- Medical Devices
- Enteral Feeding Devices Market
Enteral Feeding Devices Market Size, Share, Growth, and Regional Forecast for 2025 - 2032
Enteral Feeding Devices Market by Product (Enteral Feeding Pumps, Enteral Feeding Tubes, Enteral Syringes, Gravity Feeding Sets, and Other Consumables), Application, Age Group, End User, and Region for 2025 - 2032
Enteral Feeding Devices Market Share and Trends Analysis
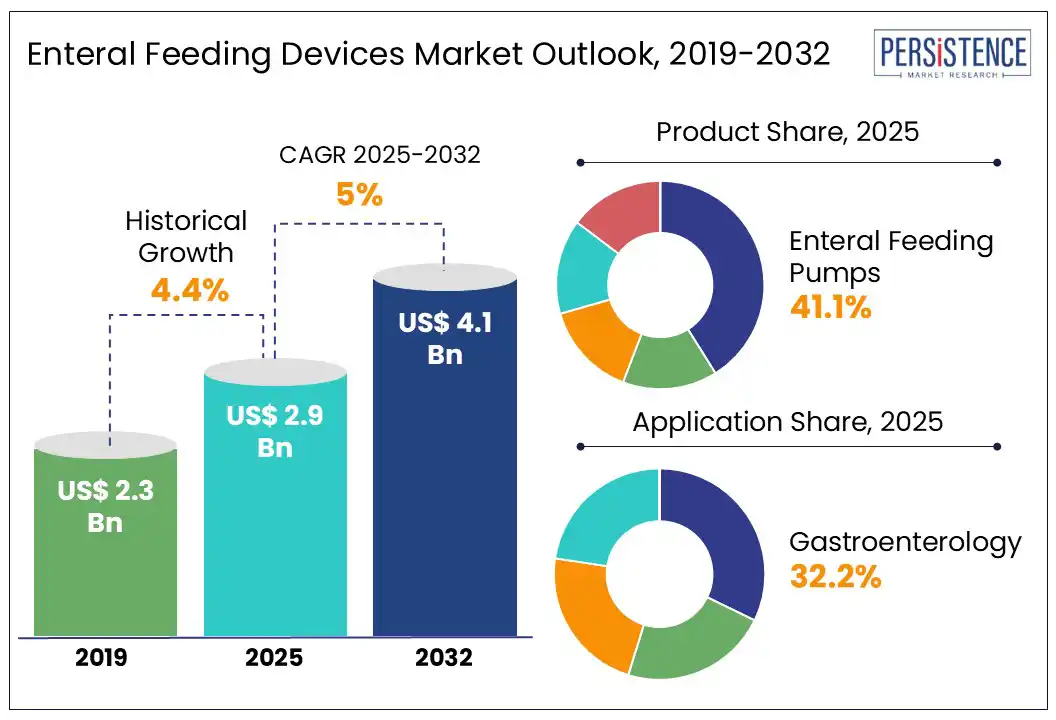
The global enteral feeding devices market size is expected to increase from US$ 2.9 Bn in 2025 to US$ 4.4 Bn by 2032. It is projected to witness a CAGR of 5% from 2025 to 2032. The rise in chronic conditions, ICU admissions, and aging-related dysphagia drives the need for enteral feeding devices. According to the WHO, the global population aged 60 and above is expected to reach 2.1 billion by 2050, resulting in a significant increase in long-term nutritional care needs. Advances such as ENFit connectors and portable feeding pumps are also accelerating adoption across hospitals, home care, and oncology care settings.
