
- Medical Devices
- Robotic Surgery Market
Robotic Surgery Market Size, Share, and Growth Forecast, 2025 - 2032
Robotic Surgery Market By Product Type (Surgical Robotic Systems, Others), End-user (Hospitals, Ambulatory Surgical Centers, Others), Deployment Model (On-premises, Remote), Application (General Surgery, Others), and Regional Analysis for 2025 - 2032
Robotic Surgery Market Share and Trends Analysis
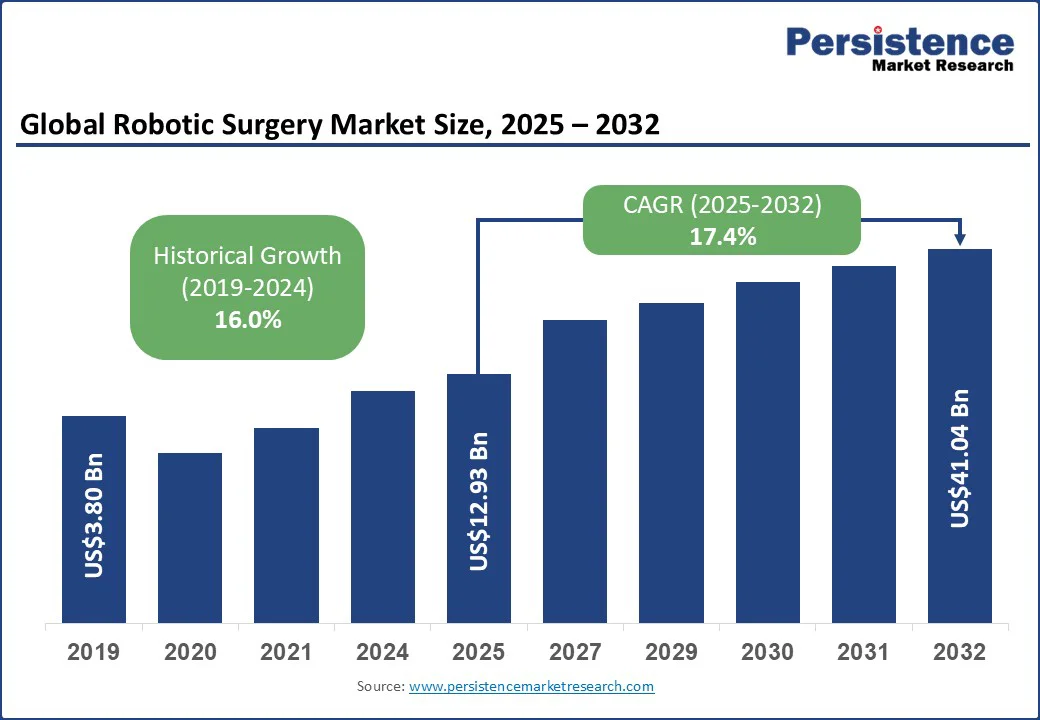
The global robotic surgery market size is likely to be valued at US$12.93 Bn in 2025 and is expected to reach US$41.04 Bn by 2032 growing at a CAGR of 17.4% during the forecast period from 2025 to 2032.
The expansion of telesurgery and remote procedure capabilities, enabling specialists to operate on patients in rural or remote hospitals via ultra-high-speed networks, is emerging as a prime demand driver for robot-assisted surgeries in developing economies.
Robotic surgery, or robot-assisted surgery, uses cutting-edge robotic platforms to increase surgeons’ precision and control during complex procedures, resulting in smaller incisions, reduced complications and errors, and faster patient recovery.
With technologies such as 3D high-definition visualization and motion filtering, these systems are setting new standards for minimally invasive care. A recent study in the U.K. found robot-assisted cystectomy reduced hospital stays by two days and halved readmissions compared to traditional open surgery.
The robotic surgery market expansion is gaining momentum on account of the superior clinical outcomes, streamlined hospital workflows, and cost savings offered by robot-assisted surgical procedures. Current market dynamics are governed by the integration of artificial intelligence (AI) for intraoperative analytics, a growing preference for telesurgery via high-speed networks, and the deployment of compact, mobile robots in outpatient settings.
