
- Pharmaceuticals
- U.S. Viscosupplementation Market
U.S. Viscosupplementation Market Size, Trends, Share, and Growth Forecast, 2025 - 2032
U.S. Viscosupplementation Market By Product (Single Injection Viscosupplementation, Three Injection Viscosupplementation, Five Injection Viscosupplementation), Application, Distribution Channel, and Regional Analysis for 2025 - 2032
U.S. Viscosupplementation Market Size and Trends Analysis
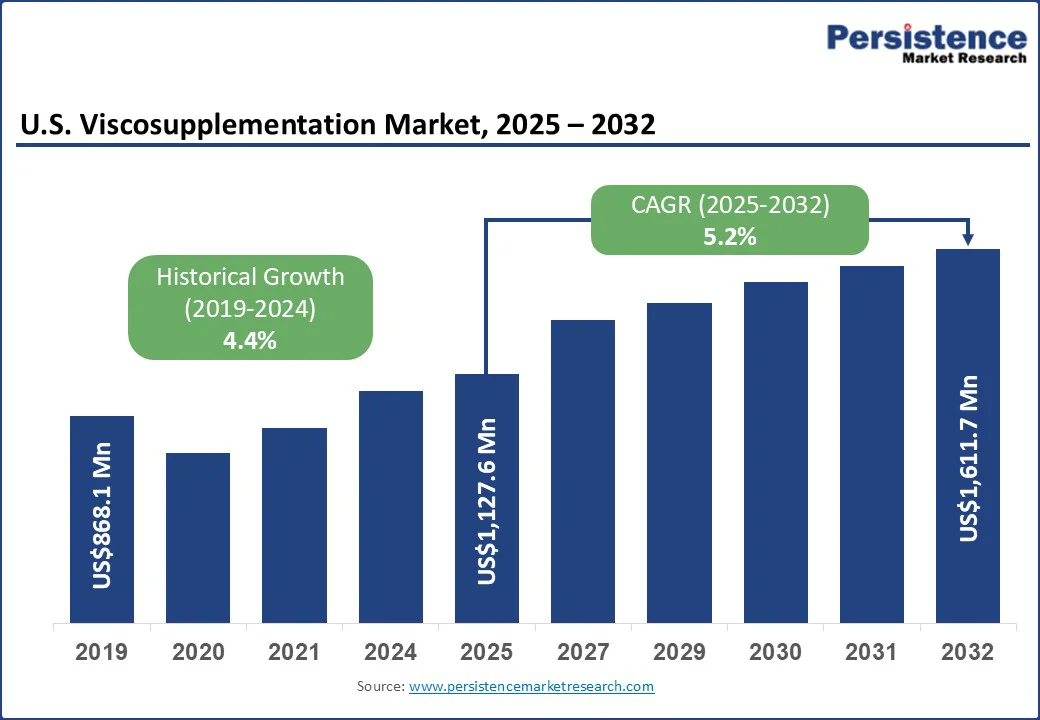
The U.S. viscosupplementation market size is likely to be valued at US$1,127.6 Mn in 2025 and is expected to reach US$1,611.7 Mn by 2032 growing at a CAGR of 5.2% during the forecast period from 2025 to 2032, driven by the rising prevalence of osteoarthritis (OA), a multifactorial joint disease marked by progressive breakdown of articular cartilage, synovial inflammation, and subchondral bone remodeling that leads to pain, stiffness, and impaired mobility.
Key Industry Highlights:
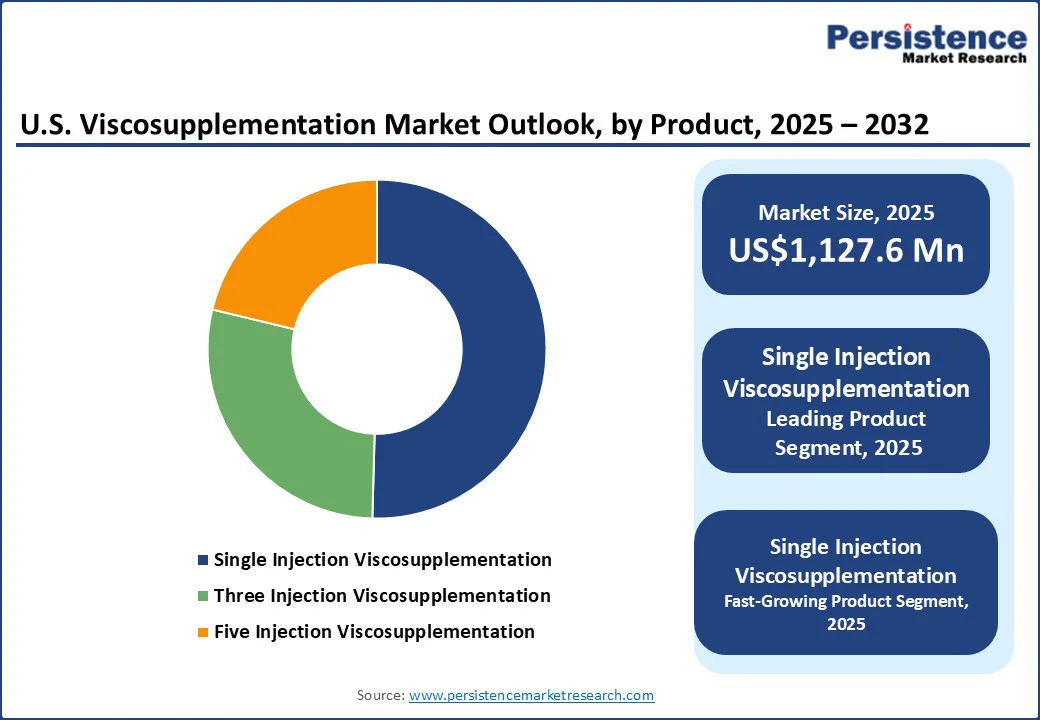
- Leading Product Segment: Single injection viscosupplementation leads the product segment due to convenience, patient compliance, and reduced clinic visits.
