- Pharmaceuticals
- Adult Vaccines Market
Adult Vaccines Market Size, Share and Growth Forecast, 2026 - 2033
Adult Vaccines Market by Vaccine Type (Influenza, Pneumococcal, HPV, COVID-19, Herpes Zoster, Others), Technology Platform (mRNA, Recombinant, Conjugate, Inactivated, Viral Vector), Immunization Type (Routine, Travel, Occupational, Pandemic, Others), and Regional Analysis for 2026 - 2033
Adult Vaccines Market Share and Trends Analysis
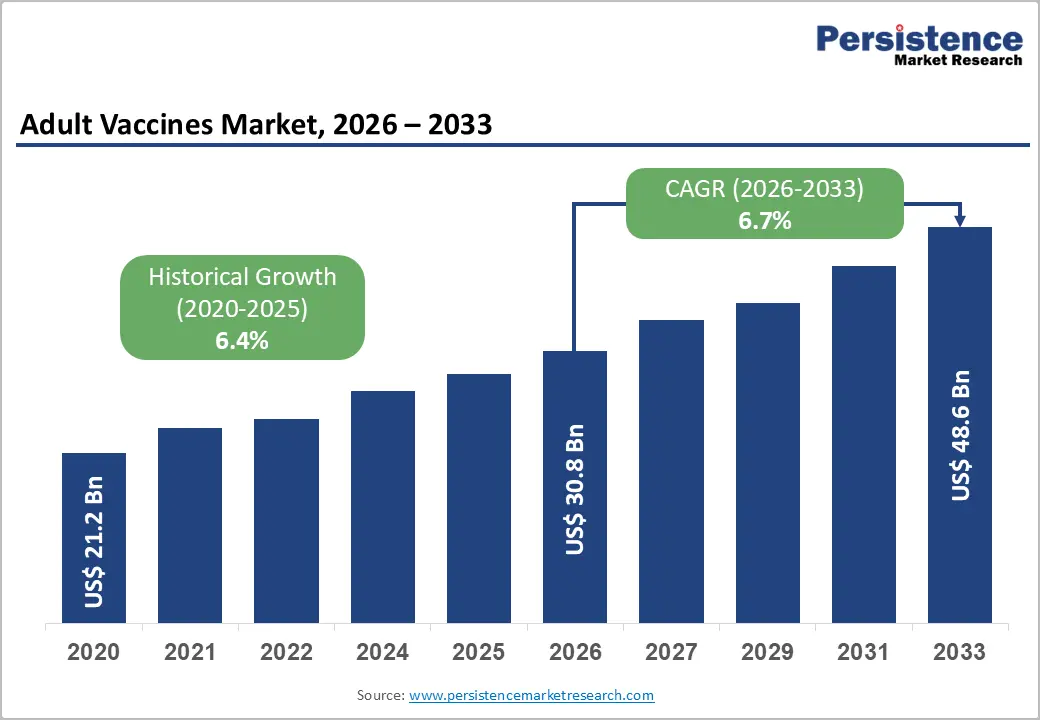
The global adult vaccines market size is likely to be valued at US$ 30.8 billion in 2026 and is projected to reach US$ 48.6 billion by 2033, growing at a CAGR of 6.7% during the forecast period 2026–2033.
The adult vaccines market is expanding as consumer preferences shift toward preventive healthcare and long-term disease risk reduction. Rising awareness of vaccine-preventable diseases, supported by public health campaigns from organizations such as WHO and CDC, is increasing adult immunization uptake. Technological advancements, including mRNA and recombinant vaccines, offer higher efficacy, faster development timelines, and improved safety profiles, strengthening confidence among healthcare providers and patients. Additionally, the integration of vaccines into routine adult care and workplace health programs is broadening their application across multiple end-use settings. Governments are increasing investments in national immunization programs, while private healthcare providers are promoting vaccination as a cost-effective preventive measure. These combined factors are creating sustained demand and reinforcing the market’s long-term growth trajectory.
Key Industry Highlights
- Dominant Vaccine Types: Influenza vaccines are estimated to command approximately 30% of revenue share in 2026, while COVID?19 vaccines are likely to grow the fastest at an estimated 11.8% CAGR through 2033, driven by ongoing booster requirements and pandemic preparedness initiatives.
- Leading Technology Platforms: Inactivated vaccines are projected to lead with around 29% share in 2026, while mRNA vaccines are expected to register the fastest growth at approximately 12.3% CAGR from 2026 to 2033, reflecting accelerated innovation and expanded pipelines.
