- Healthcare Services
- Small Molecule CMO/CDMO Market
Small Molecule CMO/CDMO Market Size, Share, and Growth Forecast for 2025 - 2032
Small Molecule CMO/CDMO Market by Product (Standard API, HP API), Service (Process Development, Analytical Method Development, GMP Manufacturing Service, Scale-up and Tech Transfer), Company Size, Scale of Operation, and Regional Analysis
Small Molecule CMO/CDMO Market Size and Trends
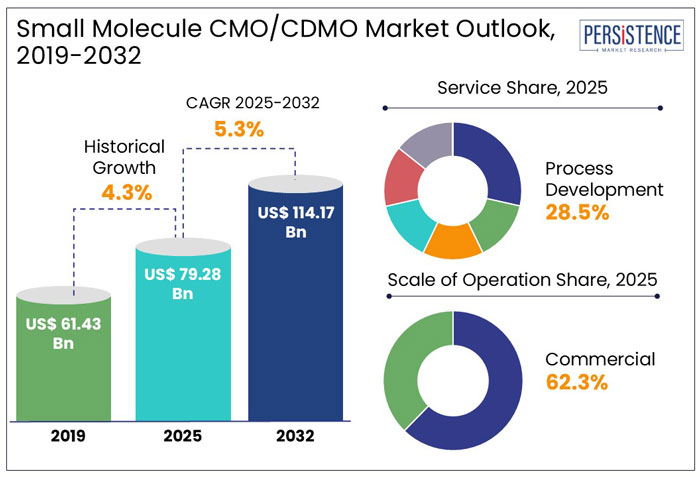
The global small molecule CMO/CDMO market size is anticipated to rise from US$ 79.28 Bn in 2025 to US$ 114.17 Bn by 2032. It is projected to witness a CAGR of 5.3% from 2025 to 2032.
As per Persistence Market Research (PMR), biotechnology and biopharmaceutical companies utilize CDMOs to streamline operations and reduce costs in research and manufacturing services, including fill finishing, packaging, and development, often outsourcing drug manufacturing and research.
- The U.S. Food and Drug Administration (FDA) reports that small molecules hold 59% of new drug approvals in 2022, representing 22 out of 37 new drugs approved. This growth is attributed to the rising prevalence of cancer and high demand for safe therapeutic options.
Key Highlights of the Small Molecule CMO/CDMO Market
- Pharmaceutical companies are outsourcing drug development and manufacturing to achieve cost-efficient production solutions.
- Rising demand for complex small-molecule drugs presents opportunities for CMOs with specialized manufacturing capabilities and expertise.
- Focus on rare diseases and personalized medicine necessitates flexible manufacturing facilities capable of handling smaller batch sizes.
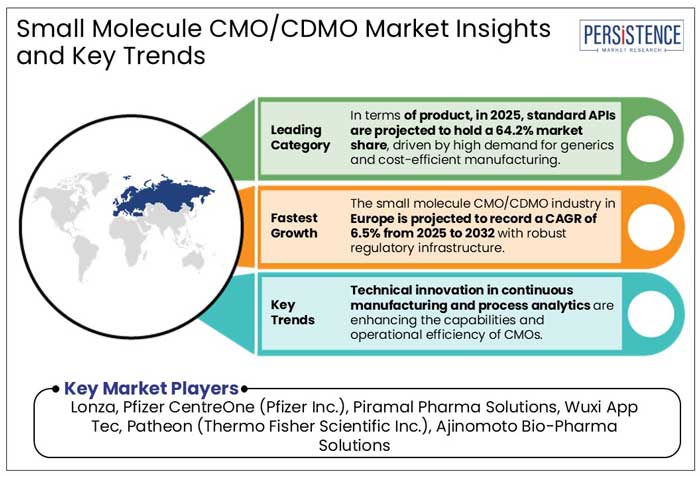
- Technical innovation in continuous manufacturing and process analytics are enhancing the capabilities and operational efficiency of CMOs.
- In 2025, the commercial scale of operation segment is projected to dominate the small molecule CMO/CDMO market, holding around 62.3% share.
|
Global Market Attributes |
Key Insights |
|
Small Molecule CMO/CDMO Market Size (2025E) |
US$ 79.28 Bn |
|
Market Value Forecast (2032F) |
US$ 114.17 Bn |
|
Projected Growth (CAGR 2025 to 2032) |
5.3% |
|
Historical Market Growth (CAGR 2019 to 2024) |
4.3% |
Historical Growth and Course Ahead
Investments in New Facilities and Cutting-edge Technologies Boosted Growth from 2019 to 2024
The global small molecule CMO/CDMO industry witnessed a CAGR of 4.3% in the historical period between 2019 and 2024. To develop new product lines and increase their clientele, small molecule CMO/CDMO businesses are giving acquisitions and growth first priority. Additionally, they are using new technology in the research, manufacturing, and packaging of drugs. They are also focusing on the development of innovative facilities to extend their production capacities. For example,
- In July 2023, Adare Pharma Solutions sold its Adare Biome business unit to dsm-firmenich.
