- Healthcare Services
- Cosmetic CDMO Market
Cosmetic CDMO Market Size, Trends, Share, and Growth Forecast 2026 - 2033
Cosmetic CDMO Market by Service (Contract Development, Contract Manufacturing), Product (Hair Products, Skin Products, Lip Products, Eye Products, Body Hygiene, Oral Hygiene), Form (Solid Form & Powder, Lotions, Creams, Others), and Regional Analysis, 2026 - 2033
Cosmetic CDMO Market Size and Trend Analysis
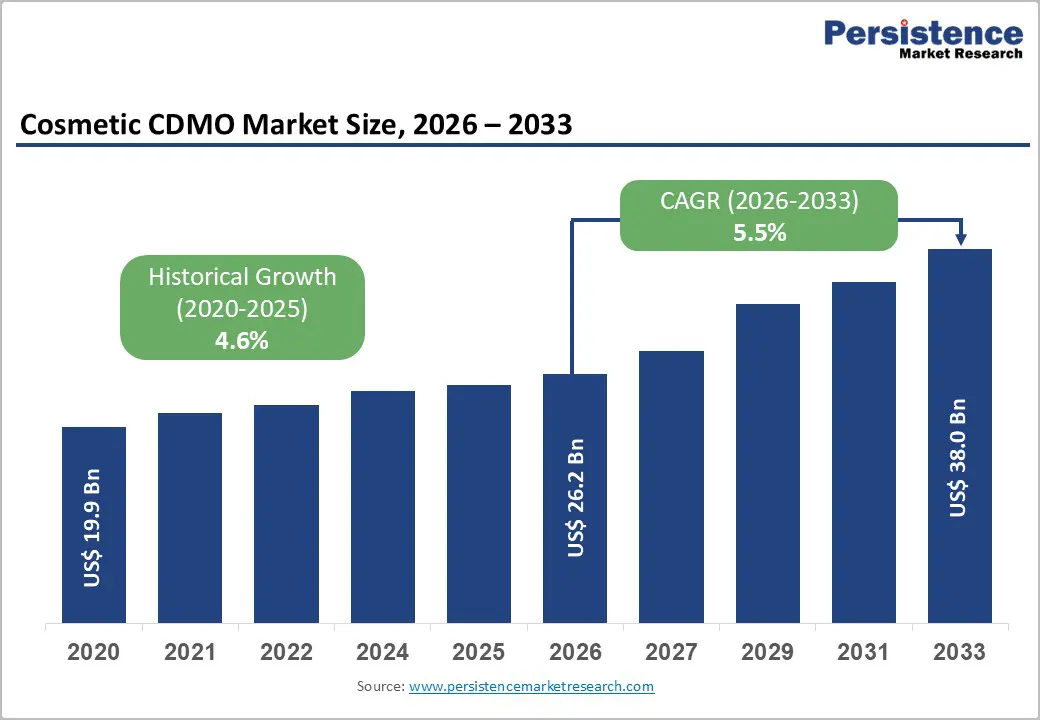
The global cosmetic CDMO Market size is expected to be valued at US$ 26.2 billion in 2026 and projected to reach US$ 38.0 billion by 2033, growing at a CAGR of 5.5% between 2026 and 2033.
The cosmetic contract development and manufacturing organization (CDMO) market is expanding as beauty brands increasingly outsource product development, formulation, and large-scale manufacturing to specialized partners. Growing consumer demand for personalized, premium, and clean-label beauty products is encouraging companies to rely on CDMOs for innovation, quality control, and faster product launches.
These organizations provide expertise in formulation, regulatory compliance, packaging, and scalable production, helping brands manage costs while maintaining high product standards. Rising demand for skincare, haircare, and wellness-oriented products, along with the adoption of sustainable ingredients and advanced manufacturing technologies, is further strengthening the role of CDMOs in the global cosmetics industry.
