- Medical Devices
- Elastomeric Infusion Pumps Market
Elastomeric Infusion Pumps Market Size, Trends, Share, Growth, and Regional Forecast, 2025 - 2032
Elastomeric Infusion Pumps Market by Product (Continuous Rate Elastomeric Pumps, Variable Rate Elastomeric Pumps), Application (Pain Management, Antibiotic/Antiviral, Chemotherapy, Others), End-user (Hospitals, Ambulatory Surgical Centers, Homecare Settings, Clinics), and Regional Analysis from 2025 - 2032
Elastomeric Infusion Pumps Market Share and Trends Analysis
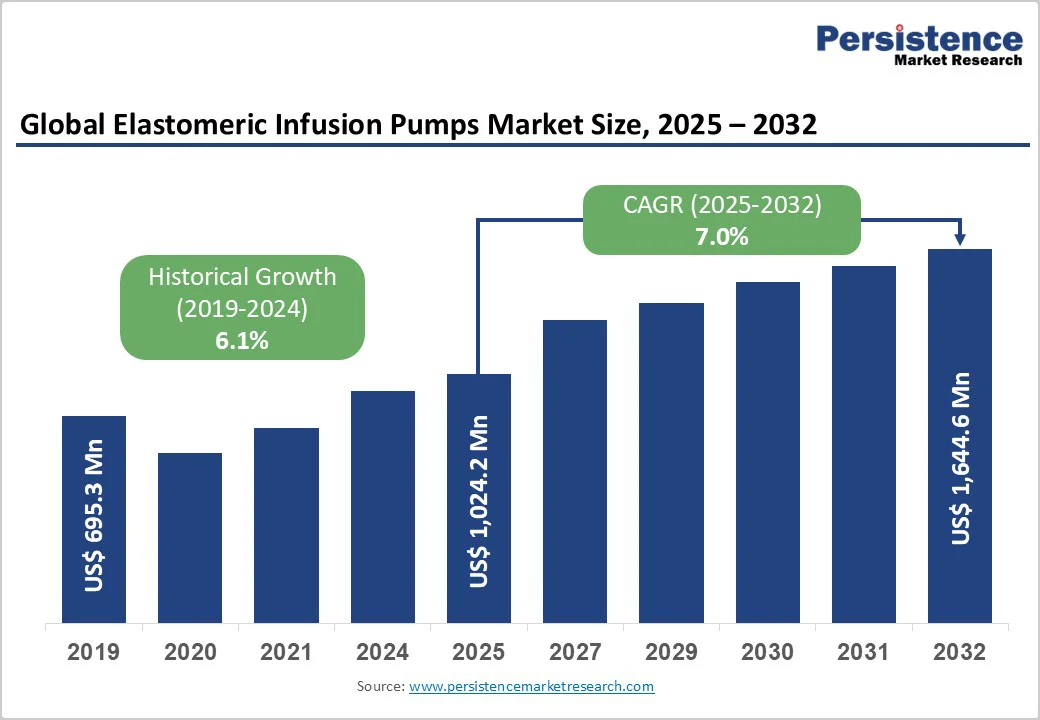
The global elastomeric infusion pumps market size is valued at US$1,024.2 million in 2025 and projected to reach US$1,644.6 million at a CAGR of 7.0% during the forecast period from 2025 to 2032. The global elastomeric infusion pumps market is expanding steadily, driven by the growing adoption of ambulatory and home-based infusion therapies, the increasing prevalence of chronic diseases such as cancer and infectious diseases, and continuous advancements in pump design and drug-delivery safety.
