
- Pharmaceuticals
- Oral Solid Dosage Contract Manufacturing Market
Oral Solid Dosage Contract Manufacturing Market Size, Share, and Growth Forecast for 2025 - 2032
Oral Solid Dosage Contract Manufacturing Market by Dosage Form (Tablets, Capsules, Softgels, Powders, Granules, Gummies, Others), Mechanism (Immediate Release, Delayed Release, Controlled Release), Application (Drug Product Development, Fill & Finish Product Manufacturing, Packaging /Labelling, Others), End user (Pharmaceutical Companies, Biopharmaceutical Companies, Emerging/Virtual Pharma Companies, Nutraceutical Companies), and Regional Analysis for 2025 - 2032
Oral Solid Dosage Contract Manufacturing Market Size and Trends
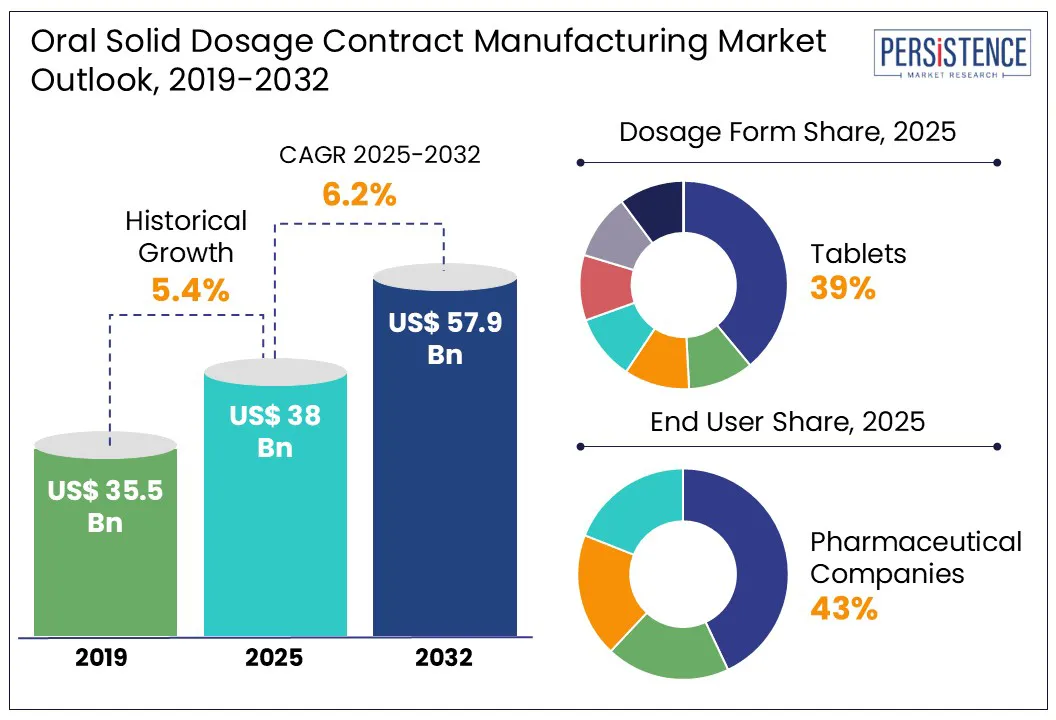
Global oral solid dosage contract manufacturing market is likely to be valued at US$ 38.0 Bn in 2025 and is estimated to reach US$ 57.9 Bn, at a CAGR of 6.2% during the forecast period 2025-2032.
Advancements in drug delivery technology, including targeted dosage forms and 3D printing, are transforming the oral pharmaceuticals market, improving patient outcomes and ensuring precise dosage control.
The production of solid dosage pharmaceuticals is growing because it is efficient and economical; digital technologies improve process monitoring and quality control, while controlled-release technologies increase bioavailability.
With the goal of improving patient adherence, CDMOs (Contract Development and Manufacturing Organizations) are advancing the development of next-generation oral medications using advanced manufacturing technologies.
Key Highlights of the Oral Solid Dosage Contract Manufacturing Market
- The need for cost-effective drug manufacturing is driving pharmaceutical companies to outsource oral solid dosage production to specialized CMOs.
- In 2025, tablets are envisioned to lead the oral solid dosage contract manufacturing market, holding a 39.4% share, reflecting their widespread use due to cost-effectiveness, patient compliance, and ease of production.
- Immediate release formulations are expected to attain a market share of 52.3% in 2025, driven by their rapid drug absorption and effectiveness in treating acute conditions.
