- Biotechnology
- Gastrointestinal Infection Testing Market
Gastrointestinal Infection Testing Market Size, Share, and Growth Forecast from 2025 - 2032
Gastrointestinal Infection Testing Market by Product Type (Instruments, Reagents and Consumables), Test Type (Molecular Tests, Immunoassays, Culture Tests), Infection Type, Sample Type, End User, and Regional Analysis from 2025 to 2032
Gastrointestinal Infection Testing Market Size and Share Analysis
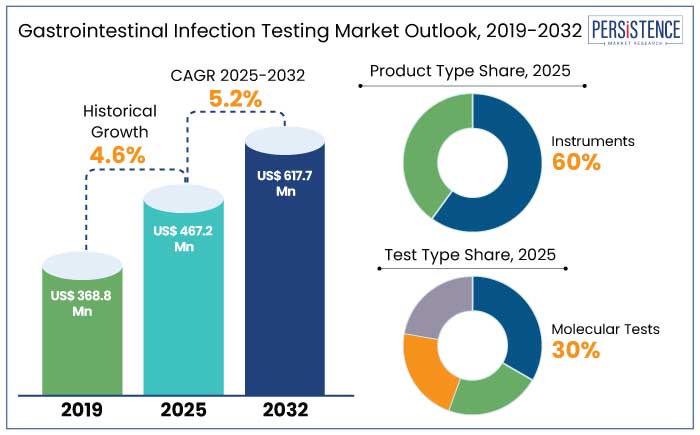
The global gastrointestinal infection testing market is predicted to reach a size of US$ 467.2 Mn by 2025. It is anticipated to witness a CAGR of 5.2% during the forecast period to attain a value of US$ 617.7 Mn by 2032.
Foodborne pathogens like Salmonella, Escherichia coli (E. coli), and Campylobacter are prominent contributors to gastrointestinal infections (GI).
- According to the World Health Organization (WHO), foodborne diseases cause an estimated 600 million illnesses and 420,000 deaths annually worldwide.
- The Centers for Disease Control and Prevention (CDC) reports that more than 48 million people in the U.S. are affected by foodborne illnesses each year.
Rising demand for rapid and accurate diagnostic tests is fostering the development of molecular diagnostic techniques like PCR-based and next-generation sequencing (NGS) tests. Point-of-care diagnostics that enable faster results while decreasing waiting times are gaining traction.
Key Highlights of the Industry
- Increasing focus on early detection of infections to prevent the spread of disease outbreaks.
- Public health campaigns and educational initiatives are surging awareness about gastrointestinal infections and the importance of timely diagnosis.
- Rise of digital health technologies is changing the landscape of gastrointestinal infection diagnostics.
- Reliance on telemedicine, remote consultations, and testing for gastrointestinal infections are becoming more common.
- Non-invasive testing options like breath tests and fecal tests are gaining traction owing to their comfort and ease of use.
- Personalized medicine plays a crucial role in gastrointestinal infection testing by offering customized treatments based on the results of genetic testing.
- By product type, the instruments segment is predicted to hold a share of 60% in 2025 owing to the rising need for accurate diagnostics.
- Based on test type, the molecular tests segment is set to hold a share of 30% in 2025 backed by rising demand for rapid testing.
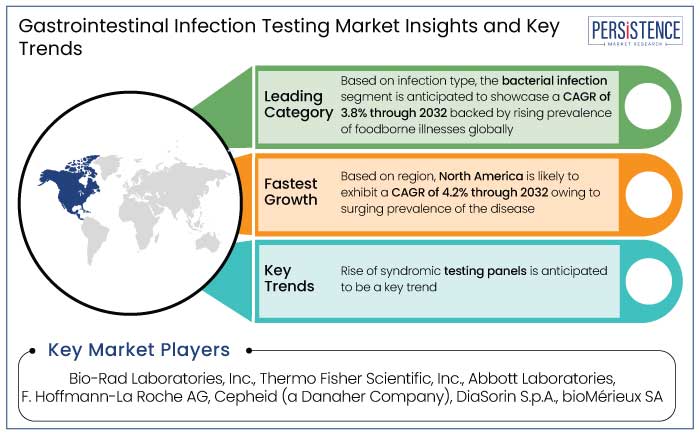
- Based on infection type, the bacterial infection segment is anticipated to showcase a CAGR of 3.8% through 2032.
- North America gastrointestinal infection testing market is projected to hold a share of 35% in 2025 because of the rising prevalence of the disease.
|
Market Attributes |
Key Insights |
|
Gastrointestinal Infection Testing Market Size (2025E) |
US$ 467.2 Mn |
|
Projected Market Value (2032F) |
US$ 617.7 Mn |
|
Global Market Growth Rate (CAGR 2025 to 2032) |
5.2% |
|
Historical Market Growth Rate (CAGR 2019 to 2023) |
4.6% |
Surging Prevalence of GI in North America to Augment Demand
North America gastrointestinal infection testing market is estimated to hold a share of 35% in 2025. The region boasts the most unique healthcare system in the world with widespread access to healthcare services and modern diagnostic technologies. This infrastructure supports the rapid adoption of the latest gastrointestinal infection testing technologies.