- Dominant Application Segment: Knee osteoarthritis remains the dominant application segment, accounting for the majority of viscosupplementation procedures nationwide.
- Opportunity: Combination therapies integrating hyaluronic acid with regenerative or anti-inflammatory agents represent a major innovation opportunity.
- Drivers: Technological advancements in HA formulations, including longer-lasting and crosslinked products, are expanding therapeutic potential and market reach.
| Key Insights | Details |
|---|---|
| U.S. Viscosupplementation Market Size (2025E) | US$1,127.6 Mn |
| Market Value Forecast (2032F) | US$1,611.7 Mn |
| Projected Growth (CAGR 2025 to 2032) | 5.2% |
| Historical Market Growth (CAGR 2019 to 2024) | 4.4% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Increasing Prevalence of Osteoarthritis and Rising Demand for Minimally Invasive Joint Treatments in the U.S.
Osteoarthritis (OA) is the most common type among over 100 types of arthritis, affecting 32.5 million U.S. adults annually. More than half of individuals with symptomatic knee OA are below 65 (Osteoarthritis (OA) Action Alliance, 2024).
About 30% of adults with arthritis find stooping, bending, or kneeling very difficult, while 20% find it very difficult to walk even three blocks or push/pull large objects. The aging population, coupled with lifestyle factors such as obesity, has significantly increased OA cases, with individuals aged 65 and older accounting for nearly 43% of cases.
Additionally, the overall economic burden associated with OA in the U.S. is estimated at US$136.8 Bn annually, more than double over the last decade, surpassing costs associated with tobacco-related health effects, cancer, and diabetes. Direct medical costs reach nearly US$65 Bn annually, and OA contributes to an estimated 1 million knee and hip replacements each year.
This demographic shift is driving demand for effective, non-surgical, or minimally invasive treatment options. Viscosupplementation injections offer a minimally invasive alternative to traditional surgical interventions, aligning with the growing preference for outpatient procedures. Increasing awareness among both patients and healthcare providers is projected to further bolster market growth.
Availability of Alternative Therapies such as Corticosteroids and Physical Therapy
Viscosupplementation, or gel injections of hyaluronic acid, replaces the synovial fluid in osteoarthritic joints, resulting in pain relief, improved joint function, and enhanced joint mobility. Other alternative treatment options, including corticosteroid injections, physical therapy, and non-surgical treatments such as genicular artery embolization (GAE), which are often used in clinical practice, challenge its adoption.
Physical therapy, focusing on strengthening muscles around the joint, remains a cornerstone for managing OA symptoms and improving function. These alternatives often enjoy broader clinical familiarity and may be perceived as more cost-effective, especially within healthcare systems that offer limited reimbursement for viscosupplementation.
Additionally, some patients and providers may choose these therapies due to their established efficacy profiles and lower initial costs, which can hinder the widespread adoption of viscosupplementation.
Growing Demand for Outpatient and Ambulatory Care Settings
Viscosupplementation or HA injections, being minimally invasive in nature, are highly preferred in outpatient clinics and ambulatory care centers. These procedures are typically quick, safe, and require minimal recovery time, making them ideal for non-hospital settings.
With growing efforts to make osteoarthritis treatment cost-efficient, maintain patient convenience, and reduce the overall burden on healthcare facilities, these outpatient settings are becoming preferred treatment destinations.
The increasing involvement of Medicare and private insurers in recognizing the value of outpatient care is supporting broader adoption of viscosupplementation in these environments. For patients with osteoarthritis, particularly knee OA, outpatient care provides an accessible option for pain management without hospitalization or lengthy downtime.
The U.S. remains a priority market for global HA manufacturers due to high adoption and reimbursement opportunities, further reinforcing the growth of viscosupplementation in ambulatory and outpatient settings. This trend creates significant opportunities for manufacturers to collaborate with outpatient networks, strengthen distribution channels, and position their products as the standard of care in these rapidly expanding healthcare environments.