- Asymchema completed the construction of a new building in Dunhua, China, in April 2022, which added nearly 500 cubic meters of batch capacity for potential commercialization projects.
Specialized Expertise in High-potency APIs and Continuous Manufacturing to Fuel Demand through 2032
In the estimated timeframe from 2025 to 2032, the global market for small molecule CMO/CDMOs is likely to showcase a CAGR of 5.3%. Demand for complex small molecule drugs is creating opportunities for contract manufacturing organizations with specialized expertise in high-potency APIs, continuous manufacturing, and innovative formulation technologies.
- In 2023, small molecule drugs accounted for nearly 70% of global pharmaceutical sales, with the CMO/CDMO market anticipated to reach US$ 150 Bn by 2027, according to studies.
Growing demand for oncology, cardiovascular, and CNS drugs is driving investments in specialized CMO/CDMO capabilities, positioning key players to benefit from innovative drug development trends.
Market Dynamics
Growth Drivers
Expanding Cancer Drug Pipeline to Push Growth in Complex Molecule Manufacturing Services
The need for complex molecule manufacturing services is rising owing to the surging medication pipeline in cancer and other therapeutic areas.
- According to studies, the worldwide sales of oncology medicine are expected to reach US$ 470 Bn by 2028, with over 40% of newly developed medications focusing on this area in 2023.
Several companies are striving to broaden their presence across developed countries and regions. They are doing so with the help of product launches, capacity expansions, and investment strategies. For instance,
- In December 2023, Cambrex Corporation completed a US$ 250 Mn expansion in the U.S. and Europe, increasing capacity for complex molecule synthesis.
- In November 2023, Lonza Group announced a US$ 1 Bn investment to extend high-potency API production.
- In October 2023, Samsung Biologics launched a new small molecule division to diversify its portfolio.
Market Restraining Factors
Trust and IP Protection Issues May Impact Long-term Collaboration in Drug Development
Pharmaceutical businesses, CMOs, and CDMOs may find it difficult to collaborate due to Intellectual Property (IP) issues, which can restrict information exchange and impede creative problem-solving and process optimization. This may reduce the advantages of outsourcing and obstruct candid dialogue. Long-term collaborations and open communication can also be hampered by trust concerns.
IP protection agreement drafting may be difficult and time-consuming, which could have an impact on project budgets and schedules. Pharmaceutical businesses would be reluctant to work together if intellectual property issues are not resolved, which might restrict market development in the projected period. For medication research and production to be effective, IP issues must be addressed.
Key Market Opportunities
Shift toward Virtual Pharma Models to Augment Demand for Integrated CDMO Services
As more businesses outsource research and development, clinical trials, and production to CMOs and CDMOs, the growing use of virtual pharma business models is opening substantial prospects for end-to-end drug development and manufacturing services. For example,
- In October 2023, Samsung Biologics won a US$ 1.2 Bn deal to supply multinational biopharma companies with end-to-end CDMO services.
To assist completely virtual biotech companies, WuXi AppTec extended its integrated research and development as well as production facilities in December 2023. Emerging biotech companies are able to launch treatments more quickly because of the move toward asset-light pharmaceutical models, especially in the areas of biologics and complicated small molecules. These companies rely on specialist CDMO partners to ensure smooth medication development and commercialization.
Small Molecule CMO/CDMO Market Insights and Trends
Product Insights
Cost-efficient Manufacturing is Pushing Widespread Adoption of Standard APIs
In terms of product, in 2025, standard APIs are projected to hold a 64.2% market share, driven by high demand for generics and cost-efficient manufacturing. Global sales of APIs are set to reach US$ 265 Bn by 2028, with standard APIs dominating due to economies of scale, according to a few studies.
Various companies are taking several steps to strengthen their position in the field of standard APIs. For example,
- In January 2024, Piramal Pharma Solutions extended its Antibody-Drug Conjugate (ADC) manufacturing facility in Grangemouth, Scotland, with a £45 Mn investment. This expansion increased the site's capacity by around 70% to 80%, adding two new manufacturing suites to the existing three.
Service Insights
Surging Pressure to Reduce Drug Development Costs to Fuel Demand for Outsourced Process Development
In 2025, the process development segment is anticipated to lead the market with a 28.5% share. It is driven by increasing demand for scalable and cost-effective manufacturing of small molecule drugs.
With pressure to reduce drug development costs, pharma companies are outsourcing early-stage and late-stage process development to CMOs/CDMOs. These service providers help in streamlining synthesis pathways, reducing waste, and improving yield efficiency, making production more cost-effective. One such example is WuXi STA-
- WuXi STA, a leading CDMO, extended its process development and manufacturing capabilities for highly potent APIs (HPAPIs) and small molecules. Due to the rising demand for complex oncology drugs and targeted therapies, the company invested in advanced process optimization and continuous manufacturing.
Regional Insights and Trends
Strict Regulatory Framework in Europe Bolsters Competitiveness and Creates Opportunities for CMO/CDMO Providers
In 2025, Europe is projected to dominate the small molecule CMO/CDMO market with a 38.2% share. It is mainly driven by its strong pharmaceutical manufacturing infrastructure, innovative technologies, and strict regulatory standards. The market is set to report a CAGR of 6.5% from 2025 to 2032. A few examples of stringent norms are-
- The European Medicines Agency (EMA) and national regulatory bodies such as the U.K.’s MHRA and Germany’s BfArM enforce Good Manufacturing Practice (GMP) guidelines, which are mandatory for both API and drug product manufacturing.