These advances, combined with the exigent need to address staffing shortages and aging populations, present significant opportunities in specialized areas such as AI-powered surgical systems and remote interventions, making investments in robotic surgery a strategic priority for healthcare providers.
Key Industry Highlights
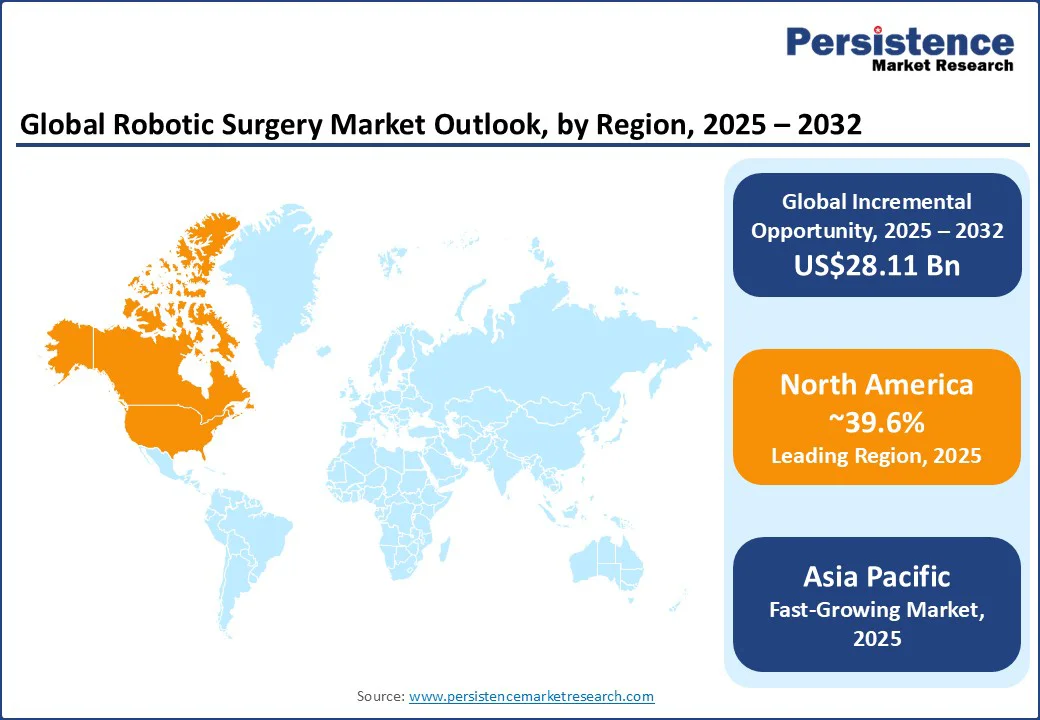
- Dominant Region: North America is likely to dominate with a share of approximately 39.6% in 2025, due to a steady inflow of capital in healthcare R&D and infrastructure, and substantial investments in cutting-edge surgical technologies.
- Fastest-growing Regional Market: Asia Pacific is projected to be the fastest-growing regional market, forecasted to grow at a CAGR of approximately 19% through 2032, supported by increasing healthcare investments and expanding medical tourism.
