- Medical Devices
- Yersinia Diagnostics Market
Yersinia Diagnostics Market Size, Share, and Growth Forecast, 2026 - 2033
Yersinia Diagnostics Market by Test Type (Stool Culture, ELISA, Radioimmunoassay, Tube Agglutination, Others), Technology (DNA Probes, Immunoassay, Monoclonal Antibodies, PCR (Polymerase Chain Reaction)), End-user, and Regional Analysis for 2026 – 2033
Yersinia Diagnostics Market Size and Trends Analysis
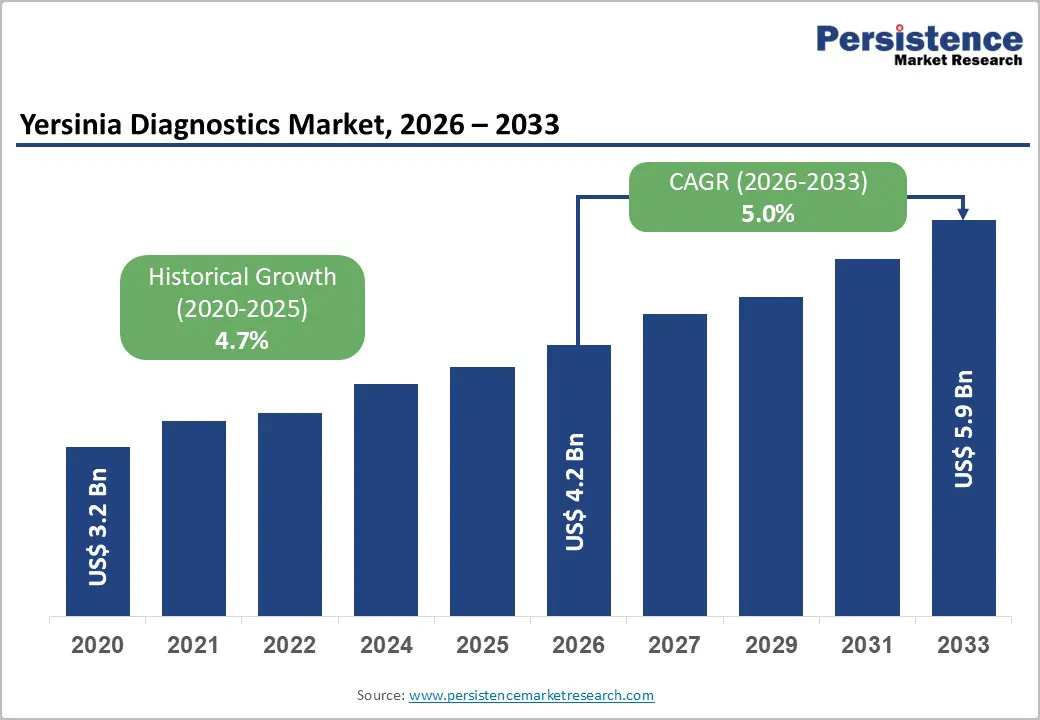
The global Yersinia diagnostics market size is likely to be valued at US$4.2 billion in 2026, and is expected to reach US$5.9 billion by 2033, growing at a CAGR of 5.0% during the forecast period from 2026 to 2033, driven by the increasing prevalence of Yersinia enterocolitica and pseudotuberculosis infections in developed and emerging economies, rising foodborne outbreak surveillance, growing demand for rapid and accurate molecular diagnostics, and expanding public health laboratory infrastructure for enteric pathogen screening. Growing demand for PCR-based and ELISA test types, especially in hospital laboratories and commercial diagnostic centers, is accelerating adoption across healthcare systems. Increasing recognition of Yersinia diagnostics as critical for early identification of yersiniosis, prevention of post-infectious complications (reactive arthritis, erythema nodosum), and outbreak control in emerging food safety and infectious disease markets remains a major driver of market growth.
