- Pharmaceuticals
- Biobetters Market
Biobetters Market Size, Share and Growth Forecast for 2025 - 2032
Biobetters Market by Product Type (Erythropoietin Biobetters, G-CSF Biobetters, Interferon Biobetters, Insulin Biobetters, Monoclonal Antibodies Biobetters, Anti-haemophilic Factor, Others), Indication (Diabetes, Cancer, Renal Disease, Neurodegenerative Disease, Genetic Disorders (Haemophilia), Others), Route of Administration (Subcutaneous, Intravenous), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and Region for 2025 to 2032
Biobetters Market Size and Share Analysis
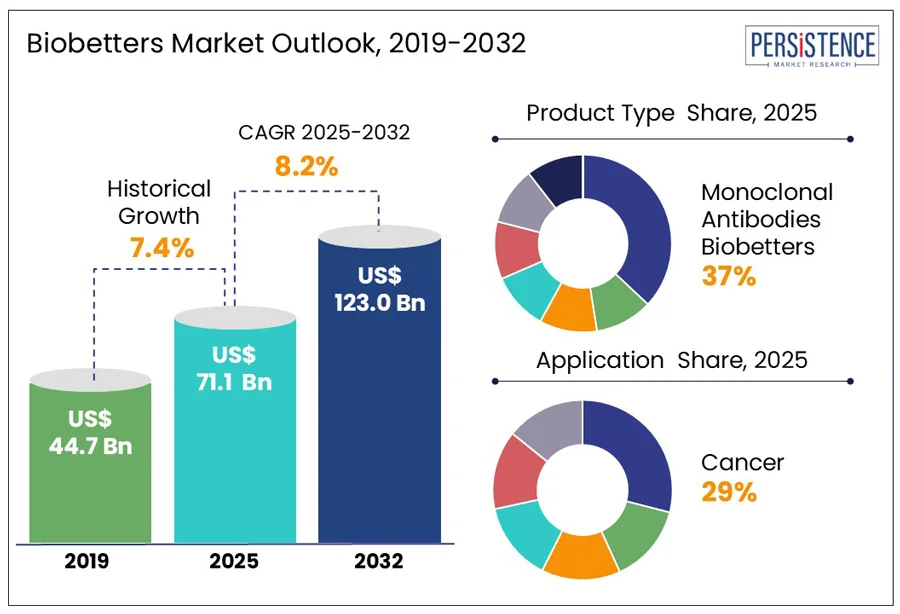
The global biobetters market size is anticipated to reach a value of US$ 71.1 Bn in 2025 and is set to witness a CAGR of 8.2% from 2025 to 2032. The market will likely attain a value of US$ 123.0 Bn in 2032. Increasing investment in developing therapeutic agents with enhanced effectiveness and safety profiles for cancers and autoimmune illnesses has influenced the biobetters market growth dynamics. The increasing investment in developing new drugs is likely to fuel market growth in the near future.
Key Highlights of the Market
- Increased Investment in developing therapeutic agents and demand for enhanced efficacy and safety are significant growth drivers.