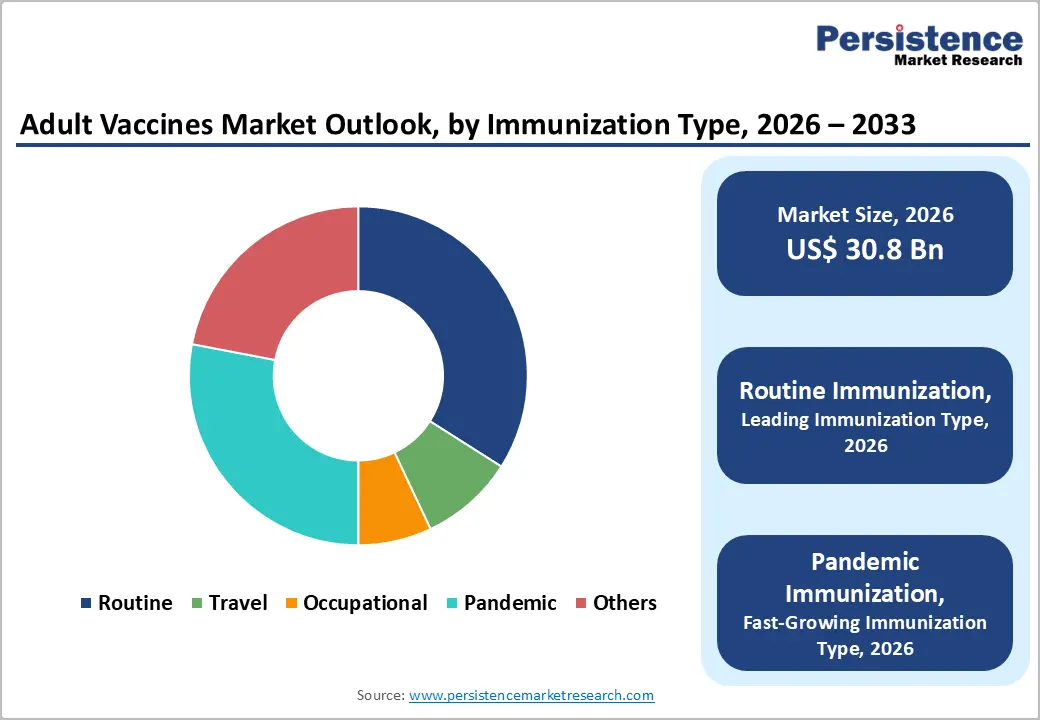
- Dominant Immunization Type: Routine immunization is anticipated to hold roughly 34% share in 2026, while pandemic immunization is predicted to be the fastest?growing segment at close to 10.9% CAGR over 2026–2033, supported by global health emergency frameworks.
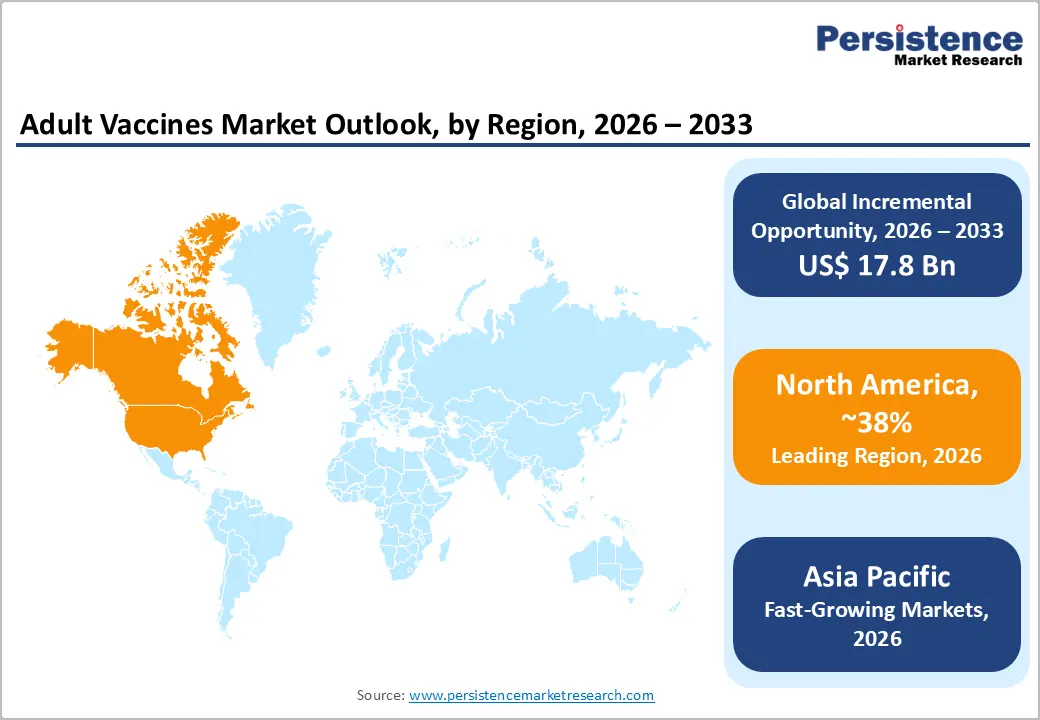
- Regional Leadership: North America is poised to lead with an estimated 38% share in 2026, while Asia Pacific is expected to be the fastest?growing region at around 9.7% CAGR through 2033, driven by rising healthcare access and market expansion across China, India, and Southeast Asia.
