- Nutraceuticals & Functional Foods
- Forskolin Market
Forskolin Market Size, Share, and Growth Forecast, 2025 - 2032
Forskolin Market By Purity (Low (<20%), Medium (20–50%), Others), Application (Dietary Supplements, Pharmaceuticals, Others), Form, and Regional Analysis for 2025 - 2032
Forskolin Market Size and Trends Analysis
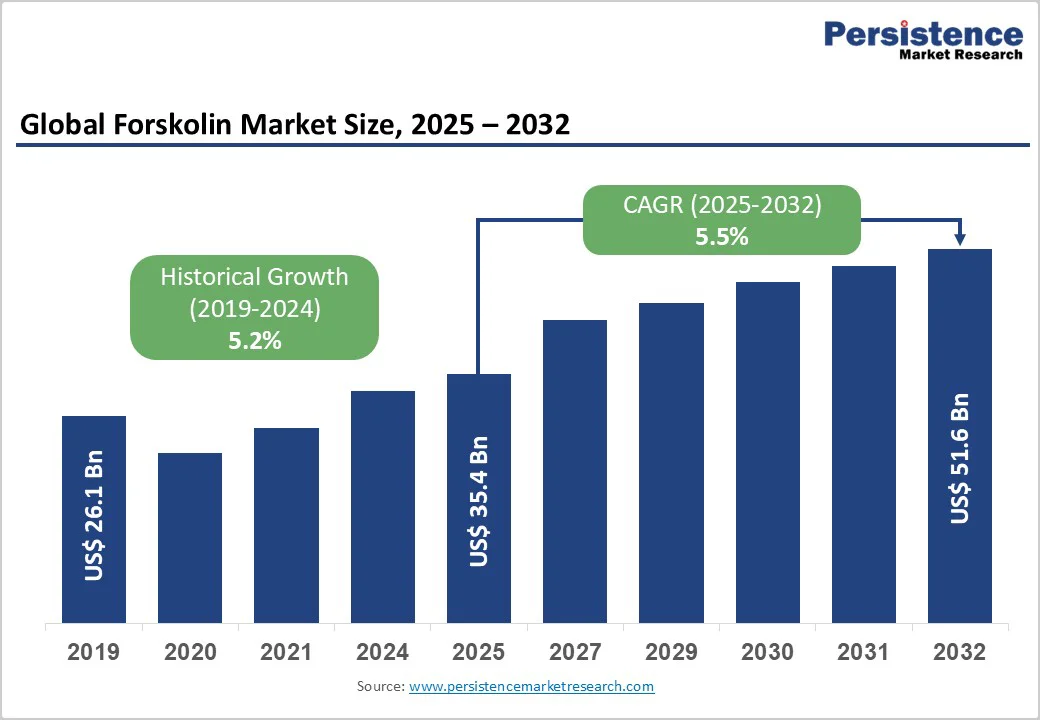
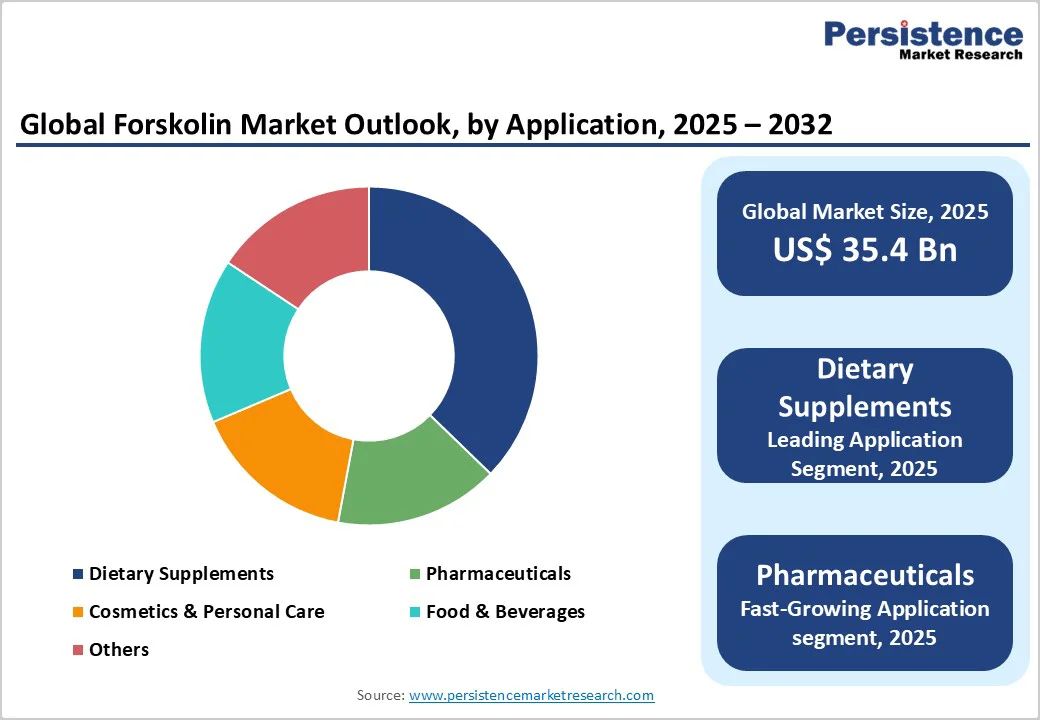
The global forskolin market size is likely to be valued at US$35.4 Billion in 2025 and is expected to reach US$51.6 Billion by 2032, growing at a CAGR of 5.5% during the forecast period from 2025 to 2032, driven by rising adoption of natural nutraceuticals, expanding