- Among end users, big pharma and biotech companies are anticipated to account for 43.4% in 2025, emphasizing their reliance on contract manufacturers for scalable production and expertise.
- For applications, drug product development is projected to hold a 60.3% share, highlighting the growing demand for innovative solid dosage formulations.
- In 2025, North America is predicted to hold a 40.5% share of the global oral solid dosage contract manufacturing industry, fueled by strong pharmaceutical infrastructure and regulatory support.
|
Global Market Attributes |
Key Insights |
|
Oral Solid Dosage Contract Manufacturing Market Size (2025E) |
US$ 38.0 Bn |
|
Market Value Forecast (2032F) |
US$ 57.9 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
6.2% |
|
Historical Market Growth (CAGR 2019 to 2024) |
5.4% |
Historical Growth and Course Ahead
Collaborative Efforts of Healthcare Sector Caters Innovation in Drug Development
The global oral solid dosage contract manufacturing industry witnessed a CAGR of 5.4% in the historical period between 2019 and 2024, as per Persistence Market Research. The need for novel treatments has increased because of the rising incidence of chronic and infectious illnesses. Oral solid dose contract manufacturing is increasing due to major pharmaceutical corporations outsourcing contract production.
In order to increase their market presence and take advantage of common technologies, businesses are concentrating on acquisitions and mergers. For instance,
- Piramal Pharma Solutions and Plus Therapeutics entered into a Master Services Agreement in March 2021 to outsource services for the Plus Therapeutics RNL Liposome Intermediate Drug Product.
Scientific Advances in Oral Solid Dosage Forms to Boost Sales through 2032
In the estimated timeframe from 2025 to 2032, the global market for oral solid dosage contract manufacturing is likely to showcase a CAGR of 6.2%. There are a lot of prospects for oral solid dosage contract manufacturing organizations throughout the world over the projection period. These businesses can concentrate on developing new technologies to enhance the morphological properties, stability, yield, and bioavailability of oral solid dosage forms.
Market players can create a variety of oral solid dosage forms, including controlled release, extended-release, and enteric-coated. For instance, using its GPEx Boost platform, Catalent's Biologics domain provides cell-line development services to produce stable, high-yielding mammalian cell lines for biosimilars.
However, because of the market's fragmentation and plenty of small CMOs, consolidation presents a substantial chance for quicker development. Big and small pharmaceutical and biotech companies are drawn to flexible business models developed by contract development and manufacturing companies. Particle engineering is a tactic that CDMOs can use to increase their market share.
Market Dynamics
Growth Drivers
Enhanced Scalability and Reduced Costs Fueling Demand for OSD CMO
Contract Manufacturing Organizations (CMOs) that specialize in oral solid dosage are becoming increasingly in demand because of their improved scalability, cost-effectiveness, and innovations in medication manufacture, which aid pharmaceutical businesses in streamlining their operations. For instance,
- In March 2024, Catalent expanded its OSD manufacturing facility in Kansas City, enhancing capabilities for high-potency drugs and controlled-release formulations.
- Lonza announced in April 2024 a US$ 100 Mn investment in its Swiss OSD facility, increasing production for oncology and rare disease treatments.
The pharmaceutical industry is predicted to grow significantly worldwide, necessitating the smart outsourcing of OSD manufacturing. Continuous production, automation, and AI-driven quality control are all combined to increase productivity and lower costs, which makes OSD contract manufacturing a fast-growing market.
Market Restraining Factors
Risk of Non-Compliance and Quality Issues as Barriers in OSD Contract Manufacturing
The OSD Contract Manufacturing industry is anticipated to have issues with quality control and regulatory compliance, which might impede market expansion. Production halts, product recalls, and financial penalties can result from strict GMP regulations enforced by organizations such as the FDA, EMA, and PMDA.
Small contract development and manufacturing organizations often struggle with high regulatory costs, complex documentation, and stringent inspections, resulting in longer drug approval timelines and slowed market expansion in developed nations. For instance,
- In 2023, the FDA approved only 37 small-molecule drugs, a decline from 50 in 2021, highlighting increasing regulatory scrutiny.
To mitigate risks, CDMOs are forming strategic partnerships and expanding globally, like Lonza's Singapore expansion in October 2021. However, outsourcing to low-cost markets like India and China presents challenges like supply chain disruptions, regulatory differences, and communication barriers.