- Key Developments: Strategic developments such as mRNA innovation, combination vaccine launches, and capacity expansion initiatives reflect heightened investment in R&D and manufacturing scale?ups, reinforcing competitive positioning in both routine and pandemic segments.
| Key Insights | Details |
|---|---|
|
Adult Vaccines Market Size (2026E) |
US$ 30.8 Bn |
|
Market Value Forecast (2033F) |
US$ 48.6 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
6.7% |
|
Historical Market Growth (CAGR 2020 to 2025) |
6.4% |
DRO Analysis
Driver - Strengthening Preventive Healthcare Adoption and Policy Support
The adult vaccines market is expanding as healthcare systems increasingly prioritize preventive care over treatment, supported by structured immunization policies. According to the U.S. CDC, annual influenza vaccination is recommended for all adults, yet only 49.4% of adults received the vaccine in a recent season, highlighting significant growth potential. Policy-driven initiatives, including expanded eligibility, workplace vaccination programs, and insurance-backed coverage, are improving accessibility and convenience, helping normalize vaccination behavior beyond traditional healthcare settings. These measures, combined with increasing awareness, are integrating adult immunization into routine health management and strengthening long-term demand.
Policy frameworks are also evolving to improve coverage and outcomes. For instance, pneumococcal vaccination coverage among adults aged ≥65 reached 65.6%, while uptake among at-risk adults aged 19–64 remained only 23.4%, prompting targeted strategies and digital health tracking systems. By reducing hospitalizations and healthcare costs, expanded funding, eligibility, and consistent recommendations are reinforcing adoption. This policy-driven momentum is creating a stable demand environment for vaccine manufacturers, benefiting the market through increased coverage and improved alignment with healthcare systems.
Innovation in Vaccine Technologies and Expanding Global Access
Technological advancements are significantly strengthening the value proposition of adult vaccines, particularly through mRNA and recombinant platforms, which enable faster development cycles, improved efficacy, and rapid adaptation to emerging variants. For example, influenza vaccination alone prevents millions of illnesses and hundreds of thousands of hospitalizations annually in the U.S., highlighting measurable health benefits that drive adoption. Innovations such as combination vaccines further improve convenience and compliance, enhancing uptake across adult populations, including high-risk groups with chronic conditions, and reinforcing confidence among healthcare providers.
Simultaneously, emerging markets are fueling demand growth through rising incomes, improved healthcare access, and expanding vaccination programs. Despite increased awareness, coverage for key adult vaccines remains below 50% in many regions, revealing significant untapped potential. Investments in local manufacturing and global partnerships are reducing costs and improving supply chain resilience, particularly in Asia Pacific, which is emerging as a key production hub. The combination of technological innovation and geographic expansion is creating a strong foundation for sustained long-term growth in the adult vaccines market.
Restraints - High Vaccine Costs and Limited Access in Low?Income Regions
Despite global immunization efforts, vaccine affordability remains a significant barrier in low? and middle?income countries (LMICs). Advanced vaccines, particularly mRNA-based products, require sophisticated production and cold-chain logistics, increasing costs compared to traditional vaccines. According to the World Health Organization (WHO), constrained financing and reduced health budgets in several LMICs in 2025 risk slowing the introduction of newer vaccines and could reduce adult coverage gains. This cost disparity limits broad adoption and creates inequities in vaccine access. Without coordinated support, many adults remain vulnerable to preventable diseases, further straining public health systems.