Category-wise Analysis
Product Insights
Single injection viscosupplementation dominates the U.S. viscosupplementation market due to its convenience, reduced clinic visits, and improved patient compliance. Compared to multi-injection regimens, single-injection formulations enhance adherence and minimize discomfort, driving higher adoption among both physicians and patients.
The development of longer-acting hyaluronic acid variants and advanced delivery systems further reinforces this preference, making single-injection therapies the leading segment in terms of sales and market penetration.
Application Insights
Knee osteoarthritis remains the leading application segment in the U.S. viscosupplementation market. As the most prevalent musculoskeletal disorder, OA affects nearly all joints but primarily impacts the knees and other weight-bearing joints, resulting in reduced mobility, decreased productivity, impaired quality of life, and increased disability and morbidity.
The annual incidence of knee OA is highest among adults aged 55 to 64, with approximately one-fourth of arthritis patients experiencing severe joint pain, scoring 7 or higher on a 0-10 pain scale.
Viscosupplementation provides targeted relief from pain and stiffness in the knee while being minimally invasive and effective in delaying surgical intervention, making it a preferred treatment choice. Growing awareness among patients and increasing recommendations from orthopedic specialists continue to reinforce the knee segment’s dominant position in the market.
Regional Insights
U.S. Viscosupplementation Market Trends
The rising prevalence of OA around the globe has led to several research efforts aimed at improving clinical outcomes. Viscosupplementation, consisting of hyaluronic acid (HA) injections, has been shown to restore the physiologic viscoelasticity of synovial fluid.
Current studies are exploring combination therapies that integrate HA with regenerative agents such as platelet-rich plasma (PRP) or anti-inflammatory drugs. These combinations aim to synergistically enhance cartilage repair, reduce inflammation, and provide a more comprehensive treatment approach for OA patients, addressing not only joint lubrication but also promoting tissue healing and slowing disease progression.
Additionally, researchers are investigating the use of natural polymers (HA, chitosan, and alginate) and synthetic polymers (polylactic acid, polylactic-co-glycolic acid, and polycaprolactone) to improve OA therapies. These biomaterials can serve as carriers for targeted drug or gene delivery, support cartilage regeneration through tissue-engineered scaffolds, and enhance joint protection when combined with viscosupplements.
Modifying or crosslinking HA is also being explored to increase its stability and prolong its therapeutic effects. Such innovations are expected to broaden the clinical applications of viscosupplementation, attract a larger patient population, and improve long-term outcomes.
Competitive Landscape
The U.S. viscosupplementation market is moderately competitive with a blend of established global players such as Anika Therapeutics, Bioventus, Seikagaku Corporation, Ferring Pharmaceuticals, Sanofi, Zimmer Biomet, Fidia Farmaceutici S.p.A., and Teva Pharmaceutical Industries Ltd.
These companies leverage their strong distribution networks, extensive product portfolios, and ongoing innovations to maintain significant market share through strategic collaborations and mergers. This helps companies expand their product offerings as well as enhance their market presence.
Key Industry Developments
- In April 2025, Seikagaku Corporation collaborated with Ono Pharmaceutical Co., Ltd. to co-develop and market the Gel-One product, an intra-articular injection, utilizing Seikagaku’s unique cross-linking technology.
- In April 2024, BioSenic, a clinical-stage Belgian-headquartered company specializing in serious autoimmune and inflammatory diseases and cell therapy, shared phase 3 study data on its late-clinical asset, a single injection of JTA-004, to be safe and efficacious, for the treatment of knee osteoarthritis at the Osteoarthritis Research Society International.
Companies Covered in U.S. Viscosupplementation Market
- Sanofi S.A.
- Zimmer Biomet Holdings Inc.
- Bioventus LLC
- Anika Therapeutics Inc.
- Fidia Pharma USA Inc.
- Ferring B.V.
- Seikagaku Corporation
- OrthogenRx Inc. (Channel-Markers Medical)
- Medical Device Business Services, Inc. (J&J)
- AVNS
- Searchlight Pharma Inc.
- Aptissen GmbH
- Teva Pharmaceutical Industries Ltd.
- TRB Chemedica International SA
- IBSA Institut Biochimique SA
Frequently Asked Questions
The U.S. market is projected to be valued at US$1,127.6 Mn in 2025.
Rising osteoarthritis prevalence and demand for minimally invasive joint pain treatments drive the U.S. viscosupplementation market.
The U.S. viscosupplementation market is poised to witness a CAGR of 5.2% between 2025 and 2032.
Innovation in longer-lasting formulations, combination therapies, and the expansion of outpatient and telehealth channels offer significant growth potential for the U.S. viscosupplementation market.
Major players in the U.S. are Sanofi S.A., Zimmer Biomet Holdings Inc., Bioventus LLC, and Anika Therapeutics Inc.