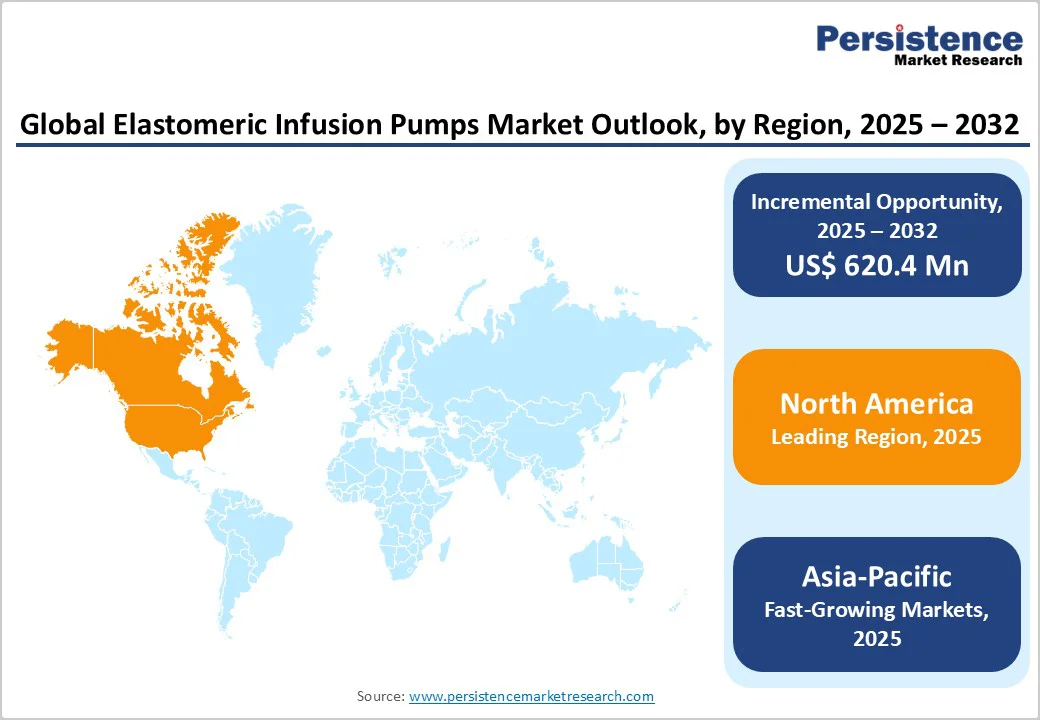
North America leads the market owing to its well-established healthcare infrastructure, high outpatient infusion volumes, and strong presence of key manufacturers. Meanwhile, the Asia-Pacific region is witnessing the fastest growth, driven by rising healthcare expenditure, expanding home healthcare services, and greater awareness among patients and clinicians of cost-effective, portable infusion solutions.
Key Industry Highlights
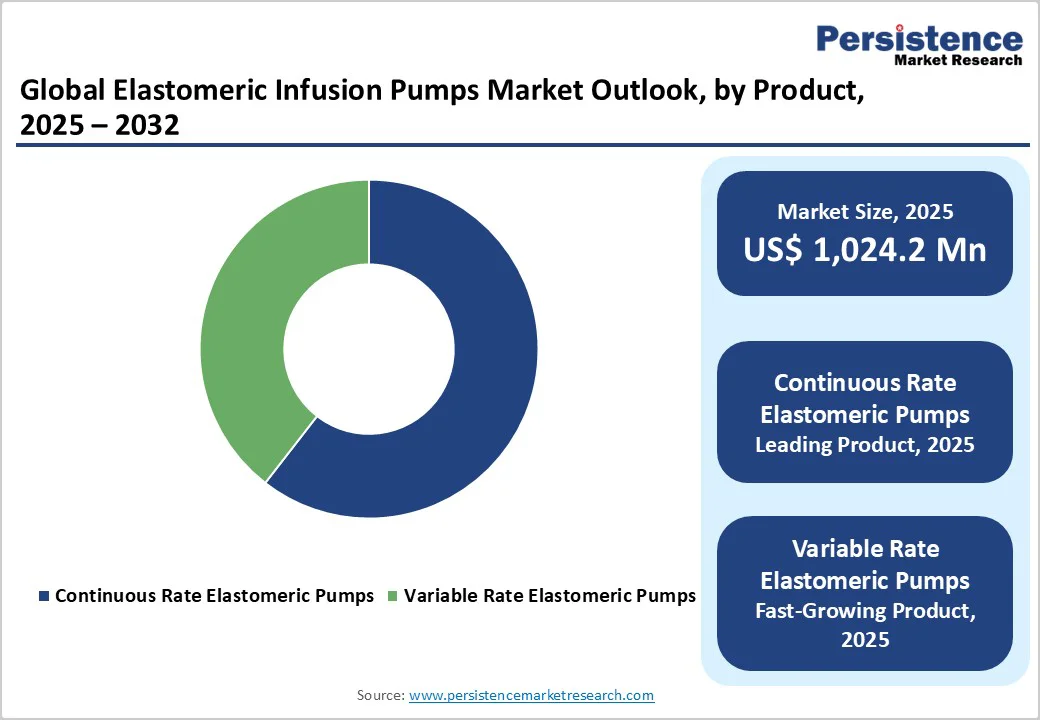
- Dominant Product: Continuous Rate Elastomeric Pumps, capturing nearly 60.5% market share, driven by their simple design, reliability, and consistent flow rate. These pumps are widely used in hospitals and home settings for antibiotic therapy, pain management, and chemotherapy, thanks to their ease of use and reduced reliance on external power sources.
- Dominant Application: Pain Management, holding around 38.6% share, dominates the Elastomeric Infusion Pumps Market due to increasing post-surgical analgesia needs, rising adoption of ambulatory pain control solutions, and growing use of elastomeric pumps for continuous peripheral nerve blocks and wound infiltration.
- Dominant Region: North America, accounting for approximately 39.7% of global revenue, leads the market owing to advanced healthcare infrastructure, high adoption of home infusion therapies, strong presence of key manufacturers, and favorable reimbursement for outpatient care.
- Investment Plans: Asia-Pacific is witnessing rapid growth, supported by expanding home healthcare programs, increasing prevalence of chronic illnesses, and growing investments by regional and global players to strengthen local manufacturing and distribution networks.