Key Industry Highlights:
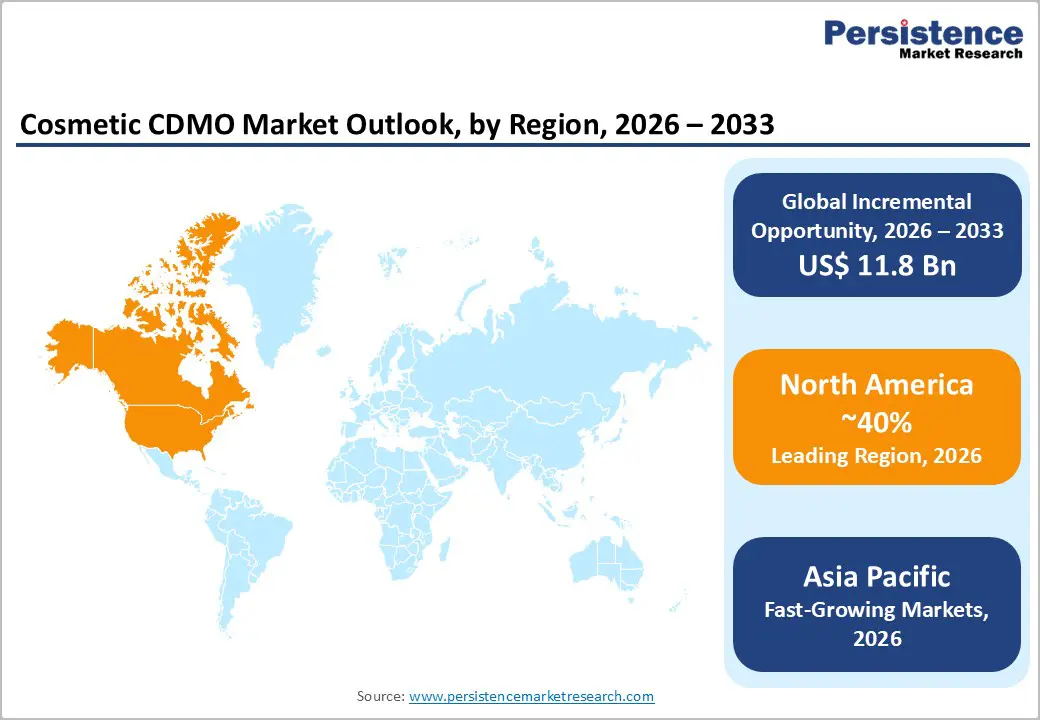
- North America leads the cosmetic CDMO market due to strong premium beauty demand, indie brand growth, and stricter MoCRA regulatory compliance requirements.
- Asia Pacific is the fastest-growing region, driven by expanding OEM/ODM hubs, K-beauty exports, cost-efficient manufacturing, and rising domestic beauty consumption.
