robust healthcare systems, high per capita healthcare spending, and strong regulatory support for innovative therapies. A key factor contributing to Europe’s dominance is the European Union’s orphan drug framework, which provides incentives such as market exclusivity, accelerated approval processes, and financial support for the development of treatments for rare diseases. These policies encourage pharmaceutical companies to invest in research and development of specialty drugs, particularly for conditions with unmet medical needs.
Germany and France have well-established reimbursement pathways and strong public and private healthcare funding, enabling rapid patient access to advanced therapies such as biologics, gene therapies, and targeted oncology treatments. Europe’s emphasis on personalized medicine, growing prevalence of chronic and rare diseases, and collaborative initiatives between industry and healthcare institutions further bolster market growth.
Asia Pacific Specialty Pharmaceuticals Market Trends
Asia Pacific commands around 20% share and is the fastest-growing region, fueled by several interrelated factors, including expanding healthcare infrastructure, rising disease prevalence, and supportive government policies. In China, comprehensive healthcare reforms have enhanced access to advanced therapies, streamlined regulatory approvals, and incentivised innovation, thereby creating a conducive environment for the adoption of speciality pharmaceuticals. India is witnessing a growing burden of chronic and complex diseases, particularly cancer, which drives demand for targeted therapies such as biologics, oncology drugs, and gene therapies.
Rising awareness among patients and healthcare providers regarding early diagnosis and advanced treatment options further contributes to market expansion. Additionally, increasing investments by multinational pharmaceutical companies in local research, manufacturing, and distribution networks enhance the availability and accessibility of specialty drugs across the region. The convergence of government support, rising healthcare expenditure, and the need for personalized, high-value therapies positions Asia Pacific as a crucial market for future growth, offering substantial opportunities for both domestic and international players in the specialty pharmaceuticals segment.
Competitive Landscape
The global specialty pharmaceuticals market is highly competitive, dominated by a limited number of large multinational companies that focus on high-value, complex therapies. These companies prioritize innovative biologics and gene therapies, targeting rare, chronic, and difficult-to-treat conditions where conventional treatments are insufficient. Biologics, including monoclonal antibodies, recombinant proteins, and cell therapies, form the backbone of specialty portfolios due to their ability to address specific molecular pathways and provide personalized treatment options.
Gene therapies are emerging as transformative solutions, offering the potential to correct underlying genetic disorders rather than merely managing symptoms, which is driving significant research investment. To strengthen their market positions and expand therapeutic capabilities, leading players actively pursue strategic acquisitions, partnerships, and licensing agreements. These moves enable access to novel technologies, specialized manufacturing capabilities, and established patient networks. Additionally, companies focus on improving patient access through specialized distribution channels, patient support programs, and innovative pricing strategies.
Key Developments
- In October 2025, Novo Nordisk entered into a licensing agreement with U.S.-based biotech firm Omeros, valued at up to $2.1 billion. This deal grants Novo Nordisk exclusive global rights to develop and commercialize Omeros' experimental drug, zaltenibart, which targets the alternative complement pathway. Zaltenibart is being developed for rare blood and kidney diseases, including paroxysmal nocturnal hemoglobinuria (PNH).
- In October 2024, BioCryst Pharmaceuticals announced a strategic acquisition of Astria Therapeutics in a deal valued at approximately $700 million. This acquisition brings Astria’s lead drug candidate, navenibart, into BioCryst’s pipeline. Navenibart is an injectable therapy in late-stage trials aimed at preventing HAE, with data expected in early 2027. Its dosing schedule of every three to six months could make it a more convenient alternative to current treatments.
Companies Covered in Specialty Pharmaceuticals Market
- Roche
- Amgen
- Johnson and Johnson
- Merck and Co
- Pfizer
- Teva Pharmaceuticals Industries
- GlaxoSmithKline
- Bristol Myers Squibb
- Vertex Pharmaceuticals
- AbbVie
- Eli Lilly and Company
- Sanofi
- Novartis
- AstraZeneca
- Gilead Sciences
Frequently Asked Questions
The global specialty pharmaceuticals market is projected to reach US$ 16.5 Bn in 2025, driven by chronic and rare disease treatments.
The complexity of these diseases promotes investment in research, development, and specialized healthcare technologies to improve patient outcomes and quality of life.
The market is poised to witness a CAGR of 37.1% from 2025 to 2032, supported by gene therapy innovations.
Expansion in gene therapies and precision medicine offers opportunities for specialty pharmaceuticals in rare disease treatments.
Roche, Pfizer, Novartis, Amgen, and Vertex Pharmaceuticals lead through innovative specialty pharmaceuticals for chronic conditions.