pharmaceutical and cosmetic research focused on forskolin’s biological activity, and improvements in extraction efficiency and supply-chain scale across key producing regions.
These factors collectively elevate both volume and value across purity grades. Europe and Asia Pacific currently lead in market presence, while North America displays the fastest pace of adoption for clinically oriented and high-purity formats.
Key Industry Highlights
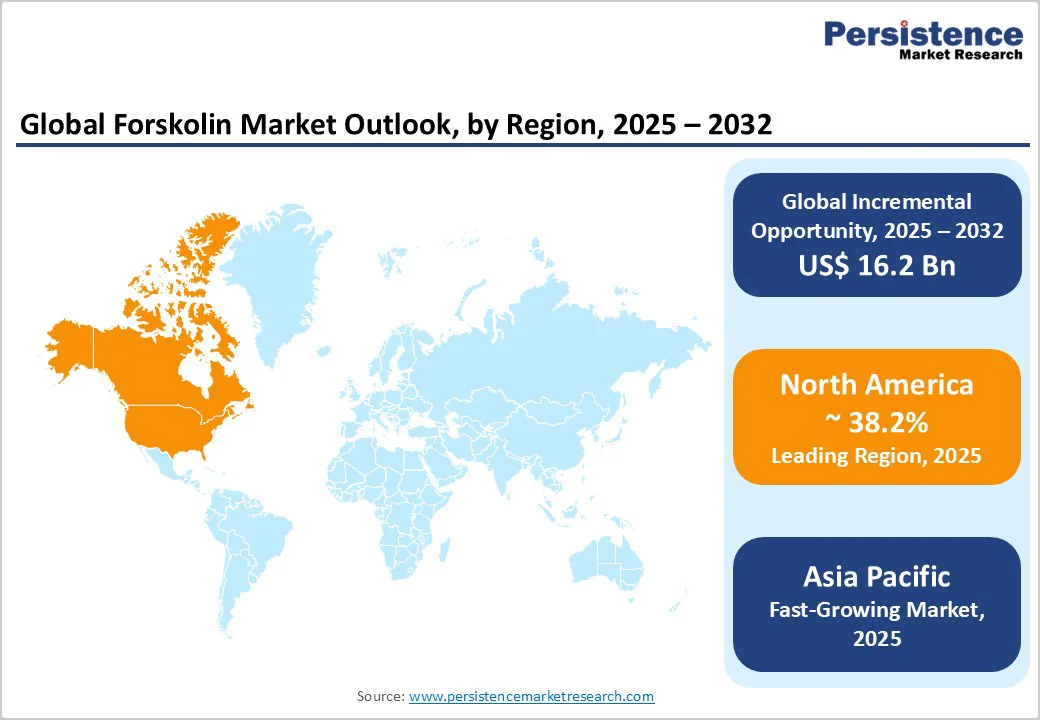
- Leading Region: North America accounted for approximately 38.2% of global market revenue in 2025, supported by high supplement spending, advanced delivery-system adoption, and strong regulatory oversight.