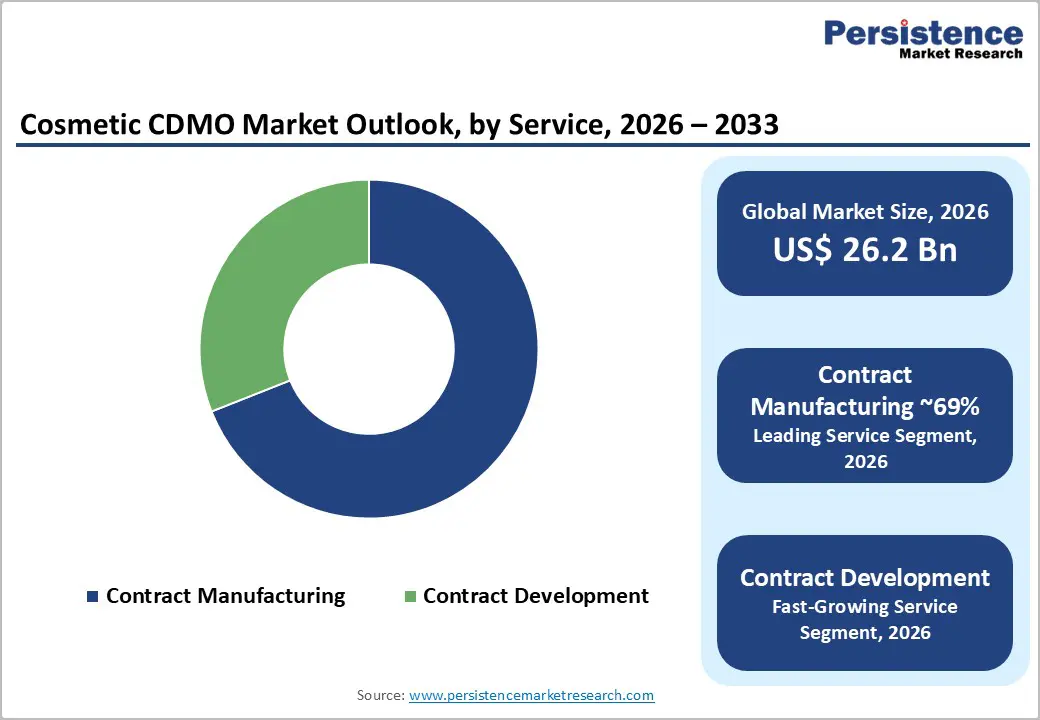
- Contract manufacturing dominates services with about 69% share as brands outsource bulk production, scale-up, testing, and filling and packaging operations.
- Skin products lead the product segment with roughly 35% share, supported by strong demand for dermocosmetics and high-performance skincare formulations.
- Major opportunities lie in clean-label contract development and increased use of Asia Pacific manufacturing hubs for faster launches and cost-efficient global supply.
| Key Insights | Details |
|---|---|
| Cosmetic CDMO Market Size (2026E) | US$ 26.2 billion |
| Market Value Forecast (2033F) | US$ 38.0 billion |
| Projected Growth CAGR (2026 - 2033) | 5.5% |
| Historical Market Growth (2020 - 2025) | 4.6% |
Market Dynamics
Drivers - Lowering Production Costs
In recent years, there has been a noticeable rise in consumer demand for natural/herbal beauty products, which has expanded the use of herbal ingredients in a wider range of cosmetic and personal care products. Businesses are also working harder to transition away from synthetic chemicals and toward natural ingredients, largely in response to rising customer demand and in an effort to locate a sustainable raw material source. Manufacturers are under intense pressure to focus on mass-producing herbal cosmetics to keep up with consumer demand. Businesses are outsourcing their manufacturing processes to third-party CMOs to fulfil this ongoing demand, which also contributes to the industry's rapid CMO expansion in the cosmetics and personal care market.
CMOs also give businesses the ability to focus on their core strengths and increase production throughput while lowering production costs. These organizations have well-known vendor lists, storage lists, large manufacturing facilities, and distribution networks. Due to these benefits, there is an increasing demand for third-party production, particularly of herbal cosmetics, thus providing vast opportunities for growth in the cosmetic CDMO market.
Demand for private label brands has increased due to changes in consumer preferences in the marketplace, which are also expected to provide CMOs with opportunities in the near future. The key reasons for the change are price, accessibility, selection, and reviews & ratings. Major drivers for the increase in demand for private-label brands are e-Commerce and social medicine.
Restraints - Threat of Intellectual Property Theft
One key downside of contract manufacturing is the danger of intellectual property theft. The process entails transferring the patented designs of items to the CMO, which provides the manufacturer and other workers access to patented information. Since the manufacturer might utilize that information without a license, there is some risk involved. Additionally, this might result in the theft or leaking of intellectual property. Suppliers might exploit it to their advantage and potentially become competitors.
Additionally, when a service is outsourced, the company loses control of the process. There is a chance of losing vital production-related knowledge and skills when this control is turned over. This may put the product's quality and other crucial components of production at risk.