Key Industry Highlights:
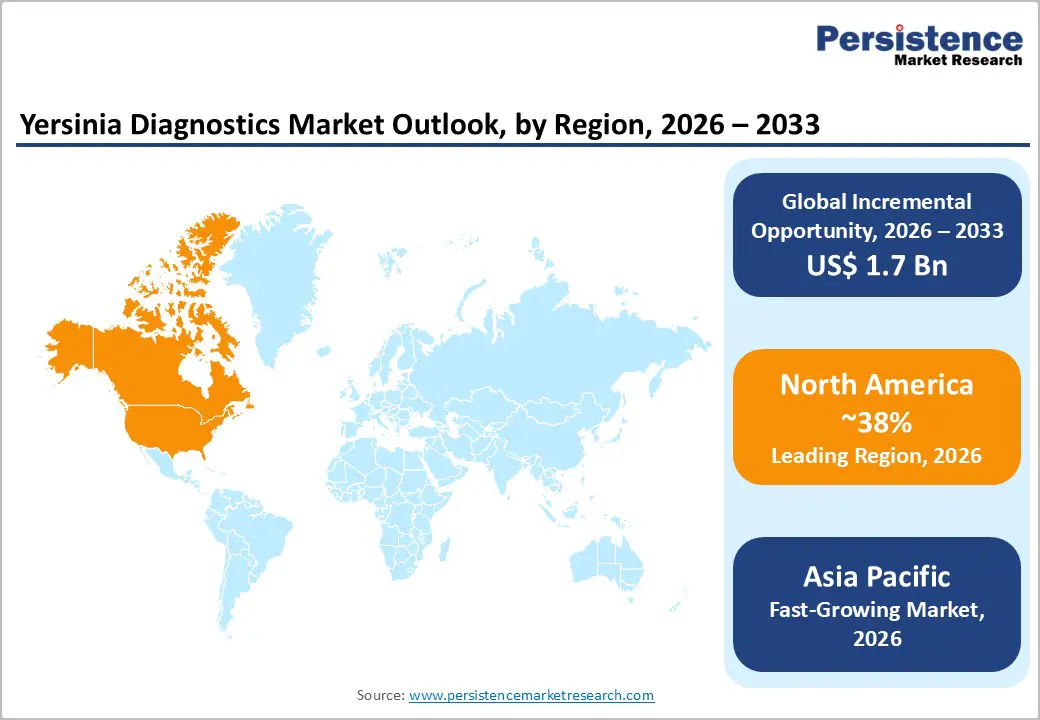
- Leading Region: North America, to account for a 38% market share in 2026, driven by high foodborne illness reporting, advanced molecular lab networks, and strong demand in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by rising enteric infection burden, expanding private lab chains, and increasing food safety regulations in China and India.
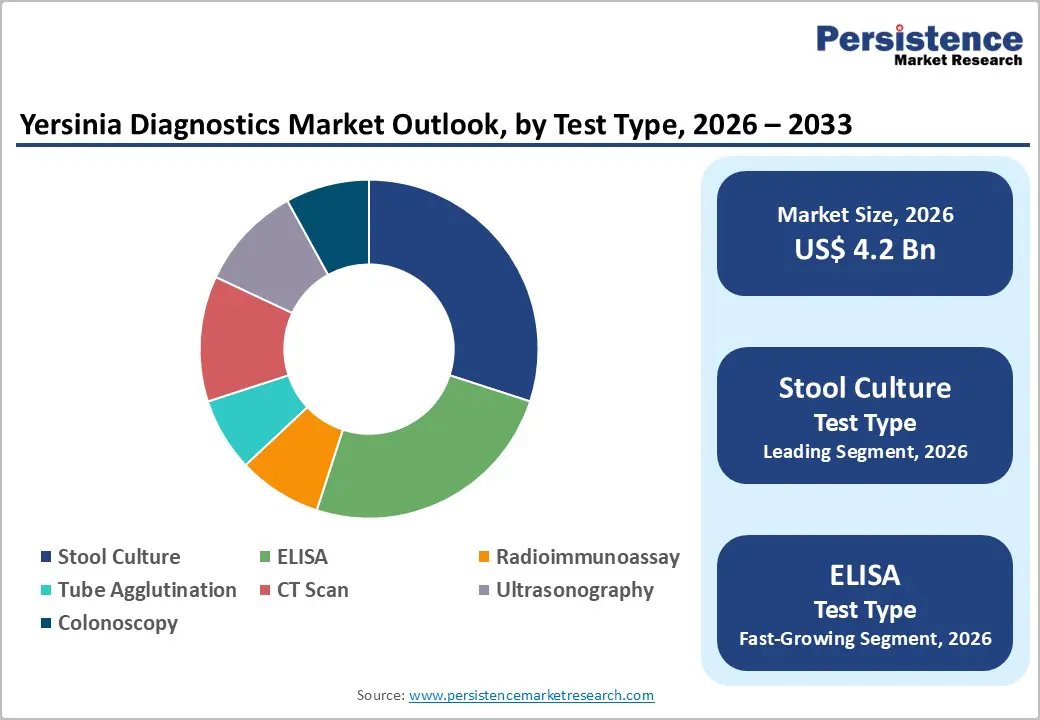
- Dominant Test Type: ELISA, to hold approximately 42% of the market share, as it remains the most widely used serological method.