- Market Drivers: Rising demand for portable and disposable infusion systems, increasing preference for home-based therapies, growing incidence of cancer and infectious diseases requiring continuous infusions, and ongoing product advancements that enhance flow accuracy and patient comfort.
- Market Opportunity: Development of smart elastomeric pumps with digital monitoring features, integration of pressure sensors for real-time flow control, expansion into untapped emerging markets with cost-effective devices, and collaborations with healthcare providers to support long-term ambulatory infusion programs.
| Key Insights | Details |
|---|---|
|
Global Elastomeric Infusion Pumps Market Size (2025E) |
US$ 1,024.2 Mn |
|
Market Value Forecast (2032F) |
US$ 1,644.6 Mn |
|
Projected Growth (CAGR 2025 to 2032) |
7.0% |
|
Historical Market Growth (CAGR 2019 to 2024) |
6.1% |
Market Dynamics
Driver - Increasing Prevalence of Chronic Diseases Drives the Market Forward
The rising prevalence of chronic diseases such as cancer, diabetes, and cardiovascular disorders is a significant growth driver for the elastomeric infusion pumps market. According to the World Health Organization (WHO), chronic diseases account for 71% of all global fatalities, creating a pressing need for efficient long-term care solutions.
Elastomeric pumps, known for their ease of use, portability, and ability to deliver continuous medication are increasingly used in home care and outpatient settings. In cancer care, these pumps allow for precise chemotherapy dosing without requiring hospital stays, improving patient convenience and reducing healthcare costs.
Use of elastomeric pumps in managing diabetes and cardiovascular conditions through consistent infusion of medications ensures effective treatment and better patient compliance. As the global population ages and chronic diseases become more widespread, the demand for elastomeric infusion pumps is expected to grow exponentially.