Also, a lack of effective communication is another reason why businesses hesitate to outsource their needs. During the production process, this could lead to several issues, including those with schedules, transition plans, internal communications, and capacity qualification, among others. These variables may restrict the expansion of contract manufacturers on a global scale.
Market Opportunities
High-value contract development for specialized and clean-beauty formulations
Growing demand for differentiated, science backed, and clean label products creates substantial opportunities in Contract Development services. Consumers increasingly favor skincare, dermocosmetics, and hybrid products with measurable efficacy and safety, driving brands to invest in advanced actives, microbiome friendly formulations, and dermatologically tested claims. CDMOs with strong R&D capabilities such as Catalent, Inc., whose consumer health division offers specialized topical and beauty formulation expertise across skin, bath & body, hair, fragrance, and color cosmetics are well positioned to capture higher margin projects that extend beyond basic manufacturing. Similarly, niche players like Swiss American CDMO focus on topical skin and wound care products, leveraging ISO 13485 quality systems and FDA registered facilities to deliver OTC and cosmeceutical solutions that command premium pricing and long-term client contracts. This shift toward complex, regulated, and high-performance formulations supports faster growth in Contract Development relative to base level production.
Category-wise Analysis
Service Insights
Within the service category, contract manufacturing is the leading segment, estimated to account for around 69% of the market share in 2026. Most beauty brands, from multinational corporations to indie labels, outsource large portions of bulk preparation, scale up, safety testing, and filling & packaging to specialized plants with validated processes and multi format lines. Major groups such as Fareva Group operate extensive networks of factories across Europe, the Americas, and Asia, offering high-capacity production of personal care and color cosmetics with strict quality control systems and multiple international GMP certifications. Similarly, INTERCOS S.p.A has expanded its hair, body, and skincare capabilities alongside its core makeup business, achieving strong double-digit growth and serving both global and emerging brands. The combination of scale efficiencies, regulatory expertise, and access to state-of-the-art equipment ensures that Contract Manufacturing remains the primary revenue driver in the Cosmetic CDMO Market.
Form Insights
By form, creams are anticipated to be the leading segment, representing approximately 40% share in 2025, while lotions are likely to be among the fastest-growing formats. Creams dominate a wide range of skin and body care applications from facial moisturizers and treatments to body butters and hand creams resulting in consistently high volumes on contract manufacturing lines. These semi-solid emulsions allow CDMOs to incorporate diverse active ingredients, textures, and sensorial properties while utilizing well-established process controls, making them attractive for both mass and prestige brands. Industry reports and company disclosures show that skincare and hair & body categories, which rely heavily on creams and lotions, have been among the quickest-expanding segments for major OEM/ODM players in recent years. As hybrid formats such as tinted moisturizers and treatment creams blur the line between skincare and makeup, demand for flexible, high-volume cream and lotion manufacturing is expected to remain strong.
Regional Insights
North America Cosmetic CDMO Market Trends and Insights
North America is the leading regional market for the Cosmetic CDMO Market, accounting for about 40% of global revenues in 2025. The region benefits from a large and sophisticated beauty and personal care consumer base, with the U.S. ranking among the world’s largest cosmetics markets and showing continued mid single-digit growth. A dense ecosystem of indie brands, dermatology backed lines, and wellness-oriented players drives demand for agile contract partners that can handle small-batch innovation as well as high volume scale-up. CDMOs such as Swiss American CDMO support this environment with FDA registered facilities and ISO 13485 quality systems, enabling development and manufacture of complex topical skin, OTC, and cosmeceutical products for leading U.S. brands.
Regulatory developments further reinforce outsourcing trends. The Modernization of Cosmetics Regulation Act (MoCRA) significantly expands FDA authority over cosmetics, including mandatory facility registration, product listing, safety substantiation, and forthcoming GMP rules scheduled to be finalized by 2025. Compliance with MoCRA pushes many brand owners especially smaller companies to rely on CDMOs with robust quality systems and regulatory teams that can maintain documentation, perform stability and microbiological testing, and manage adverse-event reporting. As a result, North America remains a critical hub for high-value development contracts, regulatory-intensive formulations, and technologically advanced manufacturing in the cosmetic CDMO market.