- Leading Technology: Immunoassay, to contribute nearly 48% of the market revenue, due to the highest routine usage volume.
| Key Insights | Details |
|---|---|
|
Yersinia Diagnostics Market Size (2026E) |
US$4.2 Bn |
|
Market Value Forecast (2033F) |
US$5.9 Bn |
|
Projected Growth CAGR (2026-2033) |
5.0% |
|
Historical Market Growth (2020-2025) |
4.7% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Growth Analysis – Increasing Foodborne Outbreaks and Surveillance Programs
Increasing foodborne outbreaks have become a major public health concern worldwide, driven by globalization of food supply chains, urbanization, changing dietary habits, and rising consumption of processed and ready-to-eat foods. Contamination can occur at multiple stages—production, processing, storage, transportation, or retail—making prevention complex. Pathogens such as Salmonella, Listeria, E. coli, and Campylobacter continue to cause significant illness, hospitalizations, and economic losses. Climate change and increased cross-border trade further contribute to the spread and persistence of foodborne pathogens, increasing the frequency and geographic reach of outbreaks.
Governments and health agencies are strengthening food safety surveillance programs to detect, monitor, and control outbreaks more effectively. Modern surveillance systems integrate laboratory testing, molecular diagnostics, genome sequencing, and digital reporting platforms to enable rapid identification of contamination sources. Traceability systems, mandatory reporting requirements, and real-time data sharing between regulatory bodies improve outbreak response times and reduce public health risks. Risk-based inspection frameworks and stricter food safety regulations also encourage manufacturers to adopt preventive controls such as Hazard Analysis and Critical Control Points (HACCP).
Rising Molecular Diagnostics Adoption in Clinical Labs
The adoption of molecular diagnostics in clinical laboratories is increasing rapidly due to the growing need for accurate, rapid, and early disease detection. Unlike conventional diagnostic methods that rely on culture or immunoassay techniques, molecular diagnostics detect specific genetic material (DNA or RNA) of pathogens or genetic mutations, enabling highly sensitive and specific results. This precision supports early diagnosis of infectious diseases, genetic disorders, and various cancers, improving patient outcomes and reducing complications. Technological advancements such as polymerase chain reaction (PCR), real-time PCR, next-generation sequencing (NGS), and automated sample-to-answer platforms have made molecular testing more efficient and accessible.
Automation reduces human error, shortens turnaround time, and increases laboratory throughput. Clinical laboratories are increasingly integrating these platforms to handle growing test volumes while maintaining accuracy and regulatory compliance. The rising prevalence of infectious diseases, antimicrobial resistance, and chronic conditions has further accelerated demand for molecular testing. Personalized medicine approaches require genetic profiling to guide targeted therapies, particularly in oncology. Hospitals and reference laboratories are investing in molecular diagnostic infrastructure to support precision treatment decisions.
Barrier Analysis – High Cost of Molecular Assays
The high cost of molecular assays remains a significant barrier to widespread adoption, particularly in resource-limited settings. Molecular testing requires advanced instrumentation such as real-time PCR systems, automated extraction platforms, and sequencing equipment, all of which involve substantial capital investment. In addition to equipment costs, laboratories must account for expensive reagents, specialized consumables, and proprietary test kits that increase per-test expenses.
Operational costs further add to the financial burden. Molecular assays require skilled laboratory personnel, ongoing training, strict quality control measures, and maintenance of highly controlled environments to prevent contamination. Many assays also depend on patented technologies, limiting pricing flexibility and supplier competition. For smaller hospitals and diagnostic centers, the high upfront and recurring costs can limit test availability or result in higher patient charges.