- Fastest-growing Region: Asia Pacific, driven by large-scale cultivation in India and China, expanding certified manufacturing capacity, and rising export-oriented production.
- Dominant Purity: High-purity extracts captured about 67.8% of the total market value due to their use in pharmaceutical and clinical-grade formulations requiring strict potency and quality validation.
- Leading Applications: Dietary supplements contributed approximately 37.6% of total demand, remaining the primary application segment supported by retail expansion, e-commerce penetration, and steady consumer adoption.
| Key Insights | Details |
|---|---|
|
Forskolin Market Size (2025E) |
US$35.4 Bn |
|
Market Value Forecast (2032F) |
US$51.6 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
5.5% |
|
Historical Market Growth (CAGR 2019 to 2024) |
5.2% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Growth Analysis - Increasing Preference for Natural Weight-Management and Wellness Supplements
Consumer movement toward plant-based health solutions has strengthened the position of forskolin within metabolic-health and weight-management formulations. Demand for botanical ingredients has been growing at steady annual rates across supplement channels, and forskolin remains a recurring inclusion within these formulations. Brands continue to diversify delivery formats through capsules, blends, powders, and enhanced bioavailability formats, supporting consistent consumption across retail and online platforms.
Expanding Pharmaceutical and Therapeutic Research Pipeline
Scientific interest in forskolin’s mechanism, particularly its influence on cyclic AMP signaling, has driven R&D across cardiovascular, respiratory, and metabolic indications. As pharmaceutical experimentation increases, demand for high-purity and GMP-compliant forskolin rises. Successful mid-stage clinical validation would expand the addressable pharmaceutical ingredient market substantially, positioning forskolin as a value-intensive botanical active with premium pricing potential.
Improved Cultivation and Extraction Efficiencies
Advances in cultivation methods such as controlled-environment farming and aeroponics have improved consistency, yield, and purity of extractable forskolin. Extraction innovation has reduced batch variability and minimized impurities, enabling producers to supply higher-quality material suited for regulated applications. These improvements strengthen supply stability and reduce production barriers, encouraging greater adoption in premium end-use categories.
Barrier Analysis - Increasing Regulatory Scrutiny of Claims and Labeling
Authorities across major regions maintain strict oversight on botanical health claims, especially those referencing metabolic or cardiovascular benefits. This requires manufacturers to invest in validated research, compliant labeling, and regulatory documentation. Smaller producers face elevated compliance costs and slower launch cycles, which can limit competitiveness in claim-driven product segments.
Variability in Purity, Quality, and Adulteration Risks
Forskolin extracts range from low to high purity, and inconsistencies in raw material quality or extraction standards can lead to variations that negatively affect buyer confidence. Concerns over adulteration further increase the need for validated certificates of analysis and traceable sourcing. Premium buyers’ demand documented quality assurance, creating a performance gap between certified suppliers and commodity-grade producers.
Opportunity Analysis - Pharmaceutical Partnerships and Clinical Validation Pathways
As clinical research progresses, pharmaceutical collaborations represent a significant revenue opportunity. Even moderate adoption within therapeutic formulations could expand premium-segment demand by large magnitudes. Manufacturers positioned with high-purity, GMP-certified supply will be best placed to capture value if clinical milestones accelerate during the forecast period.
Growth in Cosmetic and Topical Applications
The natural cosmetic and personal care segment provides attractive mid-purity volume potential due to rising demand for botanical actives with anti-inflammatory or skin-enhancing properties. If cosmetic applications capture even a modest share of total revenue, they would represent a meaningful, margin-accretive segment with stable year-round demand supported by private-label and contract manufacturing models.
Manufacturing Scale and Export Expansion in Asia Pacific
Asia Pacific holds structural advantages in cultivation, extraction capacity, and cost efficiency. As producers adopt globally recognized certifications and invest in standardized processing, export volumes to North America and Europe continue to expand. Certified supply chains enable manufacturers to secure large-scale private-label and ingredient contracts, reinforcing the region’s long-term production leadership.