Asia Pacific Cosmetic CDMO Market Trends and Insights
Asia Pacific region is projected to be the fastest-growing market in the Cosmetic CDMO industry, driven by strong domestic demand, expanding beauty exports, and cost-efficient manufacturing capabilities. Countries such as China, Japan, South Korea, and India have rapidly growing cosmetics industries, supported by rising consumer spending and innovation in skincare and beauty products. Global influence of K-beauty and J-beauty trends continues to shape product development worldwide.
Major OEM/ODM companies such as Kolmar Korea and COSMAX have reported strong revenue growth driven by exports of skincare and color cosmetics for both multinational and indie beauty brands. According to the Ministry of SMEs and Startups in South Korea, small and medium-sized enterprises contribute more than 60% of the country’s cosmetics export value, highlighting the importance of contract manufacturers in helping emerging brands scale globally.
The region is also attracting multinational brands seeking flexible manufacturing capacity, advanced packaging technologies, and competitive production costs. As demand grows for clean beauty, men’s grooming, and hybrid skincare products, Asia Pacific CDMOs are expected to secure a larger share of global outsourcing contracts.
Competitive Landscape
The cosmetic CDMO market is moderately consolidated with the presence of several global and regional players offering formulation development, manufacturing, packaging, and regulatory support services. Leading companies such as Fareva Group, Intercos Group, Kolmar Korea, Cosmax, and Ancorotti Cosmetics dominate the industry through strong R&D capabilities and extensive manufacturing networks. Companies are focusing on strategic partnerships, acquisitions, and capacity expansions to strengthen their market presence and meet rising demand for innovative and sustainable beauty products. Many CDMOs are also investing in clean-label formulations, advanced manufacturing technologies, and customized product development to support emerging beauty brands and accelerate product launches in the highly competitive cosmetics industry.
Key Developments:
- In February 2025, Granules India completed the acquisition of Switzerland-based Senn Chemicals AG for CHF 20 million (approximately INR 192.5 crore). The acquisition marked the company’s entry into the peptide therapeutics segment and expanded its presence in the CDMO sector.
- In March 2023, INTERCOS S.p.A reported net sales of approximately €988 million for 2023, an increase of about 18% year-on-year, with strong growth in skincare and hair & body segments following investments in expanded production capacity and innovation centers.
Companies Covered in Cosmetic CDMO Market
- Catalent, Inc
- Pierre Fabre group
- INTERCOS S.p.A
- Biofarma Srl C.F.
- Fine Foods & Pharmaceuticals NTM S.p.A. (Euro Cosmetics)
- Merck KGaA
- C.O.C. Farmaceutici Srl
- Cosmoproject
- Swiss American CDMO
- Zymo Cosmetics
- Fareva
- PARAGON NORDIC
- Kolmar [Kolmar Korea Holding]
- Lonza (Global)
- Others
Frequently Asked Questions
The Cosmetic CDMO Market is expected to reach approximately US$ 26.2 billion in 2026.
Key drivers include global beauty market growth, increasing indie and niche brands, and stricter regulations requiring GMP compliance, safety testing, and regulatory expertise.
North America leads the Cosmetic CDMO market due to strong premium beauty demand, a growing indie brand ecosystem, and evolving FDA regulations encouraging outsourcing.
Major opportunities include contract development for clean and science-backed formulations and leveraging Asia Pacific OEM/ODM hubs to enable innovation and cost-efficient global manufacturing.
Key players include Catalent, Pierre Fabre Group, INTERCOS, Biofarma, Fareva, Kolmar Korea, Lonza, and several regional OEM/ODM cosmetic specialists.








