- Medical Devices
- Infectious Disease In-vitro Diagnostics Market
Infectious Disease In-vitro Diagnostics Market Size, Share, and Growth Forecast from 2025 - 2032
Infectious Disease In-vitro Diagnostics Market by Product Type (Instruments, Reagents, Software and Services), Technology (Immunoassay, Molecular Diagnostics, Microbiology, Others), Application (MRSA, Clostridium Difficile, Respiratory Virus, TB and Drug-resistant TB, Gonorrhea, HPV, HIV, Hepatitis C, Hepatitis B, COVID-19, Others), End User (Point-of-care, Central Laboratories, Others), and Regional Analysis
Infectious Disease In-vitro Diagnostics Market Size and Share Analysis
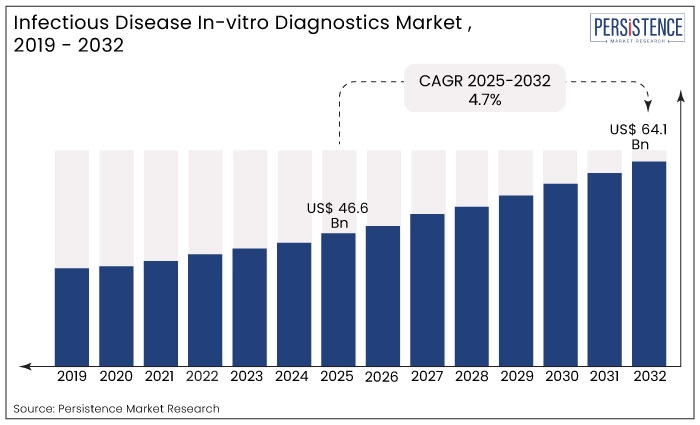
The global infectious disease In-vitro diagnostics market size is anticipated to reach a value of US$ 46.6 Bn in 2025 and is set to witness a CAGR of 4.7% from 2025 to 2032. The market will likely attain a value of US$ 64.1 Bn in 2032. Rising frequency of infections caused by fungi, bacteria, viruses, and parasites increases demand for diagnostic tests that can identify pathogens swiftly and accurately.
Over 60% of clinical decisions are influenced by in-vitro diagnostics (IVD). However, it barely represents 2% of all healthcare costs, thereby presenting a huge opportunity for affordable in-vitro diagnostics.
Developments in diagnostic technologies play a key role in driving expansion. Integration of AI and ML to enhance diagnostic accuracy and speed is also becoming a leading growth factor. Government support is crucial in the growth of the market as increased funding from the government enables research institutions to create quick analysis systems helpful for the diagnosis of a variety of diseases.
Key Highlights of the Industry
- Rising global awareness regarding the importance of early diagnosis for preventing the spread of infectious diseases is bolstering demand for IVD products.
- Government authorities along with healthcare organizations are focusing on routine screening programs for diseases like tuberculosis, STIs, HIV, and hepatitis. .
- Molecular diagnostics are witnessing a high demand as these enable the creation of personalized medicine, thereby surging growth in the in-vitro diagnostic market.
- Need for accurate diagnostic testing to identify pathogen type and profile resistance is increasing the demand for molecular and microbiology diagnostics to guide antibiotic therapy and curb AMR.
- In terms of product type, the reagents category is predicted to witness a CAGR of 5.1% through 2032 owing to the rising adoption of POC testing for infectious diseases.
- By technology, the immunoassays segment is estimated to account for a share of 36.3% in 2025 due to their ability to swiftly diagnose infectious diseases.
- Infectious disease in-vitro diagnostics market in the U.K. is projected to showcase a CAGR of 4.9% through 2032 due to high investments in research and development activities in the country.