Limited Awareness and Under-Reporting
Limited awareness and under-reporting remain key challenges in disease detection and surveillance systems. In many regions, patients may not recognize early symptoms or may avoid seeking medical care due to social stigma, financial constraints, or limited access to healthcare facilities. Numerous cases go undiagnosed or unrecorded, leading to an incomplete understanding of disease prevalence and transmission patterns.
Healthcare providers may also lack adequate training or diagnostic resources to accurately identify certain conditions, contributing to misdiagnosis or delayed confirmation. In some settings, weak health information systems and inconsistent reporting protocols hinder timely data collection and sharing. Without standardized reporting mechanisms, cases may not be systematically documented at regional or national levels. Under-reporting affects public health planning, resource allocation, and outbreak response strategies. When disease burden appears lower than reality, authorities may underestimate risks and delay preventive interventions.
Opportunity Analysis – Innovation in Multiplex PCR Panels and Point-of-Care Tests
Innovation in multiplex PCR panels and point-of-care (POC) tests is transforming modern diagnostics by improving speed, accuracy, and efficiency. Multiplex PCR technology allows simultaneous detection of multiple pathogens or genetic targets within a single test run. Instead of conducting separate assays for each suspected organism, laboratories can identify a broad panel of viruses, bacteria, or resistance markers at once. This reduces turnaround time, conserves reagents, and supports faster clinical decision-making, particularly in cases where symptoms overlap, such as respiratory or gastrointestinal infections.
Advancements in assay design, reagent stability, and automated platforms have enhanced sensitivity while minimizing cross-reactivity between targets. Compact, cartridge-based systems integrate sample preparation, amplification, and detection into a streamlined workflow, reducing manual handling and contamination risk. Point-of-care tests further extend diagnostic capabilities beyond centralized laboratories. Portable molecular devices provide rapid results at hospitals, clinics, emergency departments, and even remote settings. These systems are designed for ease of use, requiring minimal technical expertise while delivering reliable performance. Faster diagnosis enables timely treatment, improved infection control, and better patient outcomes.
Expansion in Food Safety Testing
Expansion in food safety testing is driven by increasing consumer awareness, stricter regulatory standards, and the growing complexity of global food supply chains. As food products move across multiple countries and processing stages before reaching consumers, the risk of contamination by pathogens, chemical residues, allergens, and adulterants has increased. This has compelled food manufacturers, distributors, and regulatory bodies to strengthen quality control measures through more comprehensive and frequent testing.
Advancements in analytical technologies such as rapid microbiological methods, molecular diagnostics, chromatography, and mass spectrometry have improved detection accuracy and reduced turnaround times. These tools enable identification of contaminants at very low levels, supporting preventive action before products reach the market. Automation and digital traceability systems further enhance monitoring by improving data management and enabling faster recall procedures when necessary. Food safety testing has also expanded beyond traditional pathogen detection to include testing for pesticide residues, heavy metals, mycotoxins, genetically modified organisms (GMOs), and food authenticity.
Category-wise Analysis
Test Type Insights
Stool culture is expected to dominate with over 30% share in 2026, driven by its established reliability and clinical acceptance. As the traditional gold standard for identifying bacterial enteric pathogens such as Salmonella, Shigella, Campylobacter, and pathogenic E. coli, stool culture provides definitive results that guide targeted treatment and antibiotic susceptibility. Many hospitals and diagnostic laboratories continue to use stool culture because of its accuracy, relatively low cost, and long history in clinical microbiology. Max Lab and Dr. Lal PathLabs both offer stool culture and sensitivity tests as standard clinical diagnostics to identify harmful bacteria such as Salmonella or E. coli in patients with gastrointestinal symptoms. This test involves growing bacteria from stool samples in a lab setting, which helps physicians confirm infection and select effective antibiotics.
ELISA is expected to be the fastest-growing in 2026, due to its reliability, sensitivity, and cost-effectiveness. The technique is widely used for detecting antigens and antibodies in infectious diseases, food safety testing, and clinical diagnostics. Its relatively simple workflow, compatibility with high-throughput screening, and adaptability to multiple sample types make it suitable for both small laboratories and large diagnostic centers. Mikrogen Diagnostik developed the recomWell Yersinia IgG ELISA kit, an enzyme-linked immunosorbent assay designed to detect IgG, IgA, and IgM antibodies against Yersinia enterocolitica and Yersinia pseudotuberculosis in human serum or plasma. The assay uses recombinant Yersinia outer membrane proteins (YOPs) to improve diagnostic sensitivity and specificity, enabling reliable detection of infections and differentiation between antibody classes for disease monitoring.