These financial and logistical challenges result in uneven vaccination rates, with adult immunization in many LMICs remaining below 20%. Weak healthcare infrastructure, insufficient cold-chain systems, and limited reimbursement mechanisms further exacerbate the problem. WHO has emphasized that without sustained investment and global coordination, access to essential vaccines will remain inequitable. Bridging these gaps requires international funding, local manufacturing investments, and improved supply chain management. Until addressed, these structural issues continue to restrain overall adult vaccine market penetration, slowing long-term growth potential. Governments and global agencies must prioritize adult vaccination programs to ensure equitable coverage.
Vaccine Hesitancy and Misinformation
Vaccine hesitancy continues to undermine immunization efforts globally, recognized by WHO as one of the top ten health threats. Post-COVID-19, widespread misinformation and safety concerns have amplified public skepticism, particularly in adult populations. WHO’s Strategic Advisory Group of Experts (SAGE) in 2025 warned that misinformation could derail vaccination campaigns, reduce uptake, and erode public confidence in preventive health programs. This creates demand volatility and unpredictable adult vaccine adoption patterns, posing a significant market challenge. If not addressed, hesitancy can also impact acceptance of newly introduced vaccines beyond COVID-19, affecting broader adult immunization strategy.
The impact of hesitancy was visible in early 2026 during a measles outbreak in north London, where misinformation and public distrust contributed to low vaccination coverage in adult and adolescent populations. Public health officials stressed that building trust requires sustained education, targeted communication, and community engagement, which are resource-intensive efforts. Until these behavioral barriers are addressed, hesitancy will continue to limit adult vaccine uptake, slow coverage expansion, and restrain overall market growth globally. Coordinated campaigns and transparent communication remain critical to overcoming these challenges.
Opportunity - Growth Potential in Emerging Markets with Life?Course Immunization Focus
Emerging economies in Asia Pacific, Latin America, and Africa present substantial growth opportunities as adult vaccination coverage remains comparatively low. According to the World Health Organization (WHO), while high?income countries routinely include adult vaccines in national schedules, many LMICs have not yet scaled life?course immunization, leaving significant gaps in protection. WHO and partners are prioritizing expanding vaccination across all ages, emphasizing adult coverage to strengthen public health resilience globally. This renewed focus supports infrastructure investments and policy shifts toward broader immunization strategies.
In 2025, the WHO highlighted that rapidly aging population and rising healthcare costs make adult vaccines a critical health priority, particularly in regions where preventive care systems are evolving. As governments increase healthcare budgets and integrate adult immunization into primary care, access improves, and demand accelerates. Local manufacturing initiatives and public?private partnerships reduce costs and strengthen supply chains, unlocking multi?billion?dollar expansion potential. With strategic positioning, companies can tap into these high?growth markets, aligning commercial goals with global health priorities.
Innovation in Combination & Booster Vaccines Supported by Updated Immunization Guidance
Combination and multi?valent vaccines are gaining momentum as healthcare systems aim to simplify adult immunization while enhancing disease protection. Advances in vaccine science, including updated formulations targeting multiple pathogens, have garnered attention from public health authorities. In the U.S., the CDC’s 2025 adult immunization schedule includes expanded recommendations for influenza, RSV, and pneumococcal vaccines, reflecting broader use of diverse vaccine platforms. These schedule updates indicate growing acceptance of advanced and combination immunizations among adult populations.
Booster vaccination programs are also being actively supported by government guidance. For the COVID?19 season, the CDC recommends individualized COVID?19 vaccination for adults based on risk factors, and continues to endorse targeted booster doses for high?risk individuals to maintain immunity. This approach underscores recurring demand for booster vaccines as part of routine preventive care. As immunization policies evolve to include periodic boosters and innovative vaccine combinations, manufacturers can capitalize on stable, long?term demand cycles, creating recurring revenue streams while driving broader adult vaccine adoption.
Category-wise Analysis
Technology Platform Insights
The inactivated vaccines segment is expected to continue to lead the adult vaccines market, with an estimated 29% revenue share in 2026, owing to its long?standing safety profile and widespread regulatory acceptance. The Adult Immunization Schedule published by the U.S. CDC maintains strong recommendations for inactivated influenza and pneumococcal vaccines across adult age groups, reinforcing their foundational role in national preventive programs. This platform’s ease of storage, broad clinical familiarity, and compatibility with existing healthcare infrastructure underpin its continued dominance. Established supply chains and regulatory comfort make inactivated vaccines reliable choices for routine adult immunization. Health systems also benefit from predictable dosing schedules and minimal cold?chain requirements, which support high uptake. As public health agencies reinforce routine usage, inactivated formulations sustain their leadership position.
