- Pharmaceuticals
- U.S. Bacterial Conjunctivitis Drugs Market
U.S. Bacterial Conjunctivitis Drugs Market Size, Share, and Growth Forecast, 2025 - 2032
U.S. Bacterial Conjunctivitis Drugs Market By Drug Class (Fluoroquinolones, Aminoglycosides, Macrolides, Other), Route of Administration, Indication, Distribution Channel, and Analysis for 2025 - 2032
U.S. Bacterial Conjunctivitis Drugs Market Trends Analysis
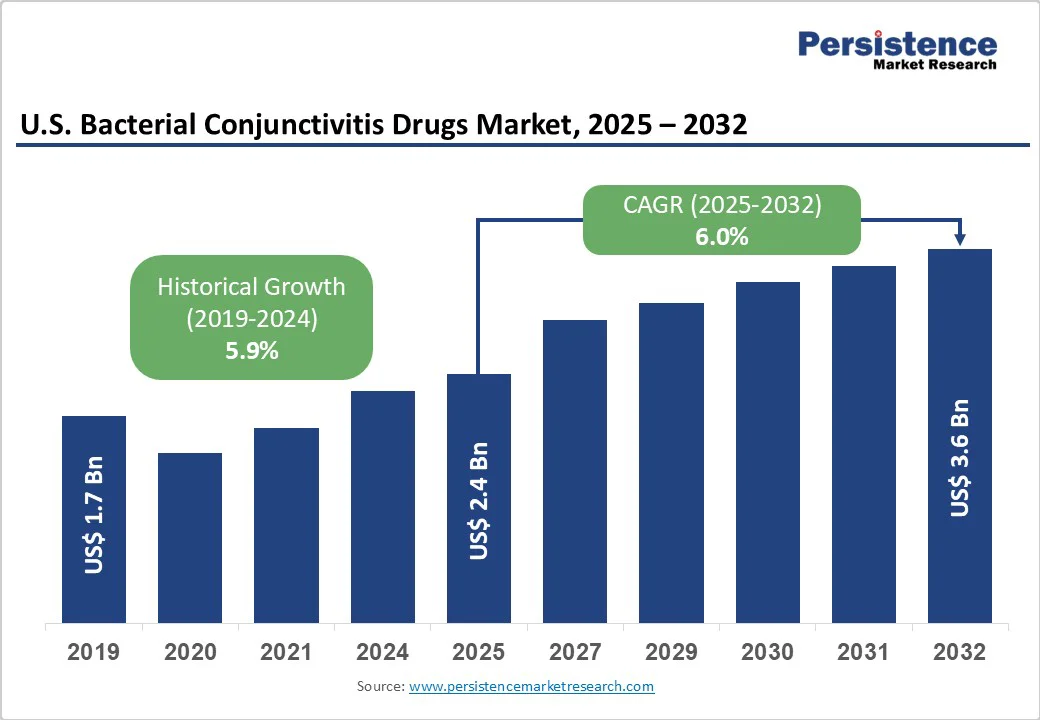
The U.S. bacterial conjunctivitis drugs market size is likely to be valued at US$651.6 Mn in 2025. It is expected to reach US$900.3 Mn by 2032, growing at a CAGR of 4.7% during the forecast period from 2025 to 2032, driven by the high prevalence of conjunctivitis and rising antimicrobial resistance, which are fueling demand for next-generation topical antibiotics and innovative ocular therapies.
Key Industry Highlights:
- Driver: Rising antimicrobial resistance drives demand for next-generation antibiotics with novel mechanisms of action.
- Leading Indication Segment: Acute bacterial conjunctivitis is anticipated to dominate the market with a 74.3% market share in 2025, as it represents the most common form of bacterial eye infection.