- According to the World Health Organization (WHO), healthcare spending in the U.S. accounted for 17.7% of its GDP in 2021, making it one of the largest healthcare markets globally.
Prevalence of gastrointestinal infections in North America remains high due to a combination of factors like foodborne illnesses, contamination, and outbreaks. Pathogens like Salmonella, E. coli, and Norovirus are common causes, resulting in an increasing demand for diagnostic testing to identify the causative organisms.
- According to the Centers for Disease Control and Prevention (CDC), 48 million people in the U.S. suffer from foodborne illnesses every year, with gastrointestinal infections being a key component.
Hospitals to Recommend Intermittent Catheters as a Safer Alternative
Instrument is anticipated to hold a share of 60% in 2025. Need for accurate and rapid diagnosis of gastrointestinal infections is one of the primary reasons behind the increasing demand for instrument-based testing. Instruments like PCR machines, immunoassay analyzers, and culture-based diagnostic tools enable high precision and speed in identifying pathogens.
Development of next-generation diagnostic instruments, like automated PCR machines and multiplex assays, has significantly improved the efficiency and accuracy of gastrointestinal infection testing. High demand for point-of-care (POC) testing is pushing the adoption of instruments that can provide quick results outside traditional laboratory settings. For example,
- According to the National Institutes of Health (NIH), funding for infectious disease research in the U.S. increased by US$ 5 Bn in recent years. Much of this goes toward innovations in diagnostic instruments.
Molecular Tests to Offer High Sensitivity and Specificity in Pathogen Detection
Molecular tests are projected to account for a share of 30% in 2025. Molecular tests, like PCR-based tests, offer high sensitivity and specificity in detecting gastrointestinal pathogens, even at very low concentrations.
Demand for rapid testing has grown owing to the need for quick identification of gastrointestinal infections, particularly in emergency settings or foodborne illness outbreaks. For instance,
- Molecular diagnostic tests can detect pathogens in as little as 2 to 4 hours, compared to traditional methods that may take 2 to 5 days.
The American Society for Microbiology states that rapid diagnostic tests, especially molecular tests, are vital in quickly identifying infectious agents. This assists in decreasing the length of hospital stay and preventing the overuse of antibiotics.
A BioTechniques article mentioned that multiplex PCR testing is becoming increasingly popular in clinical and commercial laboratories. Its ability to provide comprehensive pathogen identification in gastrointestinal cases spurs growth.
Gastrointestinal Infection Testing Market Introduction and Trend Analysis
Potential growth in the global gastrointestinal infection testing industry is predicted to be driven by the rise in availability of over-the-counter (OTC) test kits that can be used by consumers at home to identify pathogens. This improves convenience while decreasing healthcare facility burdens. Regulatory bodies like FDA and European Medicines Agency (EMA) are streamlining approval processes for gastrointestinal infection testing devices.
Historical Growth and Course Ahead
The gastrointestinal infection testing market growth was steady at a CAGR of 4.6% during the historical period from 2019 to 2023. Growth during the period was attributed to increased awareness regarding gastrointestinal diseases and rising demand for point-of-care testing.
Developments in diagnostic technologies like PCR-based molecular tests were key in driving demand. Diarrheal infections were one of the leading causes of morbidity globally. For example,
- The CDC estimated that 48 million cases of foodborne illness occur annually in the U.S. alone.
The forecast period is predicted to witness continuous developments in molecular diagnostic and automation. Increasing awareness regarding gastrointestinal diseases and their complications are likely to drive demand for innovative diagnostic methods and treatment options.
Market Growth Drivers
Increasing Prevalence of Gastrointestinal Infections
The World Health Organization (WHO) estimates that 600 million people fall ill each year due to contaminated food, resulting in 420,000 deaths annually. Rapid urbanization, especially in developing countries, has led to overcrowded living conditions and strained sanitation systems, increasing the risk of GI infections.
Changes in temperature and rainfall patterns have facilitated the spread of GI pathogens, especially waterborne diseases like cholera and typhoid. For instance, Vibrio infections, including cholera, have become more frequent in coastal areas due to rising sea temperatures. For example,
- According to the Centers for Disease Control and Prevention (CDC), up to 70% of international travelers experience traveler’s diarrhea caused by pathogens such as E. coli and Campylobacter.
Emergence of antibiotic-resistant bacteria has made some GI infections harder to treat, increasing the demand for accurate diagnostic tools to guide treatment. Pathogens like Clostridioides difficile have become a significant concern in hospital settings.
Surging Adoption of Molecular Diagnostic Technologies
Molecular diagnostic techniques, such as polymerase chain reaction (PCR) and nucleic acid amplification tests (NAATs), can detect pathogens at very low concentrations. Modern molecular diagnostics can simultaneously test for multiple pathogens in a single assay. For instance,
- Multiplex PCR panels can identify 20+ pathogens, including Salmonella, Norovirus, and Clostridioides difficile, in less than 2 hours, reducing time-to-diagnosis.