Category-wise Analysis
Purity Insights
High-purity forskolin extracts hold the largest share of 67.8% market value, since they are essential for regulated applications where dose accuracy and pharmacological consistency are mandatory. These grades are widely used in clinical trial materials, pharmaceutical formulations, and premium nutraceutical blends. High-purity extracts often undergo multiple refinement steps such as CO2 extraction, chromatographic filtering, and batch-level potency validation. Brands developing cardiovascular research supplements, as well as contract manufacturers supplying material for investigational respiratory therapies, frequently rely on these high-purity variants. Their elevated cost per kilogram reflects both the processing complexity and the stringent documentation required for regulated end uses.
Medium-purity grades are expanding at the fastest pace as they strike a practical balance between potency and affordability. This concentration range aligns with the formulation needs of mass-market dietary supplements, functional blends for sports nutrition, and cosmetic formulations where forskolin is incorporated as a supportive botanical active. Examples include mid-range weight-management capsules carried by leading online retailers, as well as cosmetic manufacturers developing skin-firming creams that incorporate botanically derived actives at moderate concentrations. The rise of third-party testing, clean-label certification, and improved supply-chain traceability is increasing confidence in medium-purity material, boosting its adoption among mainstream brands.
Applications Insights
Dietary supplements remain the dominant application with a market share of 37.6% due to widespread consumer familiarity, extensive retail availability, and forskolin’s long-standing use in weight-management and metabolic-support formulations. The category benefits from high brand turnover, frequent product launches, and growing visibility on e-commerce platforms. Capsules, softgels, and multi-ingredient blends often incorporate forskolin with complementary botanicals such as green tea extract, ashwagandha, or L-carnitine to support broader wellness claims. Major supplement labels and private-label manufacturers continue to introduce standardized forskolin offerings, helping maintain consistent repeat purchases and strengthening the category’s contribution to overall market demand.
Pharmaceutical applications exhibit the fastest projected growth as forskolin undergoes expanded evaluation across cardiovascular, respiratory, and metabolic research pathways. High-purity extracts are incorporated into controlled-study formulations examining outcomes such as bronchodilation, intraocular pressure reduction, and smooth-muscle relaxation. For example, forskolin’s interaction with adenylate cyclase pathways has supported exploratory research in glaucoma management and cardiovascular modulation. Ongoing investigations by clinical research groups and university-affiliated laboratories are improving the scientific understanding needed for possible therapeutic adoption. As evidence accumulates and formulation acceptance improves, pharmaceutical demand is expected to accelerate.
Regional Insights
North America Forskolin Market Trends - Evidence-Backed Supplement Demand and Regulatory-Driven Transparency
North America is the dominant market, holding a share of 38.2%, due to advanced nutraceutical consumption patterns and strong demand for botanical ingredients supported by clinical research. The United States anchors regional growth due to high per-capita supplement spending, the rise of subscription-based D2C brands, and early adoption of delivery formats such as liposomal liquids, micellar suspensions, and controlled-release capsules. Pharmaceutical interest also supports demand for GMP-certified, high-purity forskolin, particularly in research exploring cardiovascular and respiratory pathways.
Regulatory scrutiny from agencies such as the U.S. FDA and Health Canada strongly influences market behavior. Companies prioritizing transparent labeling, validated claims, and third-party certifications (e.g., USP, NSF, Informed Choice) are better positioned to secure shelf space in premium retail chains. The competitive ecosystem includes ingredient suppliers, specialty contract manufacturers, and supplement brands focusing on purity and evidence-backed formulations. Investment activity centers on traceability platforms, clinical partnership programs, and dual sourcing strategies to reduce dependence on single-country raw material suppliers.
Recent examples include North American supplement brands launching standardized 95 percent forskolin extracts with enhanced bioavailability claims, contract manufacturers expanding liquid-extract production lines, and ingredient distributors forming partnerships with Indian and Thai cultivation networks to secure long-term supply agreements. Companies are also integrating QR-based transparency systems that allow consumers to verify batch reports, a trend gaining traction across premium botanical categories.
Europe Forskolin Market Trends - Pharmacy-Led Botanical Adoption and High-Purity Compliance Standards
Europe maintains a substantial share of global forskolin revenue due to its strong consumer preference for natural, clinically supported ingredients and its mature regulatory framework governing botanical supplements and cosmetics. Germany and the U.K. serve as core markets with established pharmacy-led retail ecosystems, while France and Spain are experiencing growing adoption within botanical skincare and wellness categories.