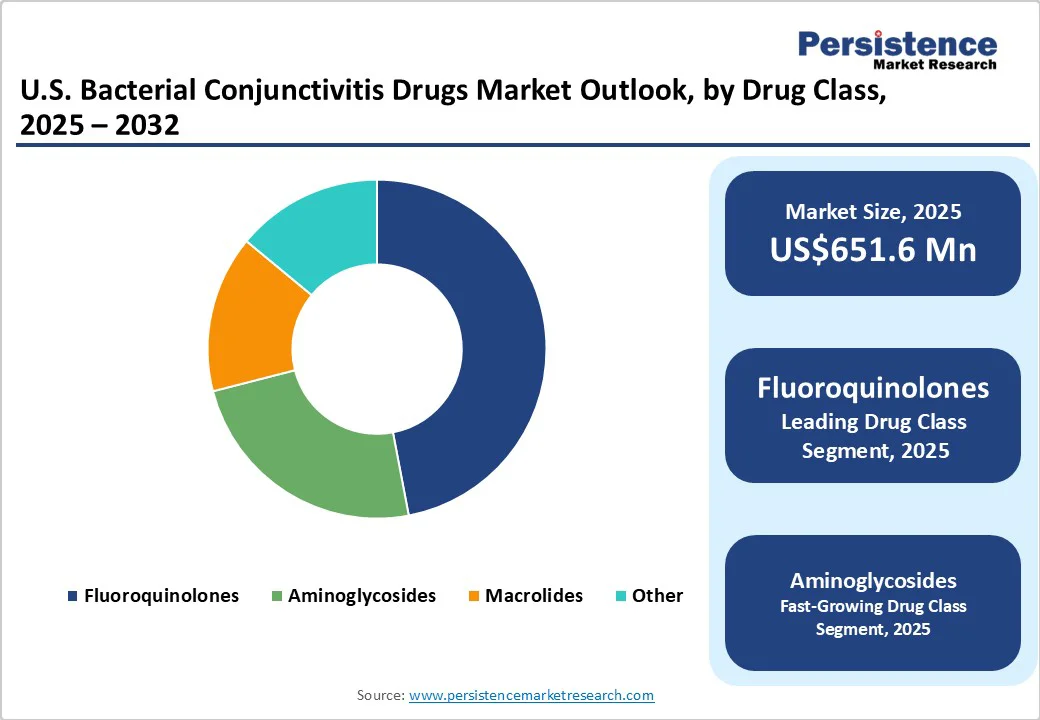
- Dominant Drug Class: Fluoroquinolones are expected to lead with a market share of 52.1% in 2025, driven by their broad-spectrum activity and high physician preference.
- Leading Route of Administration: Topical formulations are projected to dominate the market with a 68.8% market share, owing to their targeted delivery, rapid action, and improved patient compliance.
- Opportunity: Tele-ophthalmology adoption supports remote diagnosis and prescription, improving access and reducing hospital visits.
| Key Insights | Details |
|---|---|
|
U.S. Bacterial Conjunctivitis Drugs Market Size (2025E) |
US$651.6 Mn |
|
Market Value Forecast (2032F) |
US$900.3 Mn |
|
Projected Growth (CAGR 2025 to 2032) |
4.7% |
|
Historical Market Growth (CAGR 2019 to 2024) |
4.0% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Rising Prevalence of Bacterial Eye Infections Among All Age Groups
Bacterial conjunctivitis, commonly referred to as pink eye, is one of the most frequently reported eye infections in the U.S., affecting approximately 135 cases per 10,000 people annually. It represents a significant burden in both primary care and ophthalmology practices, with conjunctivitis accounting for nearly 2–5% of general practice consultations for eye-related issues. Infectious conjunctivitis accounts for approximately 35% of all eye cases, with an incidence of 13–14 cases per 1,000 individuals.
Acute bacterial conjunctivitis remains the most prevalent condition across age groups. In children, it accounts for more than 50% of conjunctivitis cases, largely driven by high transmission in schools and daycare settings. Among adults, exposure in workplaces and frequent contact lens use contribute substantially to case volumes. While Neisseria meningitidis is an uncommon cause, a notable outbreak of 41 meningococcal conjunctivitis cases occurred from February to May 2025 among military trainees at Joint Base San Antonio-Lackland in Texas.
The widespread prevalence of bacterial eye infections results in a consistently high number of outpatient visits and antibiotic prescriptions, particularly for topical treatments. As these infections cause discomfort, disrupt daily activities, and may lead to complications if untreated, timely and effective treatment remains critical. This broad patient base requiring rapid therapy continues to drive strong demand for bacterial conjunctivitis drugs in the U.S. bacterial conjunctivitis drugs market.
Patent Expiries and Rising Antibiotic Resistance Significantly Restrain U.S. Market Growth
Patent expiries of branded antibiotics pose a significant restraint to the U.S. bacterial conjunctivitis drugs market. The loss of exclusivity for widely prescribed agents, particularly topical fluoroquinolones and macrolides, allows generic manufacturers to enter the market with more affordable alternatives. While this improves patient accessibility due to cost-effectiveness and comparable therapeutic outcomes, it exerts downward pressure on branded revenues, reduces profitability, and fragments the market, ultimately lowering overall value.