Restraints - Inaccurate Delivery and Choice of Alternatives Limit Market Expansion
Many studies and reports have shown that the performance of elastomeric infusion pumps varies with the temperature and viscosity of the diluent or drug library, despite their many advantages. Interestingly, in a two-phase study, the infusion pump did not complete the flow on time in 50% of cases among patients receiving ambulatory chemotherapy. In some cases, it disrupted the treatment schedule due to sub-therapeutic dosing.
Inaccurate flow rate, inability to trace the history of the administered drug demand by the patient, and detection of abnormal drug delivery are factors limiting the adoption. The availability of alternative infusion pumps can dent market growth over the coming years.
Elastomeric infusion pump devices are among the most recalled medical devices due to their design deficiency and product errors. Other errors associated with these pumps include alarms, software issues, inadequate user interface design, damaged components, fire, sparks, battery failure, overheating, and electric shocks. These errors may impede the growth of the elastomeric infusion pumps market.
Opportunity - Increasing Initiatives to Address Infusion Pump Safety Problems
Increasing initiatives by institutes, organizations, and associations to repair elastomeric infusion pumps and address safety concerns will boost the market's growth. The US. The Food and Drug Administration (FDA) launched the infusion pump improvement initiative to address safety issues with infusion pumps and foster the development of safe, effective infusion pumps across the industry. This initiative established additional requirements for infusion pump manufacturers.
The FDA aims to raise awareness among hospital staff, administrators, and home users about mitigating the risks associated with using existing infusion pumps. Regulatory approvals and funding support will boost demand growth in the healthcare and medical device industries.
Elastomeric pump manufacturers are gradually focusing on advancing their existing products, developing new products, and introducing digital platforms to increase market penetration. Training staff to handle devices and administer drugs to patients and increase awareness would create goodwill and increase product adoption.
Category-wise Analysis
By Product, Continuous Rate Elastomeric Pumps Dominates the Elastomeric Infusion Pumps Market
The market is divided into continuous-rate and variable-rate elastomeric pumps. Out of these two prominent product types, the continuous rate elastomeric pumps lead the market, accumulating around 60.5% market share in 2025.
The dominance of the continuous-rate elastomeric infusion pumps is attributed to their consistent and regulated flow of medications, making them ideal for treatments requiring constant drug delivery, such as antibiotic therapies and pain management. Their simplicity and operational effectiveness rendered them a favored option in diverse healthcare environments, contributing to their substantial market share.
Continuous rate pumps are widely employed in hospitals and home healthcare environments due to their reliability and ease of use, stimulating demand and driving market growth. In April 2024, Baxter International Inc. launched advanced elastomeric infusion pumps with enhanced durability and user-friendly designs, reinforcing its status as a provider of infusion treatment solutions.
By Treatment, Pain Management is gaining traction due to rising chronic pain cases and a growing elderly population.
Based on treatment, the market is further classified into pain management, antibiotic/antiviral, and chemotherapy. Among these, the pain management treatment dominates the market with a significant share. Pain management treatment type to attain a 38.6% market share.
The rising incidence of chronic pain problems worldwide is resulting in an escalating demand for efficient pain treatment options. The growing elderly population, susceptible to numerous chronic conditions that necessitate pain management, is substantially driving market expansion in the sector.
Elastomeric infusion pumps are extensively utilized for pain management in both acute and chronic contexts, allowing patients to self-administer drugs as required and offering efficient and convenient analgesia. A study in the Journal of Pain Research demonstrated that elastomeric infusion pumps for post-operative pain management enhanced patient satisfaction and decreased opioid usage.
Regional Insights
North America Elastomeric Infusion Pumps Market Trends
The North America region dominates the global market with a 39.7% share in 2025, due to its aging population, advanced healthcare infrastructure, and strong reimbursement support for home infusion therapy. According to the U.S. Administration for Community Living (ACL), around 57.8 million Americans aged 65 and older, representing 17.3% of the total population in 2022, are most likely to require continuous infusion treatments for pain management, infections, and chemotherapy.