Technology Insights
Immunoassay is expected to dominate the market, contributing nearly 48% of revenue in 2026, fueled by its broad clinical applications, high throughput capability, and cost efficiency. These tests are widely used for detecting hormones, infectious disease markers, cardiac biomarkers, tumor markers, and therapeutic drug levels. Automated immunoassay analyzers enable rapid processing of large sample volumes with consistent accuracy, making them essential in hospital and reference laboratories. Abbott Laboratories’ Alinity i immunoassay system is widely adopted in clinical laboratories for high-throughput testing of infectious diseases, cardiac markers, and hormones. The Alinity i series is known for its broad test menu and automated workflow, helping labs process large volumes of samples efficiently.
PCR (Polymerase Chain Reaction) represents the fastest-growing test type, due to its exceptional sensitivity, specificity, and rapid detection capabilities. It enables the identification of minute quantities of DNA or RNA, making it highly effective for early-stage disease detection and infectious disease diagnosis. Real-time PCR and automated platforms have further accelerated adoption by reducing turnaround time and improving workflow efficiency in clinical laboratories. Thermo Fisher Scientific offers the Applied Biosystems™ TaqMan™ Yersinia pestis Detection Kit, which uses real-time PCR technology to amplify and detect pathogen-specific DNA sequences. The assay enables rapid and highly sensitive identification of Yersinia pestis through a closed-tube PCR workflow, minimizing contamination risk and reducing sample-processing time.
Regional Insights
North America Yersinia Diagnostics Market Trends
North America is projected to dominate, accounting for nearly 38% of the revenue in 2026, driven by the region’s high foodborne illness reporting, advanced molecular lab networks, and high public awareness of enteric pathogen testing benefits. Distribution systems in the U.S. and Canada provide extensive support for Yersinia diagnostics programs, ensuring wide accessibility across ELISA, immunoassay, and hospital laboratory populations. Increasing demand for rapid, convenient, and easy-to-perform forms is further accelerating adoption, as these formats improve outbreak detection and reduce barriers associated with conventional culture.
Innovation in Yersinia diagnostics technology, including stable multiplex PCR, improved immunoassay delivery, and targeted surveillance enhancement, is attracting significant investment from both public and private sectors. Government initiatives and CDC campaigns continue to promote use against foodborne risks, diagnostic accuracy concerns, and emerging outbreak threats, creating sustained market demand. The growing focus on PCR grades and specialty uses, particularly for ELISA and others, is expanding the target applications for Yersinia diagnostics.
Europe Yersinia Diagnostics Market Trends
Europe is witnessing the steady growth driven by increasing awareness of yersiniosis diagnostic benefits, strong regulatory systems, and government-led enteric surveillance programs. Countries such as Germany, France, the U.K., and the Netherlands have well-established public health frameworks that support routine Yersinia diagnostics use and encourage the adoption of innovative PCR delivery methods. These high-sensitivity formulations are particularly appealing for hospital laboratory populations, regulation-conscious labs, and commercial users, improving detection rates and coverage rates.
Technological advancements in Yersinia diagnostics development, such as enhanced multiplex panels, application-targeted delivery, and improved immunoassay grades, are further boosting market potential. European authorities are increasingly supporting research and trials for diagnostics against both routine and specialized needs, strengthening market confidence. The growing emphasis on convenient, rapid options is aligned with the region’s focus on preventive outbreak control and food safety. Public awareness campaigns and promotion drives are expanding reach in both hospital laboratories and commercial laboratories segments, while suppliers are investing in molecular assays and novel variants to increase efficacy.
Asia Pacific Yersinia Diagnostics Market Trends
Asia Pacific is likely to be the fastest-growing market for yersinia diagnostics in 2026, driven by rising enteric infection awareness, increasing government initiatives, and expanding application programs across the region. Countries such as China, India, Japan, and South Korea are actively promoting diagnostics campaigns to address foodborne growth and emerging surveillance needs. Yersinia diagnostics are particularly attractive in these regions due to their scalable administration, ease of adoption, and suitability for large-scale hospital laboratories and ELISA drives in both urban and semi-urban populations.