In parallel, the rising prevalence of antibiotic resistance further challenges market growth. Fluoroquinolones, long considered the gold standard for rapid treatment, are increasingly compromised by resistant strains, notably Staphylococcus aureus and Streptococcus pneumoniae. Resistance has also been observed across multiple drug classes, diminishing the effectiveness of traditional options such as erythromycin, which no longer provides adequate coverage for Haemophilus influenzae.
Though combinations such as polymyxin B/ B/trimethoprim and newer fluoroquinolones still manage most acute cases, high costs and emerging resistance remain limiting factors. These dynamics erode physician confidence, diminish treatment effectiveness, and underscore the pressing need for next-generation therapies with enhanced resistance profiles.
Development of Next-Generation Antibiotics with Improved Resistance Profiles
Growing antimicrobial resistance (AMR) has reduced the effectiveness of commonly prescribed antibiotics, including fluoroquinolones and macrolides, in treating bacterial conjunctivitis. In the U.S., antibiotic-resistant infections cause over 2.8 million illnesses and 35,000 deaths annually. Surveillance programs, such as Ocular Tracking Resistance in the US Today (TRUST) and Antibiotic Resistance Monitoring in Ocular Microorganisms (ARMOR), have consistently reported an increase in resistance among ocular pathogens since 2009.
An ARMOR analysis of 2,214 conjunctival isolates (2009–2020) showed high resistance among Staphylococcus species to azithromycin and oxacillin, while S. pneumoniae demonstrated notable resistance to azithromycin and penicillin. Such findings highlight the urgent need for novel therapies.
Pharmaceutical companies are investing in innovative drug delivery systems, synthetic biology, AI, and genome mining to discover new molecules. Next-generation topical antibiotics that address resistant strains could gain strong physician preference and payer support, extend the lifecycles of antibiotic classes, and mitigate generic erosion. Addressing AMR improves patient outcomes and also presents significant commercial opportunities in the U.S. bacterial conjunctivitis drugs market.
Category-wise Analysis
Drug Class Insights
Fluoroquinolones are expected to dominate the market with a 52.1% share in 2025, driven by their broad-spectrum efficacy and strong physician preference for treating bacterial eye infections. Their broad-spectrum activity against both Gram-positive and Gram-negative organisms, combined with superior efficacy, lower dosing frequency, and strong patient compliance, makes them the preferred choice for treatment. They are commonly prescribed as either first-line or fallback therapy due to their reliability in achieving rapid symptom resolution and reducing the risk of complications or transmission.
Branded formulations, such as moxifloxacin and ciprofloxacin eye drops, further reinforce this dominance, particularly in cases of acute bacterial conjunctivitis, where quick clinical improvement is critical. Clinical evidence indicates that topical fluoroquinolones lead to faster clinical and microbiological remission, typically within the first five days of treatment, compared to placebo, while also reducing recurrence rates.
Sustained physician confidence, combined with consistent demand across both hospital and retail pharmacy channels, ensures that fluoroquinolones remain the leading revenue-generating drug class in the U.S. bacterial conjunctivitis market.
Route of Administration Insights
Topical formulations are projected to lead the market with a 68.8% share, attributed to their targeted drug delivery, fast-acting relief, and higher patient compliance. Their ability to deliver treatment directly to the site of infection ensures rapid symptom relief, faster resolution, and strong patient adherence due to their non-invasive nature. This has led to significantly higher prescription volumes compared with systemic or intravitreal options.
Topical agents also generate the largest share of revenue, as most patients are managed with short courses of antibiotic drops or ointments. Clinical evidence supports their effectiveness: a 2023 Cochrane review reported that topical antibiotics improved clinical cure rates of bacterial conjunctivitis by 26% compared with placebo, while also reducing transmission risk.
Retail pharmacies, the primary distribution channel, carry a wide range of topical brands, further enhancing accessibility. In contrast, oral and intravitreal formulations remain limited to severe or resistant cases. Given their affordability, efficacy, and widespread use, topical antibiotics are expected to maintain market dominance in the U.S. for the foreseeable future.