- India infectious disease in-vitro diagnostics market is set to register a CAGR of 4.7% through 2032 as rising infectious diseases are posing a high demand for cutting-edge diagnostic methods.
|
Market Attributes |
Key Insights |
|
Market Size (2025E) |
US$ 46.6 Bn |
|
Projected Market Value (2032F) |
US$ 64.1 Bn |
|
Global Market Growth Rate (CAGR 2025 to 2032) |
4.7% |
|
Historical Market Growth Rate (CAGR 2019 to 2024) |
3.8% |
Presence of World-famous Research Institutions in the U.K. Fosters Advancement
The U.K. infectious disease in-vitro diagnostics market is estimated to witness substantial demand during the forecast period at a CAGR of 4.9%. This is owing to the country’s well-established healthcare system. The government of the country is investing in healthcare technologies and infrastructures to provide a conducive environment for innovation and implementation of in-vitro diseases.
The country is also popular for its high level of investment in research and development activities, especially in the field of biomedical and life sciences sectors. World renowned institutions like the University of Oxford, Imperial College London, and other world-class research hubs contribute to the modern advancements in diagnostic technologies.
The robust regulatory framework for medical devices and diagnostics in the U.K. further bolsters expansion. These regulations offer manufacturers a stable and predictable environment for the launch of new IVD products.
Robust Government Support in India is Bolstering Expansion
India is poised for significant growth during the assessment period at a CAGR of 4.7%. The ongoing and increasing incidence of infectious diseases is assisting in creating a robust demand for diagnostic tools to enable early detection, monitoring, and effective management.
The rise of infectious diseases in the country poses a requirement for unique, swift, and accessible diagnostic methods. It is further driving demand for point-of-care solutions, PCR testing, IVD reagents, and immunoassays.
The Government of India has been making significant investments in healthcare initiatives like Ayushman Bharat, Pradhan Mantri Jan Arogya Yojna (PMJAY), and National Health Mission (NHM). These schemes aim to improve healthcare access, affordability, and quality.
Disease specific programs like National Tuberculosis Elimination Program (NTEP) and National AIDS Control Program (NACP) focus on reducing the burden of these particular diseases. India is rapidly adopting unique diagnostic technologies like molecular diagnostics, next-generation sequencing, and immunoassays for the testing of infectious diseases.
Ability to Conduct High-precision Tests Propels Demand for Reagents
|
Category |
Market Share in 2025 |
|
Product Type - Reagents |
64.7% |
Reagents are estimated to experience a rising demand as they are essential components of diagnostic tests. These are needed in large quantities to conduct testing for a variety of infectious diseases. The ongoing disease outbreaks increase the need for large-scale diagnostics, thereby surging the demand for diagnostic reagents.
Rise of POC testing for infectious diseases also plays a key role in creating robust demand for reagents that are tailored for these applications. Surging adoption of genetic and molecular testing for infectious diseases is raising demand for reagents to conduct high-precision tests.
The market is also witnessing a growing trend toward the development of customized reagents that are tailored to cater to the specific requirements of certain diagnostic tests. Manufacturers in the industry are hence focusing on creating reagents that are specific, sensitive, and resistant to contamination.
Low Cost of Immunoassays Makes them Easily Accessible
|
Category |
Market Share in 2025 |
|
Technology - Immunoassays |
36.3% |
The ability of immunoassays to quickly diagnose infectious diseases is particularly valuable in clinical settings and during disease outbreaks. Rapid testing can assist in initiating timely treatment, decreasing disease transmission, and enabling efficient healthcare responses. Lower cost of immunoassays makes them accessible to low-resource settings, including rural areas and emerging markets.
Affordability and ease of distribution makes this technology a critical tool in global health programs, particularly in countries with limited healthcare infrastructure. Immunoassay-based diagnostics can be utilized for a wide range of infectious diseases. This ability makes them a versatile tool in clinical settings as well as public health programs.
Market Introduction and Trend Analysis
As long as endemic and emerging infectious diseases remain a significant global health burden, the spread of diseases like tuberculosis, malaria, COVID-19, influenza, and HIV/AIDS will continue to bolster demand for diagnostic solutions.
