- Pharmaceuticals
- North America Synthetic Opioids Market
North America Synthetic Opioids Market Size, Share, and Growth Forecast, 2025 - 2032
North America Synthetic Opioids Market By Product Type (Synthetic Opioids, Semi-synthetic Opioids), Application (Pain Management, De-addiction), Distribution Channel (Retail Outlets, Hospitals), and Country Analysis for 2025 - 2032
North America Synthetic Opioids Market Size and Trends Analysis
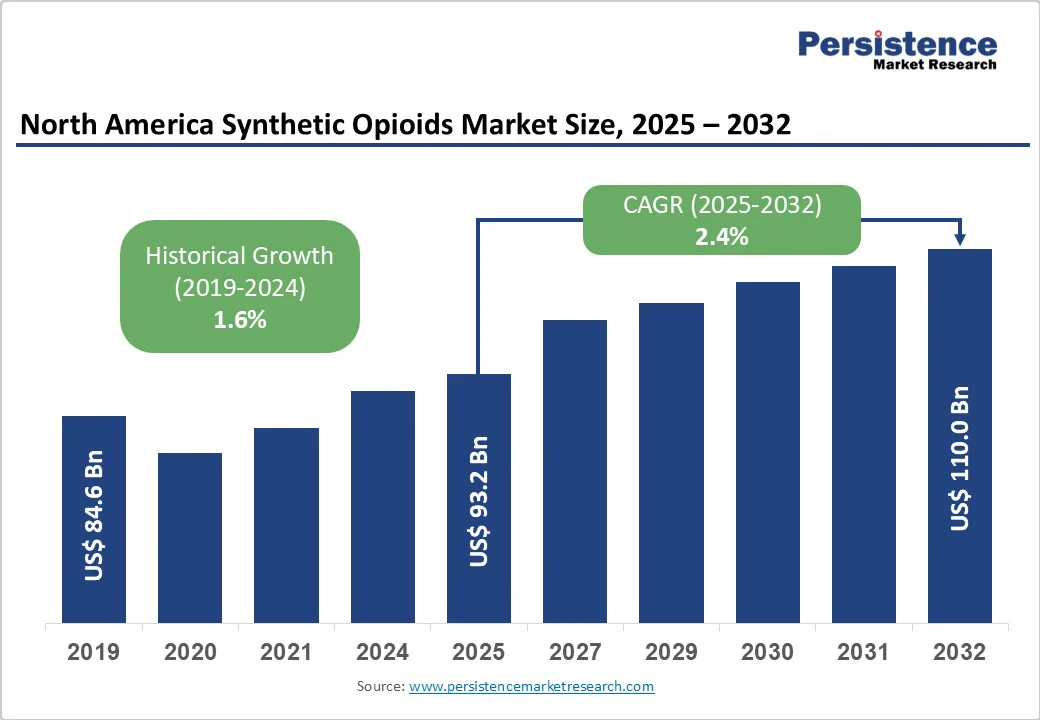
The North America synthetic opioids market size is likely to be valued at US$93.2 Billion in 2025 and is estimated to reach US$110.0 Billion in 2032, growing at a CAGR of 2.4% during the forecast period 2025-2032, driven by the proliferation of fentanyl and its analogs. These substances are often illicitly manufactured and trafficked, complicating enforcement efforts.
Key Industry Highlights
- Leading Country: The U.S., with about a 61.5% share in 2025, backed by high prescription rates and a well-established healthcare infrastructure.
- Fastest-growing Country: Canada, owing to rising opioid-related pain management requirements and expanded harm-reduction initiatives.