- The guidelines require strict quality control, batch traceability, and regular facility inspections to maintain compliance. Non-adherence to these standards can lead to manufacturing bans, warning letters, or market recalls.
The aforementioned strict regulatory norms in Europe’s market are bolstering quality improvements and global competitiveness. While compliance with GMP, ICH Q7, and Ph. Eur. standards presents challenges, these regulations are also creating positive opportunities for CMO/CDMO service providers.
Well-established Biotechnology and Pharmaceutical Industry to Strengthen North America's Position
North America, a key contributor to the global market, is set to account for a 26% share in 2025. The region is estimated to record a CAGR of 4.7% from 2025 to 2032. The region is renowned for its well-established biotechnology and pharmaceutical firms, as well as rising investments in research and development, which are set to push demand.
Increasing prevalence of illnesses like diabetes, cardiovascular disease, and cancer is also set to create new opportunities for North America. The U.S., with the largest global healthcare spending, is increasing outsourcing and supporting CDMOs to reduce operational and capital expenses, while promoting new therapies. For instance,
- The U.S. has a growing number of clinical trials, with 147,213 clinical trials conducted between 1999 and 2021, and 127,967 registered clinical studies as of December 2021.
These factors contribute to growth opportunities for domestic CDMOs in North America.
Global Pharma Companies to Invest in Asia Pacific as High-quality Manufacturing Expands
In 2025, Asia Pacific is projected to hold a 24.7% share in the small molecule CMO/CDMO market, with a CAGR of 7.8% from 2025 to 2032. Growth is fueled by lower manufacturing costs and increasing pharmaceutical research and development investments in emerging nations such as India and China.
Stringent regulatory frameworks from agencies like PMDA (Japan) and MFDS (South Korea) are ensuring high-quality production standards, attracting global pharma companies. For instance,
- Fujifilm Diosynth, a global CDMO, extended its small molecule API manufacturing capabilities in Japan, ensuring full compliance with PMDA’s stringent GMP regulations.
Small Molecule CMO/CDMO Market Competitive Landscape
Small molecule Contract Manufacturing Organizations (CMOs) and Contract Development and Manufacturing Organizations (CDMOs) are strategically focusing on acquisitions and expansions. They aim to develop innovative product lines and broaden their customer base.
The foremost companies in the sector are not only enhancing their service offerings but are also keen on integrating cutting-edge technologies into various aspects of drug manufacturing, development, and packaging processes. This proactive approach not only helps them stay ahead in the market but also enables them to meet the evolving demands of their clients and the pharmaceutical industry at large.
Key Industry Developments
- In October 2024, Symbiosis Pharmaceutical Services invested US$ 1.57 Mn in a new manufacturing facility at Castle Business Park in Stirling, Scotland, doubling the local CMO's production capacity.
- In September 2024, SK Pharmteco planned to build a US$ 260 Mn small molecule and peptide production facility in Sejong, South Korea, employing 300+ people and broadening its manufacturing network.
- In June 2024, Veranova, a leading antibody-drug conjugate development company, announced a US$ 30 Mn expansion of its Devens facility, including a new high-potency process development laboratory and two new cGMP suites.
- In January 2024, WuXi AppTec commissioned two new peptide manufacturing plants in China, marking the largest of its five API production sites, aiming to meet global demand for peptide therapeutics.
- In January 2024, Eurofins CDMO Alphora Inc. completed its pilot-scale biologics development facility in Mississauga, Canada, focusing on monoclonal antibodies and therapeutic proteins, offering upstream and downstream development services.
Companies Covered in Small Molecule CMO/CDMO Market
- Lonza
- Pfizer CentreOne (Pfizer Inc.)
- Piramal Pharma Solutions
- Wuxi App Tec
- Patheon (Thermo Fisher Scientific Inc.)
- Ajinomoto Bio-Pharma Solutions
- Siegfried Holding AG
- Eurofins Scientific
- Aurigene Pharmaceutical Services Ltd.
- Asymchema
- PCI Pharma Services
- Almac Group
- Labcorp Drug Development
- Sumitomo Chemical Co., Ltd.
- ICROM
- AGC Inc.
- FUJIFILM Corporation
- CMIC HOLDINGS Co., LTD.
- Evonik
- Terumo Pharmaceutical Solutions (TPS)
Frequently Asked Questions
The market is set to reach US$ 79.28 Bn in 2025.
CMO/CDMOs offer a range of services such as drug development, quality control, clinical trial support, and technology transfer.
Lonza, Pfizer CentreOne (Pfizer Inc.), Piramal Pharma Solutions, Wuxi App Tec, and Patheon (Thermo Fisher Scientific Inc.) are a few leading players.
The industry is estimated to rise at a CAGR of 5.3% through 2032.
Europe is projected to hold the largest share of the industry in 2025.