The rise of Antimicrobial Resistance (AMR) is further intensifying the requirement for accurate diagnostics to identify resistant pathogens and guide appropriate treatment. It is further estimated to drive continuous investments in diagnostic technologies to swiftly identify and manage these diseases. Companies have a particular focus on rapid diagnostics, precision medicine, and early detections.
The industry is likely to witness accelerated adoption of molecular diagnostics, AI-driven diagnostics, Point-Of-Care Testing (POC), and multiplexed diagnostic tests. These technologies are estimated to allow for swift, accurate, and highly sensitive pathogen detection, which is key for managing common and emerging infectious diseases.
Growth in the market is also likely to be driven by the emergence and adoption of these technologies as they will assist in expediting test result time. These also enable personalized treatment plans along with decreasing healthcare costs. The trend toward automated diagnostics will likely assist in streamlining workflows and enhancing operational efficiency.
Historical Growth and Course Ahead
The infectious disease in-vitro diagnostics market remained steady during the period between 2019 and 2024 witnessing a CAGR of 3.8%.
The diagnosis of COVID-19 was highly dependent on in-vitro testing. This was clearly demonstrated by the vast array of test kits that have been launched globally for the detection of COVID-19. All of these tests received permission under an emergency use authorization, which exempts them from usual FDA review requirements. Most of the market participants were focused on the creation of quick detection test kits for COVID-19. The market is projected to secure a CAGR of 4.7% through 2032.
Market Growth Drivers
Rising Shift toward Home Testing and Telemedicine to Skyrocket Demand
The rise in home-based testing for infectious diseases, including HIV, COVID-19, and STIs are driving demand in the IVD industry. Consumers across the globe are seeking at-home diagnostic solutions for greater convenience and timely results.
The emergence of telemedicine also supports the requirement for remote diagnostic tools that can be used at home or in non-clinical settings. Infectious disease in-vitro diagnostics market growth is closely linked to the rising demand for self-testing kits, telehealth platforms, and home diagnostics, especially with a focus on disease prevention and management.
Developments in Molecular Diagnostic Techniques to Propel Demand
The market is witnessing rapid developments in diagnostic technologies, thereby transforming the landscape of infectious disease testing. Innovations like Polymerase Chain Reaction (PCR) and other molecular diagnostic techniques assist in providing precise, accurate, and rapid detection of pathogens at the genetic level.
Next-generation sequencing enables comprehensive pathogen identification, including rare or emerging infections. Point-of-care (POC) diagnostics are witnessing rising demand in the industry as they provide fast, reliable, and cost-effective testing in remote as well as low-resource settings.
Growth of multiplexed testing enables simultaneous detection of various pathogens from a single sample. These innovations assist in enhancing the speed, accuracy, and ease of testing, thereby resulting in better clinical decision-making, effective disease management, and decreased time for treatment.
Market Restraining Factors
Uncertainty around Reimbursement Policies to Reduce Uptake
Several diagnostic tests, especially those used for infectious diseases, face challenges in securing reimbursement from public health systems or insurance companies. This uncertainty around obtaining reimbursement along with pricing pressure can discourage innovation and decrease the uptake of novel diagnostic tools.
Reimbursement policies vary between countries and regions. Tests that are not covered are hence likely to face limited adoption. Without guaranteed reimbursement, healthcare providers are set to be hesitant to invest in new diagnostic technologies, especially for infectious diseases.
Market Growth Opportunities
Rising Antimicrobial Resistance Creates a Need for Diagnostic Solutions
Global rise in antimicrobial resistance is posing a rising demand for diagnostic solutions that assist in identifying resistant pathogens and guide appropriate treatments. Concerns over drug-resistant infections and government regulations encouraging appropriate antimicrobial usage are the key drivers influencing growth in the market.
Companies focusing on developing tests for AMR detection, including rapid test identifying resistance profiles, are expected to see robust demand in developed as well as emerging markets.