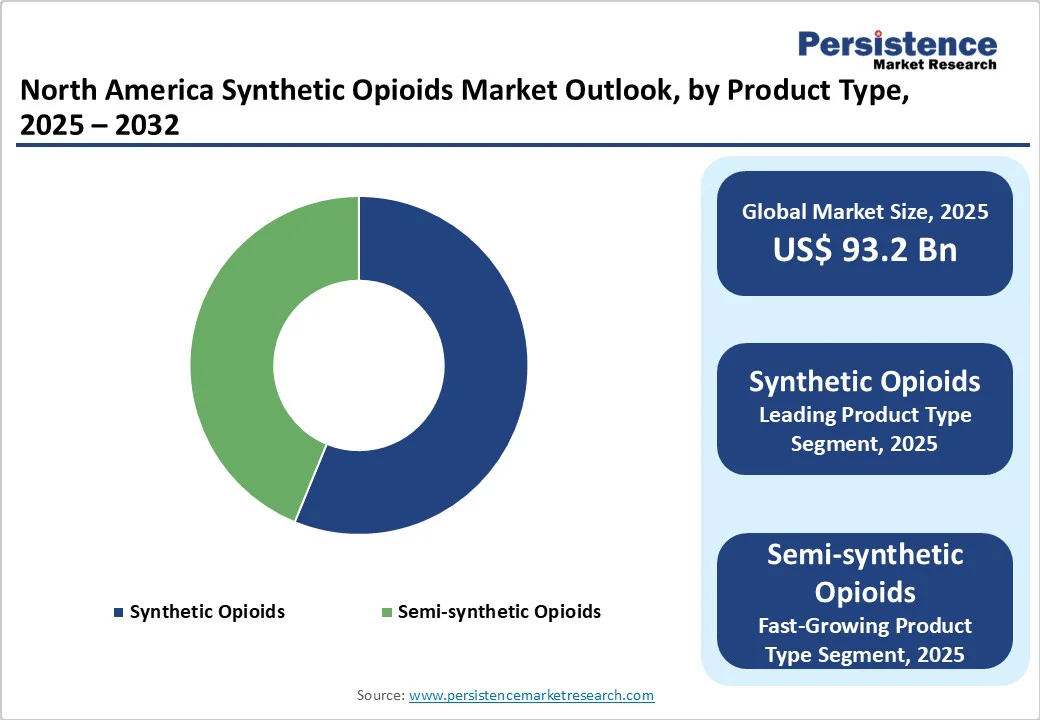
- Leading Product Type: Synthetic opioids hold nearly 56.2% share in 2025, due to their high potency, low production costs, and ease of trafficking.
- Dominant Application: Pain management, approximately 66.8% of the North America synthetic opioids market share in 2025, as synthetic opioids effectively treat acute and chronic pain, including post-surgery and cancer-related pain.
- Key Distribution Channel: Hospitals recorded about 71.4% share in 2025, as they help manage controlled administration for surgeries and trauma care, ensuring patient safety and monitoring for side effects.
- Public Health and Harm Reduction Measures: The FDA's approval of the Alltest Fentanyl Urine Test Cassette marked a milestone as the first over-the-counter test for preliminary fentanyl detection in urine. This development improves accessibility to harm reduction tools, empowering individuals to identify fentanyl presence in substances.
| Key Insights | Details |
|---|---|
|
North America Synthetic Opioids Market Size (2025E) |
US$93.2 Bn |
|
Market Value Forecast (2032F) |
US$110.0 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
2.4% |
|
Historical Market Growth (CAGR 2019 to 2024) |
1.6% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Growth Analysis - Surging Incidence of Chronic Pain
The rising prevalence of chronic pain in North America has significantly influenced the demand for synthetic opioids. Conditions such as osteoarthritis, back pain, and neuropathic disorders have become more common due to an aging population and lifestyle factors. In 2023, around 24.3% of adults in the U.S. reported experiencing chronic pain, with 8.5% experiencing high-impact chronic pain that frequently limited life or work activities.
Women are more likely to experience chronic pain, with 25.4% reporting it compared to 23.2% of men. This surge in chronic pain cases has led to an increased reliance on opioid prescriptions, including synthetic opioids such as fentanyl, as part of pain management strategies. However, the effectiveness and safety of long-term opioid use remain subjects of ongoing research and debate.
Rising Cancer Incidence and Related Pain Management
The increasing number of cancer diagnoses has contributed to a high demand for effective pain management solutions. In 2023, an estimated 1,958,310 new cancer cases and 609,820 cancer-related deaths occurred in the U.S. alone. In addition, breast cancer rates in women under 40 have been rising, with significant geographic variations, particularly high in New York, New Jersey, and Connecticut.
Synthetic opioids, mainly fentanyl, are frequently prescribed for managing moderate to severe cancer-related pain. These opioids are considered essential in palliative and end-of-life care to alleviate suffering. The challenge lies in balancing effective pain relief with the risk of opioid misuse and dependence, necessitating careful prescribing practices and monitoring.