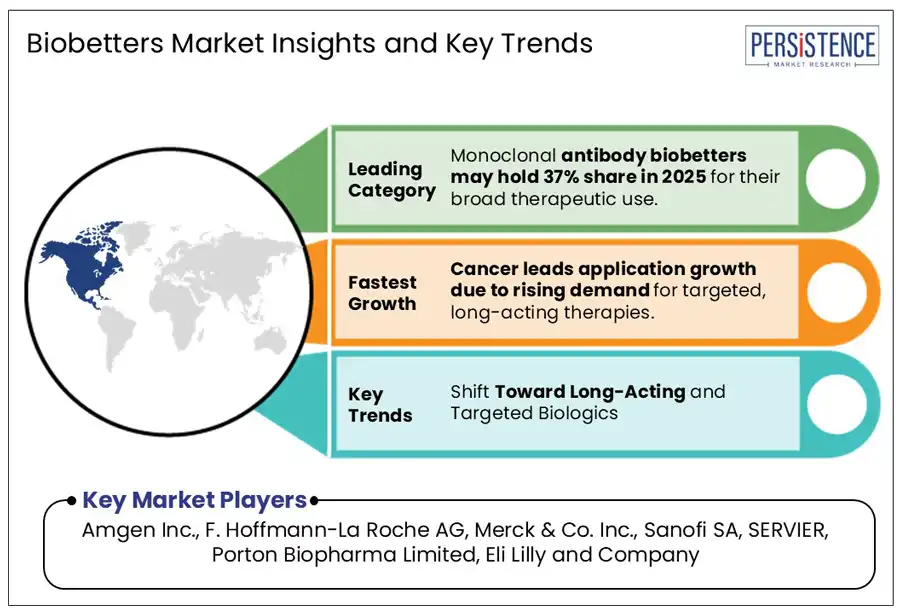
- Monoclonal antibodies biobetters segment maintains dominance in the market and commanded an impressive 37.8% market share in 2025.
- Technological advancements, product innovations, and industry trends are expected to drive market growth.
- Significant growth is expected in regions like North America, Europe, and Asia Pacific, driven by advanced biopharmaceutical infrastructure, and robust research and development capabilities.
- North America is projected to continue its dominance and to account for 45.6% market share in the 2025.
- A significant opportunity in the market lies in incorporating digital tools and data analytics into biobetter therapies to enhance patient monitoring.
- Cancer to be the largest contributor accounting for 28.5% market share.
|
Market Attributes |
Key Insights |
|
Biobetters Market Size (2025E) |
US$ 71.1 Bn |
|
Projected Market Value (2032F) |
US$ 123.0 Bn |
|
Global Market Growth Rate (CAGR 2025 to 2032) |
8.2% |
|
Historical Market Growth Rate (CAGR 2019 to 2024) |
7.4% |
North America Biobetters Market to Retain Top Position
|
Region |
Market Share in 2025 |
|
North America |
45.6% |
North America has consistently maintained a substantial share in the biobetters market, accounting for 45.6% in 2025. North America leads the biobetters market due to its advanced biopharmaceutical infrastructure, robust R&D capabilities, and strong regulatory support.
The region's leading pharmaceutical companies are pioneers in biologic drug development, driving innovation in biobetters which are enhanced versions of existing biologics. Additionally, North America's favorable regulatory environment with agencies like the FDA providing clear guidelines for biobetters accelerates market entry.
Monoclonal Antibodies Biobetters to be Top Products Category
|
Category |
Market Share in 2025 |
|
Product Type - Monoclonal Antibodies Biobetters |
37.8% |
Monoclonal antibodies (mAbs) biobetters are leading in the biobetters market due to their significant therapeutic advantages and broad applications particularly in oncology and autoimmune diseases. This product type to account for 37.8% market share.
These biobetters are engineered to offer enhanced efficacy, improved safety profiles, and longer half-lives compared to original mAbs. The ability to fine-tune their pharmacokinetics and reduce immunogenicity makes them highly effective in targeting specific disease pathways.
The high demand for innovative cancer therapies and the ongoing development of new mAbs with better clinical outcomes drive their prominence in the market. The success of mAbs in treating complex diseases ensures their leading position among biobetters.
Cancer is the Leading Application of Biobetters
|
Category |
Market Share in 2025 |
|
By Application - Cancer |
28.5% |
Cancer is the leading indication in the biobetters market due to the high prevalence of the disease and the critical need for more effective and targeted therapies. Biobetters, particularly monoclonal antibodies and other biologics have shown significant promise in oncology by offering improved efficacy, reduced side effects, and the ability to target cancer cells precisely.
The continuous innovation in cancer treatment coupled with substantial investment in oncology research drives the development of biobetters that can enhance patient outcomes. Moreover, the high mortality rates associated with cancer and the demand for advanced, life-saving treatments contribute to cancer's leadership in the biobetters market.