In contrast, mRNA vaccines are likely to emerge as the fastest?growing technology platform, projected to register approximately a 12.3% CAGR through 2033 due to rapid adaptability and innovation potential. In early 2026, Moderna filed regulatory submissions for its investigational mRNA?based seasonal influenza vaccine in multiple jurisdictions, including the U.S., EU, Canada and Australia, highlighting expanding applications beyond COVID?19. Additionally, the WHO’s mRNA Technology Transfer Programme Phase?2.0 is driving regional manufacturing capacity in Latin America, Africa, and Asia, enabling broader access and local innovation. The agility of mRNA platforms allows rapid antigen updates against evolving strains, supporting future pandemic responses. Continued investment in mRNA pipelines and supportive international initiatives position this technology as a key growth driver in adult vaccines.
Immunization Type Insights
The routine immunization segment is anticipated to hold the largest market share, estimated at 34% in 2026, anchored by comprehensive government programs and dosing strategies embedded in public health policy. The CDC’s 2025–2026 Adult Immunization Schedule outlines routine recommendations for influenza, pneumococcal conjugates, HPV, hepatitis, and other age?based vaccines for adults aged 19?years and older. This structured schedule ensures predictable demand and strengthens preventive care frameworks. Adult routine dosing, including annual influenza shots and age?or condition?based vaccines, supports consistent healthcare system integration. Public immunization reminders and established clinical workflows further increase adoption. As national health agencies emphasize life?course vaccination, routine programs reinforce sustained market demand and base revenue streams for vaccine manufacturers.
In contrast, the pandemic immunization segment is projected to grow at the fastest rate, with an estimated 10.9% CAGR through 2033, reflecting elevated preparedness efforts and continued government focus on rapid vaccine deployment. While not a “routine” schedule, the 2025 Adult Immunization Schedule includes updated guidance for COVID?19 vaccine dosing, including multiple doses based on age and risk categories. These recommendations reflect ongoing efforts to maintain immunity through periodic boosting against evolving variants. Public health agencies in multiple countries continue investing in emergency immunization planning and booster campaigns to mitigate future outbreaks. Combined with routine immunization, pandemic?focused programs are driving expansion in immunization coverage and market opportunity.
Regional Insights
North America Adult Vaccines Market Trends
North America is expected to continue its domination in the adult vaccines market, representing an estimated 38% share in 2026 thanks to high adult vaccination coverage and advanced healthcare systems. The CDC’s Adult Immunization Schedule for 2025–2026 includes updated recommendations for routine doses such as influenza and expanded RSV guidance for adults over 50, reinforcing structured dosing programs. Seasonal influenza vaccination campaigns during the 2025–26 season were supported by updated Federal recommendations and expanded age eligibility, strengthening routine immunization uptake across adult populations. Strong regulatory frameworks at the FDA ensure vaccine quality and safety, making the region attractive for product introductions.
Innovation in vaccine technology continues to shape North America’s competitive landscape. Regulatory actions in 2026, such as the FDA’s endorsement of strain selections for the 2026–2027 flu vaccine, reflect continued investment in updated seasonal vaccines that protect against evolving influenza variants. Ongoing dialogue with regulators and multi?stakeholder investment demonstrates the commitment to next?generation platforms and responsive immunization programs. Partnerships between federal agencies and private sector developers are expanding adult vaccination delivery through pharmacies and employer?based clinics. With strong reimbursement systems and public health infrastructure, North America is projected to grow with a steady growth rate, maintaining leadership in both routine and pandemic immunization efforts.
Europe Adult Vaccines Market Trends
Europe represents a mature and steadily growing adult vaccines market, anchored by coordinated public health systems and comprehensive immunization policies. Across Germany, the U.K., France, Spain, and other EU member states, governments continue to emphasize adult dosing campaigns, particularly for influenza and pneumococcal vaccines. The European Centre for Disease Prevention and Control (ECDC) plays a central role in facilitating harmonized immunization guidance and shared surveillance efforts across countries. Long?standing public healthcare frameworks ensure broad access to recommended adult vaccines, while age?based programs target older and at?risk populations to reduce disease burden.