The Centers for Medicare & Medicaid Services (CMS) implemented a home infusion therapy benefit under the 21st Century Cures Act in 2021, covering infusion pumps and related services for home care. This combination of demographic pressure, policy backing, and a well-established outpatient infusion ecosystem positions North America as the dominant region in the elastomeric infusion pumps market.
Europe Elastomeric Infusion Pumps Market Trends
Europe is one of the leading regions in the elastomeric infusion pumps market, driven by its rapidly aging population, advanced healthcare systems, and strong emphasis on home-based care. According to Eurostat (2024), about 21.6% of the European Union’s population was aged 65 years or older, and the old-age dependency ratio reached 37%, indicating a growing need for long-term medical support. Countries such as Germany, France, and the United Kingdom have well-established reimbursement mechanisms for ambulatory and home infusion services, promoting the use of elastomeric pumps for pain management, antibiotic therapy, and oncology care. Additionally, the European Commission’s health policy framework encourages home-based treatment and medical device innovation, further driving the regional demand for portable infusion solutions.
Asia-Pacific Elastomeric Infusion Pumps Market Trends
The Asia-Pacific region is the fastest-growing market for elastomeric infusion pumps because of its rapidly ageing population and expanding healthcare investment. For example, the United Nations Population Fund reports that the number of people over 60 in Asia-Pacific will triple to about 1.3 billion by 2050, making up one-quarter of the population.
Meanwhile, the World Health Organization for Economic Co-operation and Development highlight that health-care spending per capita in the region grew at about 4.7% a year between 2010-2019, higher than GDP growth at ~3.6%. These trends create strong demand for home-infusion technologies and elastomeric pumps as countries shift toward outpatient and home-based care models.
Competitive Landscape
The elastomeric infusion pumps market is highly competitive, driven by innovation, expansion, and collaborations. Baxter International and B. Braun Melsungen dominate with advanced technologies, while ICU Medical and Nipro Corporation focus on user-friendly, sustainable solutions for home care. Regional players in Asia-Pacific offer cost-effective devices, intensifying competition. Strategic partnerships, R&D investments, and eco-friendly innovations continue to shape the market’s growth and support chronic disease management globally.
Key Industry Developments:
- In April 2025, ICU Medical announced the launch of a new category of infusion devices following the U.S. FDA clearances of its Plum Solo™ and Plum Duo™ precision IV pumps. The company stated that these next-generation pumps were designed to enhance accuracy, workflow efficiency, and patient safety in both hospital and outpatient settings.
- In November 2024, Avanos Medical announced that its ON-Q elastomeric infusion pump, along with the ambIT electronic infusion pump, qualified for separate Medicare reimbursement under the NOPAIN Act, effective January 1, 2025.
Companies Covered in Elastomeric Infusion Pumps Market
- Baxter International Inc.
- B. Braun Melsungen AG
- Avanos Medical, Inc.
- Leventon, S.A.U.
- Ambu A/S
- Nipro Corporation
- Epic Medical Pte Ltd
- Ace Medical Co., Ltd.
- Heka s.r.l.
- Vygon SAS
- Promecon GmbH
- ICU Medical
- Daiken Medical Co., Ltd.
- Woo Young Medical Co., Ltd.
- Others
Frequently Asked Questions
The global elastomeric infusion pumps market is projected to be valued at US$ 1,024.2 Mn in 2025.
Rising chronic diseases, growing home healthcare demand, aging population, and advancements in portable infusion technologies drive the global market.
The global elastomeric infusion pumps market is poised to witness a CAGR of 7.0% between 2025 and 2032.
Smart pump innovations, eco-friendly designs, expanding home healthcare, and growing demand in emerging economies create major market opportunities
Baxter International Inc., B. Braun Melsungen AG, Avanos Medical, Inc., Leventon S.A.U., Ambu A/S, Nipro Corporation.