Key Market Opportunities
Pharmaceutical Companies Shifts Toward Outsourced Manufacturing Solutions
Pharmaceutical companies are outsourcing manufacturing to CDMOs to improve efficiency, reduce costs, and meet demand for specialized drug formulations due to the complexity of biologics, specialty drugs, and oral solid dosage forms. CDMOs offer advanced technology, regulatory expertise, and large-scale production capabilities. For example,
- In March 2023, Catalent expanded its oral solid dose manufacturing facility in Oxfordshire, U.K., to support the growing demand for modified-release and highly potent drug formulations.
- In December 2023, Lonza announced an approximately US$ 540 Mn investment to expand its manufacturing capacity for biologics and small molecules.
In order to concentrate on their core skills, pharmaceutical firms are outsourcing their manufacturing processes. The biopharma outsourcing industry is expected to expand quickly, and CDMOs are essential to the development of new medicinal products.
Oral Solid Dosage Contract Manufacturing Market Insights and Trends
Dosage Form Insights
Tablets Gain Popularity with Ease of Administration in Healthcare
Tablets are the dominant form in the oral solid dosage contract manufacturing market, accounting for 39.4% of the market share in 2025. They are preferred due to cost-effectiveness, ease of administration, and controlled drug release. Leading pharmaceutical CDMOs, including Catalent, Lonza, and Recipharm, continue to invest in tablet production to meet growing demand. For instance,
- In March 2024, Catalent expanded its solid dose manufacturing capabilities in its St. Petersburg, Florida facility, enhancing production efficiency and formulation innovation.
- In February 2024, Lonza revealed plans to invest in oral medication delivery technology, with a particular emphasis on formulations with controlled release.
Additionally, regulatory agencies such as the FDA and EMA are pushing for enhanced manufacturing processes, driving innovation in coating technologies, bioavailability enhancement, and excipient optimization. With the demand for oral solid dosage drugs across chronic disease management, oncology, and infectious diseases, the tablet segment is expected to maintain its dominant position in the global OSD contract manufacturing market.
End User Insights
Pharmaceutical Companies Pushes OSD Contract Manufacturing Growth
In 2025, Pharmaceutical Companies are expected to account for 43.4% of the end-user market share in OSD contract manufacturing. These companies increasingly rely on CDMOs to scale production, access specialized expertise, and accelerate drug development. The segment is projected to report a CAGR of 6.1% from 2025 to 2032, driven by rising investments in research and development and outsourcing trends.
- To meet this demand, Lonza announced in March 2024 the expansion of its OSD manufacturing capacity in Switzerland, focusing on high-potency APIs and customized solid dose formulations.
- Similarly, Catalent invested US$ 175 Mn in February 2024 to enhance its U.S. facilities for large-scale tablet and capsule production, supporting biotech firms with advanced formulation solutions.
Regulatory pressures and the high costs of in-house manufacturing push Pfizer, Novartis, and AstraZeneca to partner with CDMOs, ensuring compliance while optimizing costs. The growth of biologics and targeted therapies further solidifies CDMOs’ role in delivering scalable, cost-efficient OSD manufacturing.
Regional Insights and Trends
North America Presents Prospects for Innovations in Pharmaceuticals Development
In 2025, North America is projected to account for 40.5% of the global market, driven by expanding pharmaceutical manufacturing facilities and increased drug discovery efforts. The market is forecasted to report a CAGR of 6.3% from 2025 to 2032, supported by novel government initiatives and innovation in OSD formulations.
The region’s pharmaceutical sector continues to invest in advanced manufacturing capabilities, with CMOs playing a crucial role in ensuring scalable production and quality control. For instance,
- In June 2025, the FDA launched a pilot Commissioner’s National Priority Voucher (CNPV) program, granting select companies a 1–2 month review timeline (versus the typical 10–12 months) for CMC submissions especially for manufacturers with domestic production and innovative formulations. This will notably expedite OSD (Oral Solid Dosage) products developed and produced in North America.
- In April 2024, Lonza expanded its oral drug product development and manufacturing services at its facility in Bend, Oregon, enhancing capabilities for complex formulations.
- Catalent announced in March 2024, a US$ 350 Mn investment to expand its manufacturing operations in the U.S. to meet rising demand.
The growing reliance of pharmaceutical companies on CMOs for expertise, cost efficiency, and rapid production is expected to drive North America’s market dominance in the global healthcare sector.
Outsourcing of Pharmaceutical Manufacturing Develops Opportunities in the Asia Pacific
In 2025, Asia Pacific is expected to become a significant region due to improved social insurance schemes and rising outsourcing projects in countries like China, India, and Singapore to strengthen pharmaceutical manufacturing.