Technological advancements are supporting the development of stable, effective, and easy-to-perform Yersinia diagnostics, which can withstand challenging sample conditions and minimize false-negative dependence. These innovations are critical for reaching domestic labs and improving overall enteric coverage. Growing demand for ELISA, immunoassay, and hospital laboratory applications is contributing to market expansion. Public-private partnerships, increased healthcare expenditure, and rising investment in diagnostics research and lab capacity are further accelerating growth. The convenience of diagnostics delivery, combined with improved detection and reduced risk of outbreak spread, positions it as a preferred choice.
Competitive Landscape
The global yersinia diagnostics market is characterized by strong competition between established multinational diagnostic companies and emerging molecular-focused specialists. In North America and Europe, major players such as Thermo Fisher Scientific and CERTEST BIOTEC maintain leadership positions through extensive R&D investments, broad laboratory distribution networks, and close collaboration with public health surveillance programs. Their portfolios include advanced ELISA and PCR-based assays that support both routine screening and confirmatory testing, strengthening their market presence.
In the Asia Pacific region, regional manufacturers are expanding with cost-effective diagnostic kits, improving access in price-sensitive markets, and increasing overall testing penetration. ELISA-based solutions remain widely adopted due to scalability, affordability, and suitability for high-throughput laboratories, reducing false-negative risks in routine diagnostics. PCR innovations address the time and sensitivity limitations of conventional culture methods, supporting rapid-response testing. Strategic partnerships, acquisitions, and technology collaborations continue to enhance product portfolios, accelerate regulatory approvals, and facilitate faster commercialization across global markets.
Key Industry Developments:
- In October 2025, Hologic, Inc. announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance for its Panther Fusion® Gastrointestinal (GI) Bacterial and Expanded Bacterial Assays. The company also confirmed that the assays obtained CE marking in the European Union in compliance with the In Vitro Diagnostic Regulation (IVDR). Hologic designed these highly sensitive molecular tests to rapidly detect the most common bacterial pathogens responsible for infectious gastroenteritis.
- In January 2024, ELITechGroup announced the launch of the GI Bacterial PLUS ELITe MGB® Kit as a new addition to its diagnostic portfolio. The company developed the in vitro assay specifically for diagnosing gastrointestinal bacterial infections. The kit targeted major bacterial pathogens, including Campylobacter spp., Clostridium difficile, Salmonella spp., Shigella spp., and Yersinia enterocolitica. ELITechGroup strengthened its molecular diagnostics offerings with this launch and expanded its capabilities in the rapid and accurate detection of gastrointestinal infections.
- In February 2023, Thermo Fisher Scientific announced that its Applied Biosystems TaqPath PCR kits for infectious diseases had received licensing approval from the Central Drugs Standard Control Organisation (CDSCO) in India. The kits were manufactured in India in collaboration with Mylab Discovery Solutions to support local diagnostic needs. The PCR assays were validated for clinical and analytical use in disease screening, diagnosis, therapeutic monitoring, and genetic risk assessment.
Companies Covered in Yersinia Diagnostics Market
- Thermo Fisher Scientific
- Abbott Laboratories
- Demeditec Diagnostics GmbH
- Launch Diagnostics Limited
- ELITech Group
- Advanced Molecular Diagnostics
- SeraCare (LGC Clinical Diagnostics)
- Hygiena LLC
- Diagnostiki Athinon
- QED Bioscience Inc.
Frequently Asked Questions
The global yersinia diagnostics market is projected to reach US$4.2 billion in 2026.
Molecular diagnostics adoption is rising rapidly as advanced technologies such as PCR and NGS enable faster, highly accurate detection of infectious, genetic, and cancer-related diseases.
The yersinia diagnostics market is poised to witness a CAGR of 5.0% from 2026 to 2033.
Food safety testing is expanding due to rising consumer awareness, stricter regulations, and complex global supply chains that increase contamination risks and drive more rigorous quality control measures.
Thermo Fisher Scientific, CERTEST BIOTEC, Demeditec Diagnostics, TestLine Clinical Diagnostics, and Hygiena LLC are the key players.