Key Industry Highlights:
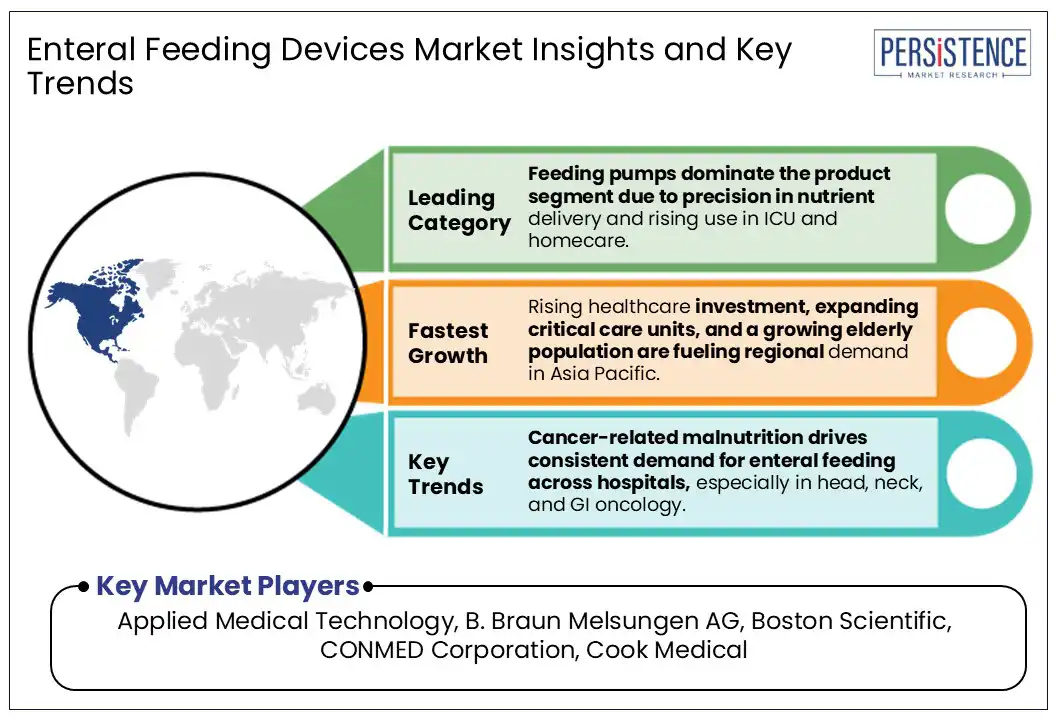
- Leading Region: Rising healthcare investment, expanding critical care units, and a growing elderly population are fueling regional demand for enteral feeding devices in Asia Pacific.
- Demand for Long-term Enteral Feeding: The growing global elderly population is fueling long-term enteral feeding demand, particularly in geriatric care and home settings.
- Leading Product Type: Feeding pumps dominate the product segment due to precision in nutrient delivery and rising use in ICU and homecare.
- Growing preference for home-based recovery has led to increased use of portable feeding pumps and syringes designed for outpatient and homecare support.
- Cancer-related malnutrition drives consistent demand for enteral feeding across hospitals, especially in head, neck, and GI oncology.
- Portable devices and caregiver-friendly features are accelerating enteral feeding device uptake in home-based nutritional therapy.
|
Global Market Attribute |
Key Insights |
|
Enteral Feeding Devices Market Size (2025E) |
US$ 2.9 Bn |
|
Market Value Forecast (2032F) |
US$ 4.4 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
5% |
|
Historical Market Growth (CAGR 2019 to 2024) |
4.4% |
Market Dynamics
Drivers - Rising Prevalence of Gastrointestinal Disorders Globally is Significantly Boosting the Demand for Enteral Feeding Tubes
The rising global burden of gastrointestinal (GI) disorders is significantly driving demand for enteral feeding tubes. Recent estimates from the GIVES-21 Study (2023) project that over 7 million individuals will be living with IBD by 2030 in North America and Europe alone, underscoring the condition’s growing prevalence. These conditions often impair digestion and nutrient absorption, creating a strong need for long-term enteral nutrition.
Manufacturers are innovating more advanced devices. In 2023, Fortune Medical launched a new line of ENFit-compatible enteral feeding tubes, including silicone PEG and nasogastric tubes featuring anti-clog tips, reinforced walls, and low-profile designs aimed at minimizing discomfort and improving usability in homecare and clinical settings. The launch also aligned with international safety standards, addressing growing concerns around misconnections. As chronic GI illnesses continue to rise, the adoption of such advanced feeding tubes is becoming integral to nutritional support strategies across hospitals, long-term care, and palliative treatment settings.