- Based on the studies, China, the world’s second-largest pharmaceutical market, has seen rapid expansion, with its pharmaceutical manufacturing sector expected to reach US$ 300 Bn in 2025.
- India, known for its strong generics market, accounts for over 20% of global generic drug exports, with leading CMOs such as Piramal Pharma Solutions and Jubilant HollisterStier expanding production capacity.
In addition, Singapore has emerged as a hub for high-quality pharmaceutical manufacturing, with companies like Lonza and Pfizer investing in advanced OSD manufacturing.
Asia Pacific's cost-effective production, skilled workforce, and regulatory advancements attract international pharmaceutical firms, making it a powerhouse in contract manufacturing and drug development.
Europe Cultivates Innovative Research in Pharmaceutical Drug Manufacturing
Europe is set to boost pharmaceutical drug manufacturing research and development through increased investments and strategic acquisitions, leveraging its position as a hub for innovation.
- In January 2024, Ardena signed an agreement to acquire a Catalent facility in Italy, strengthening its drug product development and manufacturing capabilities. This acquisition enhances Europe’s position in advanced pharmaceutical production, particularly in complex oral solid dosages and biologics.
- Along with this, Recipharm announced in February 2024 a significant investment in expanding its sterile drug development capabilities, reflecting the growing need for advanced drug formulations.
Europe's regulatory agencies, such as the EMA, promote innovation by enticing pharmaceutical businesses to invest in new technologies, presenting the region as a leader in next-generation manufacturing, increasing drug development efficiency, and boosting its worldwide healthcare role.
Oral Solid Dosage Contract Manufacturing Market Competitive Landscape
The global oral solid dosage contract manufacturing market is fiercely competitive, with leading companies dominating the landscape through their integrated business operations. Key companies are set to focus on new product launches with unique characteristics, strengthening distribution networks, as well as geographical expansions.
The increasing demand for pharmaceutical manufacturing is driving companies to expand their manufacturing units, while key providers of oral solid dosage contract manufacturing products are utilizing acquisitions and expansion strategies to strengthen their market presence in the forthcoming period.
Key Industry Developments
- In November 2024, Thermo Fisher Scientific launched Accelerator™ Drug Development, offering expanded clinical research organization and contract development services for biotechs and pharma, aiming to save costs and time for drug developers.
- In October 2024, Catalent, a leading provider of better patient treatments, agreed to sell its oral solids development and small-scale manufacturing facility in Somerset, New Jersey to Ardena, a CDMO with locations in Belgium, Spain, the Netherlands, and Sweden.
- In October 2024, CornePharma invested US$ 10.8 Mn in a new GMP Clinical Trial Development facility at its Plankstadt site, aiming to meet customer demands and improve Oral Solid Dose capabilities.
- In April 2023, NextPharma completed the acquisition of a state-of-the-art chewable tablets manufacturing site in Asker, Norway, from Takeda.
- In January 2023, WuXi STA launched its first continuous manufacturing line for oral solids in Wuxi, China, utilizing real-time analytical technology for compliance with global cGMP regulations.
Companies Covered in Oral Solid Dosage Contract Manufacturing Market
- Recipharm
- Aenova Holding
- Catalent
- AbbVie
- Patheon N.V. (Thermo Fisher Scientific)
- NextPharma
- Lonza Group AG
- Merck KGaA
- Aurobindo Pharma Limited
- Siegfried AG
- Piramal Pharma Solutions
- Corden Pharma
- HERMES PHARMA GmbH
- Medipaams India Private Limited
- Alpex Pharma
- Abaris Healthcare Pvt Ltd
- Ardena Holdings N.V
- Aphena Pharma Solutions
- Actiza Pharmaceutical Private Limited
- Sunwin Healthcare PVT. LTD
- Kosher Pharmaceuticals
- Boehringer Ingelheim BioXcellence
- Others
Frequently Asked Questions
The Global market is estimated to increase from US$ 38.0 Bn in 2025 to US$ 57.9 Bn in 2032.
Rising pharmaceutical outsourcing, cost-efficiency, generic drug demand, and advanced manufacturing technologies drive the global oral solid dosage contract market.
The market is projected to record a CAGR of 6.2% during the forecast period from 2025 to 2032.
Opportunities include high demand for generics, specialized formulations, emerging markets expansion, and partnerships for sustained-release and high-potency drug manufacturing.
Major players include Recipharm, Aenova Holding, Catalent, AbbVie, Patheon N.V. (Thermo Fisher Scientific), NextPharma, Lonza Group AG and Others.