Barrier Analysis - Bilateral Tensions over Tariff Threats
In early 2025, the U.S. administration imposed a 25% tariff on all Mexican imports, citing insufficient action against fentanyl trafficking. This move strained U.S.-Mexico relations, with Mexico deploying 10,000 National Guard troops to combat fentanyl smuggling.
Despite these efforts, only about 51,000 fentanyl pills were seized, whereas U.S. authorities intercepted over 400,000. It highlights challenges in enforcement and the requirement for more effective cooperation.
Deployment of Unique Cargo Scanners
The U.S. has significantly improved border security by deploying AI-based cargo scanners such as multi-energy portals to detect fentanyl shipments. These scanners have increased cargo inspection efficiency.
They allow Customs and Border Protection to scan nearly six times as much freight daily. However, the sheer volume of cross-border trade and the sophistication of smuggling operations continue to pose key challenges.
Opportunity Analysis - Nationwide Medicaid Coverage for Fentanyl Test Strips
In 2025, the U.S. extended Medicaid coverage to include Fentanyl Test Strips (FTS) across all 50 states. This policy aims to improve harm-reduction efforts by enabling individuals to test illicit substances for fentanyl contamination, thereby reduce the risk of overdose. The integration of FTS into Medicaid programs facilitates broad access to these tools, supporting public health initiatives and community-based organizations in their overdose prevention strategies.
By identifying fentanyl presence, users can make informed decisions, potentially decreasing the demand for illicit synthetic opioids. However, challenges persist in ensuring equitable distribution and addressing legal barriers in certain jurisdictions. Despite these obstacles, the nationwide coverage represents a major step toward comprehensive harm reduction in the opioid crisis.
FDA Approval of Nalmefene Nasal Spray for Overdose Reversal
In May 2023, the U.S. Food and Drug Administration (FDA) approved Opvee, a nalmefene hydrochloride nasal spray, for the emergency treatment of opioid overdoses. Nalmefene is an opioid receptor antagonist that provides a longer duration of action compared to naloxone, potentially providing extended protection against opioid-induced respiratory depression. This approval introduces an additional tool for first responders and harm reduction programs, improving their capacity to address opioid overdoses effectively.
The availability of nalmefene nasal spray complies with the FDA's Overdose Prevention Framework, aiming to reduce opioid-related fatalities through diversified treatment options. While nalmefene presents a promising development, its integration into clinical practice and community settings requires further evaluation to establish its comparative efficacy and safety profile.
Category-wise Analysis
Product Type Insights
Synthetic opioids are poised to capture a share of nearly 56.2% in 2025, owing to their high potency and low production costs. Fentanyl is approximately 50 to 100 times more potent than morphine, making it attractive for illicit production and distribution. Its small size allows for easy transport, and it can be synthesized in clandestine labs without the requirement for large-scale farming. These factors contribute to its widespread availability and use in both medical and illicit contexts.
Semi-synthetic opioids, including oxycodone and hydrocodone, are derived from natural opiates but are chemically modified to improve their effectiveness. They are commonly prescribed for pain management and have a well-established presence in the medical community. Their growth is propelled by their efficacy in treating moderate to severe pain and their familiarity among healthcare providers. However, the potential for misuse and addiction has led to increased scrutiny and regulation.
Application Insights
Pain management is speculated to hold around 66.8% of the share in 2025, backed by its effectiveness in treating acute and chronic pain conditions. Opioids interact with the central nervous system to alleviate pain, providing relief for patients undergoing surgery, injury, or suffering from conditions such as cancer. Despite concerns over addiction and overdose, opioids are often considered essential for managing severe pain when other treatments are insufficient.
De-addiction is a major application for synthetic opioids, especially methadone and buprenorphine, which are used in Medication-Assisted Treatment (MAT) for opioid use disorder. These medications help reduce withdrawal symptoms and cravings, supporting individuals in their recovery journey. MAT has been shown to improve treatment retention and reduce the risk of overdose, making it a cornerstone of addiction treatment strategies.
Distribution Channel Insights
Hospitals are projected to lead with a share of approximately 71.4% of share in 2025, spurred by their role in acute pain management during surgeries, trauma care, and post-operative recovery. Healthcare providers in hospitals are trained to administer opioids safely and monitor patients for adverse effects. The controlled environment allows for appropriate dosing and management of potential side effects, ensuring patient safety.