The region places emphasis on traceability, toxicology documentation, and compliance with standards such as EFSA guidelines and COSMOS certification, favoring suppliers with advanced quality-control systems. European companies often prioritize premium-grade delivery formats, clinical substantiation, and innovation in topical formulations, especially for cosmetic applications targeting firming, skin-brightening, and microcirculation benefits.
Cross-border movement of certified products benefits from regulatory harmonization under the EU framework, supporting scalable distribution across multiple countries. Recent developments include European cosmetics firms introducing botanical complexes containing forskolin for use in firming and contouring products, ingredient suppliers expanding CO2 extraction facilities to improve purity levels, and supplement brands rolling out pharmacy-exclusive forskolin capsules backed by standardized testing. Several regional distributors have also entered agreements with Asian extraction companies to secure certified, sustainably sourced material that meets EU quality requirements.
Asia Pacific Forskolin Market Trends - Global Extraction Hub with Advanced Standardization and Export Focus
Asia Pacific functions as the core production and supply hub for Coleus forskohlii cultivation and forskolin extraction. India and China dominate raw material supply and extraction capacity due to agricultural scale, cost efficiencies, and established supply chain networks. Japan contributes significantly high-value demand driven by stringent quality expectations, while ASEAN markets, including Thailand, Vietnam, and Indonesia, are emerging as secondary centers for cultivation and small-scale extraction.
Regional growth is supported by abundant raw materials and increased investments in standardized-extraction practices, and expanding certified manufacturing infrastructure aligned with global standards such as ISO, HACCP, and GMP. Export-oriented producers increasingly focus on high-purity grades, adopting advanced processes such as supercritical CO2 extraction, solvent-free purification, and chromatographic refinement to meet stringent expectations in North America and Europe.
Partnerships between Asian extraction companies and global nutraceutical brands are common, enabling knowledge transfer, contract farming programs, and the development of premium-grade ingredients. Recent developments include Indian exporters upgrading facilities to pharmaceutical-grade processing lines, Chinese manufacturers introducing dual-refinement technologies for more consistent purity levels, and collaborative agreements between Japanese nutraceutical firms and ASEAN cultivators to develop high-yield strains of Coleus forskohlii.
Competitive Landscape
The global forskolin market is moderately concentrated among established botanical extract suppliers and specialized nutraceutical ingredient manufacturers. High-purity product lines account for a significant share of total market value, while medium-purity grades support volume expansion. Competitive differentiation centers on extraction expertise, purity consistency, certification standards, clinical data support, and the ability to supply both ingredients and finished dosage formats.
Prevailing strategies include emphasis on high-purity ingredient portfolios, vertical integration from cultivation through extraction, investment in bioavailability-enhancing delivery technologies, and adoption of traceability and third-party certification systems. Collaboration with research institutions for evidence generation supports premium positioning and regulatory compliance.
Key Industry Developments
- In August 2025, Sabinsa Corporation launched six new retail products under its revamped portfolio (America’s Finest, Sanutra, Nature’s Formulary), including a Weight Management formula featuring its branded ingredient ForsLean® plus BioPerine®.
Companies Covered in Forskolin Market
- Sabinsa Corporation
- Sami-Sabinsa Group
- Nutra Green Biotechnology
- Arjuna Natural
- Alchem International
- Numen India
- BGG World
- Natural Remedies
- Vidya Herbs
- Herbo Nutra
- Ekologie Forte
- Lotus Herbals
- BioActives Japan
- Xi’an Natural Field Bio-Technique
- Botanic Healthcare
- Synthite Industries
- BioGen Extracts
- Carrubba Inc.
- Gencor Pacific
- Applied Food Sciences
Frequently Asked Questions
The forskolin market size in 2025 is estimated at US$35.4 Billion.
By 2032, the forskolin market is projected to reach US$51.6 Billion.
Key trends include the growing adoption of high-purity forskolin extracts for pharmaceutical and clinical applications, the expansion of advanced delivery formats such as liposomal and liquid systems, and increased investment in traceability and clean-label certification.
The high-purity segment leads the market, contributing the largest share due to its use in pharmaceutical-grade and clinical formulations requiring strict potency validation.
The forskolin market is expected to expand at a CAGR of 5.5% between 2025 and 2032.
Major companies include Sabinsa Corporation, Arjuna Natural, Alchem International, BGG World, and Nutra Green Biotechnology.