Market Introduction and Trend Analysis
The biobetters market is experiencing significant growth as a result of the biopharmaceutical industry's shift toward optimizing existing biologic therapies. Biobetters, which are improved versions of original biologic drugs offer advantages such as enhanced efficacy, reduced side effects, longer duration of action, and improved patient compliance.
The market is gaining traction due to the increasing demand for biologics with better clinical outcomes and the need for more cost-effective treatments in the face of patent expirations of first-generation biologics. The regulatory environment is also supportive with agencies like the FDA providing clear pathways for the approval of biobetters encouraging innovation in this space.
Monoclonal antibodies are at the forefront of the biobetters market driven by their effectiveness in treating cancer and autoimmune diseases. The market is also witnessing a shift toward subcutaneous administration, which offers great convenience and enhances patient adherence to treatment regimens.
The growing emphasis on personalized medicine is propelling the development of biobetters tailored to specific patient populations, particularly in oncology. As biopharmaceutical companies continue to invest in research and development and seek to extend the lifecycle of biologics through biobetters, the market is expected to see sustained growth.
Historical Growth and Course Ahead
Biobetters, also known as biosuperiors are recombinant protein drugs from an identical class as a present set of biopharmaceuticals is not similar to the source. It is neither a wholly novel medicine nor a generic version of an old drug. It is a biological product designed to increase clinical efficacy while also improving tolerance and lowering dosing/administration frequency.
The biobetters market is expected to witness a CAGR of 8.6% due to the advantages of biobetters such as superior therapeutic efficacy, less side effects, easy and less expensive production methods, and the availability of high investment prospects.
Market Growth Drivers
Simple and Less Expensive Manufacturing Process
The manufacturing procedure of biobetters is identical to that of biologics. However, modern technologies such as pegylation, and albumin replacement are used as well. Several manufacturers are effectively employing these techniques in the production of different biobetters.
Biobetters have a low development cost and take roughly 10 years to develop, whereas biologics demand a great investment and take around 15 years to develop. While biosimilars have a cost of development that is almost half that of biobetters, their therapeutic effectiveness is far superior.
Increased Investment in Developing Therapeutic Agents
Increasing investment in developing therapeutic agents with enhanced effectiveness and safety profiles for cancers and autoimmune illnesses has influenced the biobetters market growth dynamics. There have been considerable attempts in recent years to reduce the economic impact of chronic illnesses. The increasing investment in developing new drugs is likely to fuel market growth in the near future.
Factors Impeding the Market
High Investments Required for R&D Activities
Developing novel medicinal drugs necessitates large investments in the research and development process, which limits the market growth. However, since the Covid-19 virus outbreak, there has been a huge rise in research and development spending for developing novel biobetters. Apart from that, rising biosimilar competition is also restricting the market growth.
The biobetters market faces significant challenges due to the high investments required for research and development activities. Developing biobetters involves complex processes, including advanced bioengineering techniques, rigorous clinical trials, and extensive regulatory approval procedures, all of which demand substantial financial resources.
High cost acts as a restraint particularly for small biotech firms that may struggle to secure the necessary funding. Additionally, the need to demonstrate clear clinical advantages over existing biologics adds to the R&D burden, as companies must invest significantly to prove efficacy and safety improvements. These financial pressures can slow down innovation and limit market entry affecting the overall growth potential of the biobetters market.
Stringent Regulatory Environment
The stringent regulatory environment is a significant restraint for the biobetters market, as it imposes rigorous standards for development and approval. Regulatory agencies require extensive clinical trials and detailed data to demonstrate that biobetters offer clear advantages over existing biologics. This process ensures safety and efficacy but also leads to lengthy and costly approval timelines.
Companies must navigate complex regulatory pathways and meet high compliance standards, which can be especially challenging for smaller firms with limited resources. The stringent regulatory requirements can thus hinder the speed of innovation and increase the financial burden on companies impacting the overall growth and accessibility of biobetters in the market.