Retail outlets, including pharmacies, are key distribution channels of synthetic opioids as they dispense prescription medications to patients for home use. Pharmacies play a key role in ensuring that prescriptions are filled accurately and that patients receive proper counseling on medication use. However, the accessibility of opioids through retail outlets has raised concerns about misuse and diversion, leading to increased regulation and monitoring.
Country Insights
U.S. Synthetic Opioids Market Trends
In 2025, the U.S. is predicted to account for approximately 61.5% of the share. Synthetic opioids, specifically fentanyl, have drastically impacted the U.S. healthcare system, leading to increased overdose deaths, emergency room visits, and challenges in treatment. In 2022, nearly 74,000 deaths involved synthetic opioids, predominantly fentanyl, marking a substantial rise from previous years. This surge has overwhelmed emergency departments and strained healthcare resources.
The healthcare system has responded by expanding access to naloxone, an opioid overdose reversal drug, and increasing the availability of medication-assisted treatments such as buprenorphine and methadone. These interventions have contributed to a decline in overdose deaths in certain areas. For instance, San Francisco reported a 54% decrease in overdose deaths in October 2024 compared to the previous year, attributed to improved access to addiction treatment medications and harm-reduction strategies.
Canada Synthetic Opioids Market Trends
In Canada, synthetic opioids, especially fentanyl and its analogs, have become a key public health concern. These substances are primarily illicitly produced and have contributed to a substantial increase in overdose deaths. For instance, in 2023, British Columbia reported 2,511 overdose deaths, approximately a 1,000% increase since the introduction of supervised injection sites in 2003. The pharmaceutical industry in Canada plays an important role in the opioid crisis, both in terms of prescription practices and the regulation of opioid medications.
While fentanyl is a controlled substance under Schedule I of the Controlled Drugs and Substances Act, its illicit use has overshadowed its medical applications. The introduction of tamper-resistant formulations such as OxyNEO aimed to reduce misuse but inadvertently led to increased demand for illicit fentanyl. Canada has hence implemented various harm reduction strategies, including supervised consumption sites and the distribution of naloxone.
Competitive Landscape
In the North America synthetic opioids market, companies such as Purdue Pharma, once a leading player in opioid production, have faced legal challenges and bankruptcy due to their role in the opioid crisis. Purdue's aggressive marketing of OxyContin and other opioids contributed to widespread misuse and overdose deaths, leading to its restructuring as Knoa Pharma in 2023.
In response to the evolving synthetic opioid field, regulatory agencies are adopting more dynamic enforcement strategies. For example, FinCEN's 2024 advisory emphasizes vigilance in identifying suspicious activities related to the procurement of precursor chemicals and manufacturing equipment used in the synthesis of illicit fentanyl.
Key Industry Developments
- In February 2025, the U.S. government imposed duties on synthetic opioid precursor chemicals imported from China, aiming to curb the illicit production of fentanyl and its analogs. This move emphasizes a strategic shift toward addressing the synthetic opioid supply chain at its source.
- In August 2024, the U.S. Food and Drug Administration (FDA) approved Zurnai. It is the first nalmefene hydrochloride auto-injector for the emergency treatment of known or suspected opioid overdose in adults and pediatric patients 12 years of age and older.
Companies Covered in North America Synthetic Opioids Market
- Johnson & Johnson Services, Inc.
- Purdue Pharma L.P.
- Hikma Pharmaceuticals PLC
- AbbVie Inc.
- Sanofi
- Sun Pharmaceutical Industries Ltd
- Pfizer, Inc.
- AstraZeneca Plc.
- Mallinckrodt Pharmaceuticals
- Teva Pharmaceutical Industries Ltd.
- Endo Pharmaceuticals Inc.
- Boehringer Ingelheim International GmbH
Frequently Asked Questions
The North America synthetic opioids market is projected to reach US$93.2 Billion in 2025.
Rising prevalence of chronic pain and surging harm-reduction initiatives are the key market drivers.
The North America synthetic opioids market is poised to witness a CAGR of 2.4% from 2025 to 2032.
Expansion of Medicaid coverage for fentanyl test strips and development of emergency overdose reversal treatments are the key market opportunities.
Johnson & Johnson Services, Inc., Purdue Pharma L.P., and Hikma Pharmaceuticals PLC are a few key market players.