Molecular tests typically provide results within 1 to 4 hours, compared to 24 to 72 hours for culture-based methods. This speed is particularly critical in acute GI infections, where timely diagnosis can prevent complications such as dehydration or sepsis. Automated molecular diagnostic platforms like Cepheid's GeneXpert and Roche's cobas Liat System have made these technologies accessible even in smaller healthcare settings. For instance,
- In 2023, approximately 40% of hospital laboratories in developed countries adopted molecular platforms for GI infection testing. This adoption rate is estimated to exceed by 65% by 2030.
Market Restraining Factors
Inadequate Diagnostic Infrastructure in Developing Regions
Healthcare facilities in developing regions lack the necessary infrastructure for conducting advanced diagnostic tests, like polymerase chain reaction (PCR) or multiplex pathogen detection panels. For example,
- According to the World Health Organization (WHO), approximately 40% of healthcare facilities in low-income countries lack basic diagnostic tools for infectious diseases.
Unique diagnostic technologies require skilled technicians to operate equipment and interpret results. A 2021 study by the International Journal of Infectious Diseases reported that 70% of laboratories in rural Asia and Africa operate with underqualified staff, leading to lower test accuracy.
Market Growth Opportunities
Innovations in Diagnostic Technologies
Technologies like polymerase chain reaction (PCR), nucleic acid amplification tests (NAATs), and isothermal amplification have revolutionized pathogen detection. For example,
- Molecular diagnostics offer 95% to 99% accuracy, significantly higher than the 60% to 70% range seen in traditional culture methods.
Portable diagnostic devices like Cepheid GeneXpert and Abbott ID NOW provide results within 30 minutes to 1 hour, enabling immediate clinical decisions. NGS facilitates detailed pathogen profiling, including strain typing, resistance gene identification, and microbiome analysis.
Microfluidic platforms integrate multiple diagnostic processes, including sample preparation and pathogen detection in a single chip. These systems decrease testing time, cost, and required sample volume, making them ideal for low-resource settings.
Rise of Syndromic Testing Panels
Syndromic panels detect numerous pathogens in a single test, addressing the challenge of overlapping symptoms caused by various GI infections. Early and accurate detection of pathogens facilitates prompt treatment, reducing patient morbidity and the risk of complications. For instance,
- The BioFire FilmArray GI Panel can detect 22 pathogens, including Clostridium difficile, Salmonella, Norovirus, and E. coli within 1 hour, fostering their expansion.
- According to a report by the Journal of Clinical Pathology, syndromic testing panels have been shown to decrease hospital stay duration by 25% to 30%.
Syndromic panels streamline diagnostic workflows by combining multiple tests into one, minimizing the need for repeated testing. As per data from the American Society of Microbiology, laboratories report a 40% decrease in workload when switching to multiplex syndromic panels.
Competitive Landscape for the Gastrointestinal Infection Testing Market
Companies in the gastrointestinal infection testing market are investing in molecular diagnostic technologies like polymerase chain reaction and next-generation sequencing, to improve the accuracy and speed of GI infection testing. For instance, BioFire Diagnostics offers the BioFire FilmArray GI Panel, which detects 22 pathogens within 1 hour.
Automated platforms assist in decreasing manual errors and improve workflow efficiency. This is particularly essential for high-throughput laboratories. Businesses are focusing on developing rapid diagnostic kits that provide results within minutes, thereby enabling timely treatment.
Companies are progressively partnering with healthcare providers to promote the adoption of advanced diagnostic technologies. Joint research initiatives with academic institutions enable the development of innovative testing methods.
Recent Industry Developments
- In August 2024, MP Biomedicals introduced a new series of in vitro diagnostic tests utilizing immunochromatographic technology to detect gastrointestinal pathogens. The kit focuses on Helicobacter pylori, Salmonella typhi, and Vibrio cholerae serogroups O1 and O139.
- In May 2024, Geneoscopy, Inc. announced that the U.S. Food and Drug Administration (FDA) has approved its noninvasive colorectal cancer screening test, ColoSense. ColoSense was indicated as a screening test for adults aged 45 years or older who were at typical average risk for developing CRC.
Companies Covered in Gastrointestinal Infection Testing Market
- Bio-Rad Laboratories, Inc.
- Thermo Fisher Scientific, Inc.
- Abbott Laboratories
- F. Hoffmann-La Roche AG
- Cepheid (a Danaher company)
- DiaSorin S.p.A.
- bioMérieux SA
- Quidel Corporation
- Hologic, Inc.
- Becton, Dickinson and Company (BD)
- Meridian Bioscience, Inc.
- Qiagen N.V.
- PerkinElmer, Inc.
- Illumina, Inc.
- Accelerate Diagnostics, Inc.
- Seegene, Inc.
- MP Biomedicals
- Geneoscopy, Inc.
- Others
Frequently Asked Questions
The market is anticipated to reach a value of US$ 617.7 Mn by 2032.
Tests for GI include molecular tests, immunoassays, and culture tests.
North America is anticipated to emerge as the leading region with a share of 35% in 2025.
Prominent players in the market include Bio-Rad Laboratories, Inc., Thermo Fisher Scientific, Inc., and Abbott Laboratories.
The market is predicted to witness a CAGR of 5.2% throughout the forecast period.