Increasing Demand for Personalized Treatments to Spur Need for NGS
Next Generation Sequencing (NGS) has witnessed significant developments, thereby offering comprehensive and efficient ways to sequence genomes. This further enables the detection of a wide range of infectious agents, including bacteria, viruses, and fungi.
There is an increasing demand for personalized treatment strategies that push the use of next-generation sequencing. Low sequencing costs coupled with the ability to detect rare or emerging pathogens further bolster the utilization of NGS. This technology is likely to play a key role in diagnostics and research, thereby making it an attractive area for investment and development.
Competitive Landscape for the Infectious Disease In-vitro Diagnostics Market
Established players in the infectious disease in-vitro diagnostics industry are deploying strategies like acquiring small portfolios of emerging companies and collaborating with big laboratories to launch new diagnostic products. Emerging companies are planning on launching accurate and modern diagnostic solutions to assist healthcare professionals to enhance patient outcomes and improve the quality of life by providing better diagnostic results.
Companies in the infectious disease in-vitro diagnostics market are investing in research and development activities to discover new insights. Mid-sized and small-scale businesses are expanding their market presence by releasing new items at low prices owing to technical improvements and product innovations.
Recent Industry Developments
- In March 2025, QIAGEN announced that it received U.S. regulatory clearance for its second QIAstat-Dx mini gastrointestinal panel, marking a significant expansion of its syndromic testing portfolio in the United States. This clearance further strengthens QIAGEN’s presence in the infectious disease diagnostics market by providing healthcare professionals with a compact, rapid, and accurate molecular testing solution for gastrointestinal infections.
- In March 2025, Bio-Rad Laboratories announced that it received the IVDR quality certificate for 40 Exact Diagnostics-branded infectious disease molecular quality control (QC) products. This milestone marked a significant step in ensuring regulatory compliance under the European Union's In Vitro Diagnostic Regulation (IVDR).
- In March 2024, SEKISUI Diagnostics, based in Massachusetts, obtained the Emergency Use Authorization (EUA) clearance for the OSOM Flu SARS-CoV-2 Combo Test. It is approved for use in professional as well as home testing settings.
- In October 2023, Tangen Biosciences and SD Biosensor received Emergency Use Authorizations (EUAs) from the U.S. FDA for point-of-care SARS-CoV-2 tests.
Infectious Disease In-vitro Diagnostics Industry Segmentation
By Product Type
- Instruments
- Reagents
- Software and Services
By Technology
- Immunoassay
- Molecular Diagnostics
- Microbiology
- Others
By Application
- MRSA
- Clostridium Difficile
- Respiratory Virus
- TB and Drug-resistant TB
- Gonorrhea
- HPV
- HIV
- Hepatitis C
- Hepatitis B
- COVID-19
- Others
By End User
- Point-of-care
- Central Laboratories
- Others
By Region
- North America
- Europe
- East Asia
- South Asia and Oceania
- Latin America
- Middle East and Africa
Companies Covered in Infectious Disease In-vitro Diagnostics Market
- QIAGEN
- Becton, Dickinson and Company
- bioMérieux SA
- F. Hoffmann-La Roche, Ltd.
- Hologic, Inc. (Gen Probe)
- Abbott
- Quidel Corporation
- Siemens Healthineers AG
- Bio-Rad Laboratories, Inc.
- Danaher
- OraSure Technologies, Inc.
Frequently Asked Questions
The Global market is estimated to increase from US$ 46.6 Bn in 2025 to US$ 64.1 Bn in 2032.
Rising infectious disease prevalence, rapid diagnostic innovations, pandemic preparedness, and increased global healthcare spending drive the infectious disease IVD market.
The market is projected to record a CAGR of 4.7% during the forecast period from 2025 to 2032.
Opportunities include point-of-care testing growth, AI integration, emerging markets expansion, home diagnostics, and demand for rapid, accurate tests.
Major players include QIAGEN, Becton, Dickinson and Company, bioMérieux SA, F. Hoffmann-La Roche, Ltd., Hologic, Inc. (Gen Probe), Abbott and Others.