Collaborative initiatives in 2025–2026 have focused on strengthening manufacturing and distribution infrastructure to support adult immunization programs. National health ministries are investing in capacity expansions that improve on?time vaccine supply for seasonal campaigns. Cross?country procurement agreements and shared forecasting models have helped reduce regional stockouts and enhance program efficiency. Public awareness efforts, including targeted adult vaccination drives in urban centers, have increased demand for routine shots such as influenza, RSV, and tetanus boosters. As Europe continues to expand its preventive healthcare priorities, its role as both a stable market and a production hub for adult vaccines remains integral to the global supply ecosystem.
Asia Pacific Adult Vaccines Market Trends
Asia Pacific is expected to be the fastest?growing region with around 9.7% CAGR through 2033 in the adult vaccines market, driven by expanding healthcare systems and enhanced government immunization efforts. In 2025, the Asia?Pacific Economic Cooperation (APEC) Health Working Group released a regional dashboard highlighting the need to strengthen life?course immunization policies, including expanding vaccine access for adults amid declining confidence and coverage in several economies. This underscores a policy priority across member countries to broaden vaccination beyond childhood, acknowledging that adult immunization contributes to healthier and more productive populations. Governments are increasingly aligning life?course immunization strategies with broader public health objectives, setting the stage for stronger adult vaccination programs.
National efforts underscore this regional momentum. For example, India’s Universal Immunisation Programme (UIP) in early 2026 launched a nationwide HPV vaccination campaign and introduced an indigenous Tetanus–Diphtheria (Td) vaccine, expanding adult immunization under government auspices. These initiatives reflect a commitment to broadening adult vaccination coverage and reducing disease burden through enhanced local production and public outreach. In addition, the WHO South?East Asia Regional Working Group on Immunization convened in July 2025 to assess progress and strengthen partner?aligned strategies for immunization system resilience, including adult vaccine delivery frameworks. These developments collectively highlight both government engagement and strategic planning that are accelerating adult vaccine access and uptake across the Asia Pacific, positioning it as a key long?term growth driver for the global market.
Competitive Landscape
The global adult vaccines market structure is moderately consolidated, with major pharmaceutical and biotech companies holding significant influence across key segments. Established players such as Pfizer, Moderna, GlaxoSmithKline (GSK), Sanofi, and Merck & Co. leverage deep regulatory expertise, extensive distribution networks, and diversified vaccine portfolios to maintain leadership. These companies continue to invest heavily in research and development, particularly in next?generation platforms such as mRNA, recombinant technologies, and multi?valent vaccines, ensuring innovation remains a core competitive differentiator.
Regional and specialized firms, including Serum Institute of India, Bharat Biotech, CSL Limited, and Sinovac Biotech, focus on cost-effective manufacturing and local market expansion. Strategic collaborations, government partnerships, and integration with digital immunization platforms enhance vaccine access and adherence. Market growth is further supported by mergers, acquisitions, and technology transfer programs to expand production and diversify regional supply.
Key Industry Developments
- In February 2026, Sanofi finalized its US $2.2?billion acquisition of Dynavax Technologies, adding HEPLISAV-B and the Z?1018 shingles candidate to its portfolio. This strategic move strengthens Sanofi’s adult immunization offerings by combining Dynavax’s differentiated products with Sanofi’s global commercial infrastructure, enhancing market reach and pipeline diversity.
- In January 2026, Novavax entered a non-exclusive licensing agreement with Pfizer for its Matrix-M adjuvant, receiving US $30?million upfront and up to US $500?million in potential milestones. This collaboration allows Pfizer to develop vaccines for two infectious disease areas while leveraging Novavax’s adjuvant technology, creating new revenue streams and fostering innovation in adult vaccine development.
Companies Covered in Adult Vaccines Market
- Pfizer Inc.
- Moderna, Inc.
- GlaxoSmithKline plc
- Sanofi
- Merck & Co., Inc.
- AstraZeneca plc
- Johnson & Johnson
- CSL Limited
- Serum Institute of India
- Sinovac Biotech Ltd.
- Bharat Biotech
- Daiichi Sankyo Company
- Novavax, Inc.
Frequently Asked Questions
The global adult vaccines market is projected to reach US$ 30.8 billion in 2026.
Rising disease burden, government immunization policies, and rapid adoption of next-generation vaccines are driving the market.
The market is expected to grow at a 6.7% CAGR from 2026 to 2033.
Emerging markets, the development of combination vaccines, and expanding adult booster programs present key growth opportunities.
Pfizer, Moderna, GSK, Sanofi, Merck & Co., Serum Institute of India, Bharat Biotech, CSL Limited, and Sinovac Biotech are leading companies.