Indication Insights
Acute bacterial conjunctivitis is projected to hold the largest market share of 74.3% in 2025, as it remains the most prevalent type of bacterial eye infection. It is routinely managed with topical antibiotics, particularly fluoroquinolones, due to their rapid efficacy and ease of use. Acute conjunctivitis of all causes affects an estimated 6 million individuals annually in the U.S., generating a consistently high prescription volume.
Epidemiological patterns vary by age: approximately 71% of conjunctivitis cases in children are bacterial, whereas 78% of cases in adults are viral. As acute bacterial cases require immediate treatment to reduce transmission, alleviate discomfort, and prevent complications, they account for the majority of drug utilization and revenue within the market.
In contrast, chronic bacterial conjunctivitis, defined as symptoms persisting for more than four weeks despite standard therapy, is uncommon and typically requires referral to ophthalmology. Hyperacute cases are even rarer and require specialized management. Consequently, both chronic and hyperacute forms contribute minimally to overall market revenues compared with acute presentations.
Regional Insights
U.S. Bacterial Conjunctivitis Drugs Market Trends
The growing adoption of telehealth is a significant trend in the U.S. bacterial conjunctivitis drugs market. The rising prevalence of eye infections and the accelerated shift toward virtual consultations, particularly following the COVID-19 pandemic, have led to a broader use of tele-ophthalmology platforms for remote evaluation. These platforms allow physicians to conduct high-resolution video assessments, provide provisional diagnoses, and issue electronic prescriptions, enhancing patient convenience, reducing hospital visits, and improving treatment adherence.
Supporting this trend, a single-center retrospective study conducted at the University of Miami’s Bascom Palmer Eye Institute analyzed 6,227 patients and 7,138 telehealth encounters. A review of ICD-10 codes revealed 7,138 primary diagnoses, with the most common being general external adnexal concerns (44.1%), followed by conjunctival disorders (15.5%) and ocular surface symptoms (15.4%). The study validated synchronous tele-ophthalmology as a valuable adjunct for triaging acute eye concerns, particularly within large academic institutions facing increasing patient volumes and extended lead times.
For healthcare providers, tele-ophthalmology optimizes resource allocation and extends patient reach, especially in underserved areas. As telemedicine infrastructure advances and reimbursement frameworks evolve, this trend is expected to reshape prescribing patterns further, stimulate online pharmacy growth, and contribute to the expansion of the U.S. bacterial conjunctivitis drugs market.
Competitive Landscape
The U.S. bacterial conjunctivitis drugs market is moderately competitive, with a mix of branded and generic manufacturers. Leading players are dominating through established products, while emerging competition from generic manufacturers leveraging cost-effectiveness and distribution reach has increased competition on pricing and accessibility.
Key Industry Developments:
- In April 2025, Scientific Reports published a study analyzing 6,603 FAERS records from 2004–2024, identifying 38 drugs associated with conjunctivitis. Ophthalmic agents such as brimonidine and latanoprost showed faster onset than systemic drugs, highlighting the need for vigilant pharmacovigilance.
- In December 2024, NTC announced positive Phase II results for NTC014, an eye drop combining a quinolone antibiotic and an NSAID. The MIRAKLE study showed reduced ocular inflammation and improved outcomes, highlighting its potential to treat bacterial conjunctivitis effectively and enhance patient compliance.
Companies Covered in U.S. Bacterial Conjunctivitis Drugs Market
- Novartis AG
- AbbVie
- AdvaCare Pharma
- Bausch + Lomb
- Pfizer Inc.
- Thea Pharma Inc.
- SAVAL Pharmaceuticals
- Harrow, Inc
- Lupin
- STERI-MED PHARMA
- Innoviva Specialty Therapeutics
Frequently Asked Questions
The U.S. bacterial conjunctivitis drugs market is projected to be valued at US$651.6 Mn in 2025.
Rising infection prevalence, increasing AMR concerns, and growing demand for effective topical therapies drive the U.S. bacterial conjunctivitis drugs market.
The U.S. bacterial conjunctivitis drugs market is poised to witness a CAGR of 4.7% between 2025 and 2032.
Development of next-generation antibiotics, innovative delivery systems, and tele-ophthalmology-linked prescribing models presents major U.S. opportunities.
Major players in the U.S. bacterial conjunctivitis drugs market are Novartis AG, AbbVie, AdvaCare Pharma, Bausch + Lomb, and Pfizer Inc.