Restraint - Tube Dislodgement and Clogging in Pediatric Patients Challenges Widespread Adoption of Enteral Feeding Devices
Complications such as tube dislodgement and clogging in pediatric patients significantly hinder the widespread adoption of enteral feeding devices. Pediatric studies have reported tube dislodgement rates as high as 40%, posing serious concerns for safe and effective long-term use. Dislodged tubes not only interrupt nutritional support but also increase the risk of infection, trauma, and the need for repeated clinical interventions. Clogging, especially in smaller-diameter pediatric tubes, further disrupts feeding schedules and adds more responsibility to the caregiver. These challenges make clinicians and families more cautious about initiating or continuing enteral nutrition, particularly in home care and outpatient settings.
Opportunity - Increasing Adoption of Home Enteral Nutrition Presents Strong Growth Opportunities for Device Makers Offering Portable Feeding Pumps
The growing shift toward home-based care is driving strong demand for portable enteral feeding devices, offering a significant opportunity for manufacturers. Home Enteral Nutrition is increasingly preferred for managing chronic conditions such as cancer, neurological disorders, and gastrointestinal diseases outside hospital settings. For instance, Geratherm Medical launched a battery-operated portable feeding pump in May 2024 tailored for home use. Similarly, Abbott’s FreeGo® pump, designed for mobility, allows patients to carry their nutrition in a backpack. Cardinal Health’s Kangaroo OMNI™ pump is approved for both hospital and homecare settings, reflecting the trend toward device versatility.
Category Wise Insights
Product Insights
Enteral feeding pumps remain the most in-demand product category, primarily due to their critical role in delivering precise and automated nutrition support. These pumps ensure accurate flow rates, reduce the risk of human error, and support continuous or intermittent feeding, especially vital for critically ill, elderly, or pediatric patients. Their widespread adoption in hospitals, long-term care facilities, and increasingly in homecare settings reflects the global push for safe, consistent, and efficient enteral nutrition. Technological advancements such as programmable features, portability, and alarm systems further enhance their clinical utility and reinforce their market leadership.
In early 2024, Vygon introduced the EasyMoov6 enteral feeding pump, designed for hospital and home use. Featuring a digital touchscreen, peristaltic rotary motor, and intuitive alarms, it supports continuous or blended diets and prioritizes ease and safety for clinicians and caregivers
Application Insights
Oncology patients account for the largest application segment in the global market due to their consistent need for nutritional support during chemotherapy, radiotherapy, and post-surgical recovery. Cancer treatments often impair a patient’s ability to consume food orally, leading to high rates of malnutrition. According to the U.S. National Cancer Institute, between 30% and 85% of cancer patients experience malnutrition during treatment.
Boston Scientific announced its upcoming Intera 3000 infusion pump, intended to support targeted cancer therapy and nutritional access. With the rising global cancer burden and longer survival rates, the need for reliable, effective enteral feeding devices in oncology care continues to grow rapidly across hospitals and home care settings.
Regional Insights and Trends
North America Enteral Feeding Devices Market Trends
The transition to ENFit-compliant connectors in North America is significantly driving safety-focused innovation in the global market. ENFit connectors, introduced as part of the ISO 80369-3 standard, are designed to prevent misconnections between enteral devices and other clinical systems such as IV lines. Their adoption has prompted manufacturers to redesign feeding tubes, syringes, and accessories to ensure compatibility and reduce medical errors. For instance, Fortune Medical launched ENFit-compatible silicone gastrostomy and nasojejunal tubes in 2023, designed to enhance patient safety and reduce misconnections. These low-profile tubes feature anti-clog tips and reinforced walls for durability.
Hospitals and homecare providers are increasingly mandating ENFit-compliant devices, accelerating market demand. This shift not only enhances patient safety but also fosters innovation in connector ergonomics, labeling, and usability, reinforcing North America's leadership in enteral safety standards.