Future Opportunities for the Market Players
Innovations in Production Techniques
Innovations in production techniques such as cell line optimization are revolutionizing the biobetters market by significantly reducing costs and enhancing product quality. By refining cell lines used in the production of biologics, companies can achieve higher yields, great consistency, and improved purity of the final product.
Advanced techniques including gene editing and optimized growth conditions enable efficient protein expression and processing. This optimization not only lowers production costs but also enhances the therapeutic efficacy and safety profiles of biobetters.
The opportunity lies in leveraging these innovations to gain a competitive edge in the market, meet increasing demand for high-quality biologics, and potentially lower the overall cost of treatment for patients.
Incorporating Digital Tools and Data Analytics into Biobetter Therapies
Incorporating digital tools and data analytics into biobetter therapies represents a significant opportunity to enhance patient monitoring and treatment outcomes. By integrating digital health technologies such as wearable devices, mobile apps, and remote monitoring systems, biobetters can be more effectively tracked and managed. These tools enable real-time data collection on patient responses, side effects, and adherence to therapy allowing for more personalized and timely interventions.
Data analytics can provide deeper insights into treatment efficacy and patient behavior leading to improved dosing regimens and tailored therapeutic strategies. This approach not only enhances patient care but also supports efficient clinical trials and post-market surveillance, potentially accelerating the development and adoption of biobetters.
Competitive Landscape for the Biobetters Market
The biobetters market is highly competitive with key players like Amgen Inc., F.Hoffmann-La Roche AG, Merck & Co. Inc. leading the charge. These companies along with Sanofi SA, SERVIER, Porton Biopharma Limited are introducing novel products and leveraging innovative technologies to seize new opportunities.
Industry leaders such as Eli Lily and Company, Novo Nordisk A/S, Biogen Inc. are diversifying their portfolios and expanding their global presence. CSL Behring GmbH, Teva Pharmaceutical Industries Ltd. are also investing significantly in research and development to maintain a competitive edge and drive market growth.
Recent Developments in the Biobetters Market
- In May 2024, Merck KGaA, Darmstadt, Germany, announced that it intends to acquire Mirus Bio, a US-based life science company, for $600 million, or approximately €550 million. This acquisition strengthened Merck KGaA’s offerings in the viral vector manufacturing sector because Mirus Bio specializes in the development and commercialization of transfection reagents.
- In January 2024, The FDA approved a new biosimilar, Zymfentra (infliximab-dyyb) from Celltrion, Inc. Zymfentra a subcutaneous (SC) formulation of Inflectra, a biosimilar to Remicade. It is the world’s first and only subcutaneous infliximab product. Zymfentra is a novel tumor necrosis factor (TNF) blocker indicated in adults for maintenance treatment of moderately to severely active ulcerative colitis following treatment with an infliximab product administered intravenously and moderately to severely active Crohn’s disease.
Companies Covered in Biobetters Market
- Amgen Inc.
- F.Hoffmann-La Roche AG
- Merck & Co. Inc.
- Sanofi SA
- SERVIER
- Porton Biopharma Limited
- Eli Lily and Company
- Novo Nordisk A/S
- Biogen Inc.
- CSL Behring GmbH
- Teva Pharmaceutical Industries Ltd.
- Celltrion Healthcare Co., Ltd.
Frequently Asked Questions
The Global market is estimated to increase from US$ 71.1 Bn in 2025 to US$ 123.0 Bn in 2032.
Improved efficacy, fewer side effects, extended dosing, patent expirations, rising chronic diseases, and growing R&D investments drive the biobetters market.
The market is projected to record a CAGR of 8.2% during the forecast period from 2025 to 2032.
Opportunities include targeting niche indications, enhancing drug delivery, expanding in emerging markets, and leveraging AI for biobetters development and design.
Major players include Amgen Inc., F. Hoffmann-La Roche AG, Merck & Co. Inc., Sanofi SA, SERVIER, Porton Biopharma Limited, Eli Lilly and Company and Others.