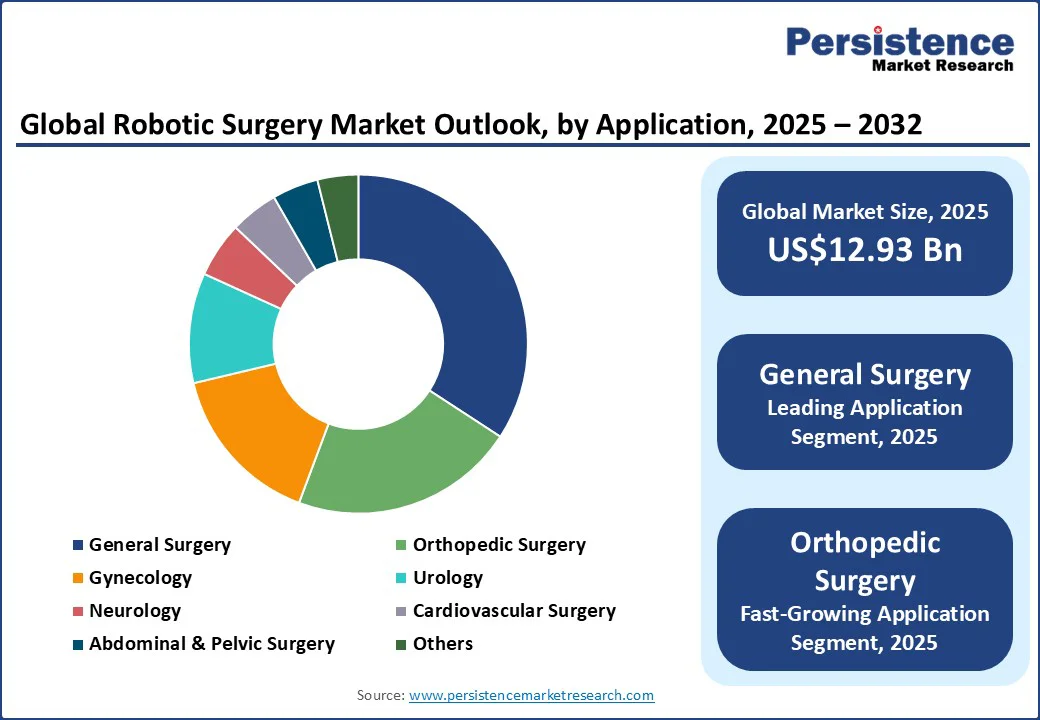
- Leading Segment: General surgery is anticipated to lead the application segment in 2025 with a market share of approximately 34.2%, driven by high surgery volumes and AI-enhanced robotic platforms improving patient outcomes.
- Fastest-growing Segment: Orthopedic surgery is likely to be the fastest-growing application segment, expected to expand at a high CAGR through 2032, fueled by innovations in implant precision and minimally invasive procedures.
- Investment Focus: Integration of artificial intelligence with robotic platforms is revolutionizing surgical procedures by providing real-time decision support and expanding the scope of minimally invasive surgeries.
|
Global Market Attribute |
Key Insights |
|
Robotic Surgery Market Size (2025E) |
US$12.93 Bn |
|
Market Value Forecast (2032F) |
US$41.04 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
17.4% |
|
Historical Market Growth (CAGR 2019 to 2024) |
16.0% |
Market Dynamics
Driver - Strong Demand for Minimally Invasive Surgery to be the Primary Growth Catalyst for the Market
The foremost driver propelling the robotic surgery market growth is the surging demand for minimally invasive surgery (MIS) procedures enabled by the unparalleled precision and control that robotic systems deliver.
A 16-year study conducted by medical researchers from the University of Maryland, U.S., found that while the overall operative volume has increased nationally for surgical residents, the representation of open cases has steadily declined since the advent of MIS in the 1980s.
The high demand for MIS also largely stems from the increasing prevalence of chronic diseases, such as cancer, diabetes, and cardiovascular conditions, which require complex surgeries that traditional methods may prove inadequate to address.
For instance, robotic surgery has been clinically proven to significantly reduce complications, blood loss, and recovery time, as evidenced by a landmark U.K. study reporting a 52% reduction in hospital readmissions after robot-assisted cystectomy, making it an increasingly preferred option among both patients and surgeons.
The adoption of robotic surgery is also reinforced by technological innovations such as AI integration and high-definition real-time visualization, which drastically enhance surgical accuracy and reduce human errors, resulting in higher success rates and operational efficiencies.
Restraint - Exorbitantly High Costs and Complex Adoption Procedure to Limit Market Outlook
The prohibitively high cost structure of robotic surgery systems, spanning acquisition, maintenance, and operational expenses, significantly limits widespread adoption of robotic surgery, especially in lower-resource settings and mid-tier hospitals.
For example, the industry-standard da Vinci surgical system demands an initial investment exceeding US$2 Mn alongside annual maintenance fees of around US$100,000, erecting a steep financial barrier for many healthcare providers.
These costs are further increased by the extensive training surgeons must undergo to master these advanced technologies, which can lengthen operating times and raise labor expenses, and delay the return on investment. Moreover, this cost challenge hinders accessibility in emerging markets with constrained healthcare budgets, impeding their ability to deploy advanced robotic-assisted surgery technologies.