Europe Enteral Feeding Devices Market Trends
Strong government focus on elderly nutritional care across Europe is significantly boosting demand for PEG and low-profile feeding tubes. With a rapidly aging population, oropharyngeal dysphagia affects approximately 30–40% of independently living older adults and rises to around 47% in geriatric acute-care settings.
European health systems in countries like Germany, France, and the UK now emphasize long-term enteral nutrition, particularly using PEG and button-style tubes, in hospitals, nursing facilities, and home care. National guidelines, such as those from ESPEN and ESSD, recommend early intervention for dysphagia lasting over 4–6 weeks, prompting the adoption of low-profile feeding solutions that deliver reliable nutrition, require minimal maintenance, and enhance patient comfort.
Asia Pacific Enteral Feeding Devices Market Trends
Asia Pacific is experiencing strong growth in the adoption of enteral feeding devices, driven by the dual forces of ICU infrastructure expansion and a rising cancer burden. Between 2021 and 2023, China increased its ICU bed capacity by 18% as part of national efforts under the "Healthy China 2030" initiative to strengthen critical care systems. At the same time, the WHO South-East Asia Region reported 2.4 million new cancer cases and 1.5 million cancer-related deaths in 2022, with forecasts projecting an 85% increase in cancer cases by 2050. These developments underscore the growing demand for long-term nutritional support among critically ill and oncology patients.
Enteral feeding tubes and pumps are increasingly being integrated into hospital protocols and homecare regimens across countries like India, China, and Indonesia. This rapid healthcare evolution is positioning Asia Pacific as a high-potential market for enteral nutrition technologies focused on chronic care and critical support.
Competitive Landscape
The global enteral feeding devices market is characterized by moderate consolidation, with competition driven by innovation, regulatory compliance, and regional expansion. Manufacturers are focusing on developing ENFit-compliant connectors, user-friendly portable pumps, and low-profile tubes to meet evolving patient and provider needs. The shift toward home-based enteral nutrition and rising demand in emerging markets are prompting companies to localize production and expand distribution networks.
Technological advancements aimed at reducing complications such as tube dislodgement and clogging are becoming key differentiators. Players are increasingly investing in strategic partnerships, clinical validations, and training programs to strengthen their position in both developed and developing regions.
Key Industry Developments:
- In April 2025, NanoVibronix acquired enteral feeding device maker EnVue, integrating EnVue’s advanced low-profile G-tubes and infusion sets into its portfolio. The acquisition aims to expand NanoVibronix’s capabilities in home healthcare with improved patient comfort, compatibility with ENFit systems, and streamlined supply chain support.
- In February 2025, Cardinal Health launched the Kangaroo OMNI™ enteral feeding pump in Europe, Australia, and New Zealand. Designed for use with blended and thickened formulas, the pump features a night mode display, 30-day feeding history, and a portable, water-resistant design ideal for both clinical and home enteral nutrition settings.
- In May 2024, Vygon launched the NutriSafe2 enteral feeding system designed specifically for neonates. This advancement features a precision flow regulator, ENFit-compatible connectors, and an anti-reflux system to ensure safe and accurate nutrition delivery for premature and critically ill infants in neonatal intensive care units.
Companies Covered in Enteral Feeding Devices Market
- Applied Medical Technology
- B. Braun Melsungen AG
- Boston Scientific
- CONMED Corporation
- Cook Medical
- Cardinal Health, Inc.
- Medline
- Moog, Inc.
- Nutricia
- Vygon SA
- Amsino International, Inc.
- Fuji Systems
- Neomed
- ICU Medical, Inc.
- Abbott Nutrition
- QMD
- JMS Co.Ltd.
- ANGIPLAST PRIVATE LIMITED
- Romsons
- Fresenius Kabi
- Avanos Medical, Inc.
- Advin Health Care
Frequently Asked Questions
The global enteral feeding devices market is projected to value at 2.9 bn in 2025.
The enteral feeding devices market is driven by the rising prevalence of gastrointestinal disorders.
The enteral feeding devices market is poised to witness a CAGR of 5% between 2025 and 2032.
Increasing adoption of home enteral nutrition presents strong growth opportunities for device makers offering portable feeding pumps.