Opportunity - Increasing Integration of AI into Robot-assisted Surgical Procedures
A highly promising opportunity in the robotic surgery market is the rapid integration of advanced AI systems with robotic platforms, revolutionizing surgical precision, decision-making, and scalability.
AI-enhanced tools, such as the Canady Robotic AI Surgical System introduced in 2023, combine real-time data analytics, 3D visualization, and adaptive machine learning to provide surgeons with unprecedented intraoperative guidance, reducing human errors and optimizing patient outcomes.
This fusion of AI and robotics simultaneously stimulates the broader adoption of robotic surgeries by lowering cognitive load on surgeons and enabling more complex minimally invasive surgeries.
In addition to the above, the convergence of AI and robotics is also accelerating the transition from traditional open surgeries to robotic-assisted procedures across diverse specialties such as orthopedics, urology, and general surgery. What needs to be emphasized is that the entry of Industry 4.0 technologies in the fields of medicine and healthcare can potentially unlock scalable, cost-effective robotic surgery adoption worldwide.
Category-wise Analysis
Application Insights
General surgery is expected to be the leading application segment for 2025, commanding approximately 34.2% of the market revenue share. The segment growth is driven by high procedure volumes in common surgeries such as cholecystectomy, hernia repair, and colorectal surgery.
Continuous technological advancements of AI-enabled robotic systems that enhance surgical precision, minimize tissue trauma, and reduce patient recovery times will also drive market growth. Hospitals prioritize general surgeries for robotic integration due to their scalability and the ability to improve throughput without compromising clinical quality. Moreover, AI algorithms provide real-time adaptive decision support during procedures, substantially enhancing safety and efficiency in general surgical procedures.
Orthopedic surgery is anticipated to grow at the highest projected CAGR through 2032. The growing prevalence of musculoskeletal disorders worldwide, combined with the increasing adoption of robotic-assisted joint replacements and spine surgeries, is driving the growth of this segment.
The opportunity in this space is catalyzed by the aging global population and a lifestyle shift toward unhealthy diets and sedentary habits that drive the prevalence of osteoarthritis and degenerative spine conditions. Robotic systems in orthopedics enhance implant accuracy and alignment, reducing revision surgeries, which is a major cost driver in traditional interventions, thereby delivering superior functional outcomes and patient satisfaction.
Noteworthy recent developments include the integration of smart sensors and haptic feedback in orthopedic robots, enabling surgeons to perform complex, MIS with a much superior tactile perception.
Deployment Model Insights
Among deployment models, the on-premises segment is slated to dominate, securing roughly 88% of the market share in 2025. This dominance is due to the preference among hospitals to maintain full control over highly sensitive surgical operations, data privacy, and system maintenance in delicate environments. On-premises robotic surgical systems enable seamless integration with existing hospital IT infrastructure, real-time connectivity with surgical teams, and easier compliance with stringent healthcare regulations.
Moreover, healthcare providers are investing heavily in upgrading on-premises operating rooms with advanced AI-enabled robotic platforms that enhance surgical precision and outcomes. The presence of trained surgical staff and dedicated support systems further justifies the continued reliance on on-premises deployment.
The remote deployment segment is projected to record the highest CAGR through 2032, as advancements in telecommunication infrastructure, low-latency 5G and beyond networks, and AI-driven remote surgical assistance platforms open up new frontiers for telesurgery and decentralized care. Remote robotic surgery eliminates geographical barriers, enabling expert surgeons to perform procedures on patients in underserved or rural areas, thus addressing global disparities in access to high-quality surgical care.
Regional Insights
North America Robotic Surgery Market Trends
In 2025, North America is slated to dominate the robotic surgery market share with approximately 39.6%, propelled by a steady inflow of capital in healthcare R&D and infrastructure, and substantial investments in cutting-edge surgical technologies by both private and public healthcare providers. The U.S. FDA's timely approval of novel robotic systems, combined with favorable reimbursement policies by insurance companies, has accelerated clinical adoption in hospitals and surgical centers.
Leading players such as Intuitive Surgical, Medtronic, and Stryker, headquartered in North America, continue to drive innovation and deployment of robot-assisted surgical systems in the region. The growing integration of AI for enhanced surgical precision, expansion of outpatient robotic surgical procedures, and investments in surgical training facilities are collectively entrenching North America's top position in this market.
Asia Pacific Robotic Surgery Market Trends
Asia Pacific, which is estimated to hold around 23% of the market share in 2025, is distinguished as the fastest-growing market, forecasted to experience a robust CAGR through 2032. This surge is the result of significant investments in medical technologies and healthcare infrastructure by public and private entities in the region, rising disposable incomes, and soaring popularity of medical tourism in China, India, Japan, and South Korea.
Indigenous manufacturers are producing cost-competitive robotic systems tailored to regional needs while collaborating with global firms to enhance technology penetration. Innovative initiatives include China’s pioneering use of satellite communications, which has empowered remote robotic surgeries, and India’s development of multi-disciplinary robotic surgical centers targeting both domestic and international patients. The growing prevalence of chronic diseases and an increasing inclination toward minimally invasive surgeries are also catalyzing the adoption of robotic surgery systems in the region.
Europe Robotic Surgery Market Trends
Accounting for approximately 26% of the robot-assisted surgery market in 2025, Europe offers lucrative opportunities due to the growing adoption of next-generation surgical solutions in university hospitals and specialty care centers after CE-mark approvals.
European healthcare providers are focused on integrating robotics to expand surgical precision in specialties such as gynecology and urology, which can also meet the health management needs of the continent’s aging populations. Noteworthy is Europe's emphasis on cross-border healthcare innovation programs, such as the eHealth Network and the European Health Data Space (EHDS), and early adoption of autonomous surgical features, enabling faster dissemination of robotic technologies across member states.
Competitive Landscape
The global robotic surgery market is profoundly shaped by relentless technological innovation, aggressive mergers and acquisitions, and strategic collaborations among key industry players. Competition among these players is intensifying as companies strive to improve surgical precision, outcomes, and operational efficiency.
Providers are rapidly integrating artificial intelligence, machine learning, and real-time imaging into robotic platforms to offer advanced features such as haptic feedback, autonomous functions, and cloud-based analytics, setting new performance benchmarks.
For example, Intuitive Surgical’s da Vinci Surgical System, with over 1.8 million procedures performed in 2024, exemplifies market leadership supported by continuous product refinement and a broadened global user base. On the other hand, startups and established players alike are leveraging partnerships to accelerate regional penetration, customize solutions for emerging markets, and expand ambulatory surgical robotics applications, signaling a shift toward a more decentralized, AI-driven competitive model.
Key Industry Developments:
- In July 2025, at the Society of Robotic Surgery conference in France, Intuitive showcased telesurgery by linking two surgeons 4,000 miles apart with a dual-console da Vinci 5 system, allowing shared control and force feedback on a tissue model. The technology remains experimental and is not yet approved for clinical use or sale in the U.S. or the EU.
- In July 2025, Medtronic received CE Mark approval in Europe for its first-ever robotic-driven LigaSure™ RAS vessel-sealing instrument, integrating rapid vessel sealing into its Hugo™ robotic surgery system for gynecologic, general, and urologic procedures.
- In July 2025, CARE Hospitals in Hyderabad launched the AI-powered Stryker Mako Robotic System for joint replacements, offering personalized 3D CT planning and real-time robotic guidance with haptic feedback for sub-millimeter accuracy, improved precision, less pain, faster recovery, and better outcomes.
Companies Covered in Robotic Surgery Market
- Intuitive Surgical, Inc.
- Medtronic PLC
- Stryker Corporation
- Johnson & Johnson Services, Inc.
- Zimmer Biomet Holdings, Inc.
- Smith & Nephew plc
- CMR Surgical Ltd.
- TransEnterix Surgical, Inc.
- Renishaw PLC
- Avatera Medical GmbH
Frequently Asked Questions
The global robotic surgery market is projected to reach US$12.93 Bn in 2025.
The surging demand for minimally invasive surgery (MIS) and the increasing global prevalence of chronic diseases that require complex surgeries are driving the market.
The robotic surgery market is expected to witness a CAGR of 17.4% from 2025 to 2032.
The rapid integration of advanced AI systems with robotic platforms and the entry of Industry 4.0 in medical technology and healthcare are key market opportunities.
Key players in the robotic surgery market include Intuitive Surgical, Inc., Medtronic PLC, and Stryker Corporation.







