- Executive Summary
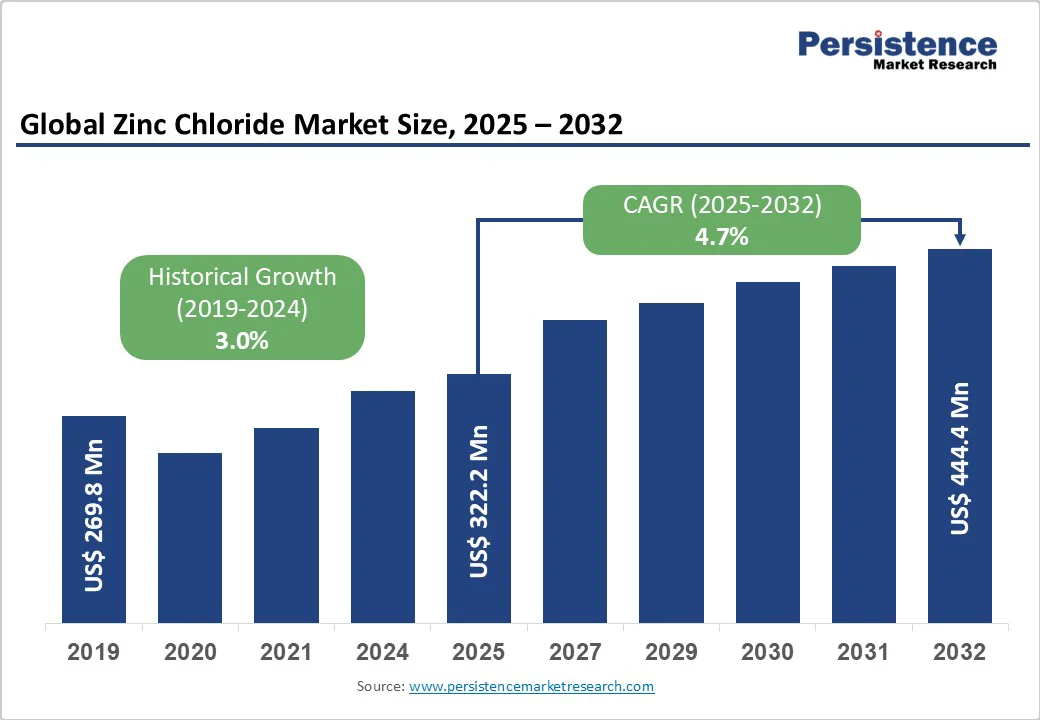
- Global Zinc Chloride Market Snapshot 2025 and 2032
- Market Opportunity Assessment, 2025-2032, US$ Mn
- Key Market Trends
- Industry Developments and Key Market Events
- Demand Side and Supply Side Analysis
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definitions
- Value Chain Analysis
- Macro-Economic Factors
- Global GDP Outlook
- Global Chemical Industry Overview
- Global Textile Industry Overview
- Global Wood Industry Overview
- Forecast Factors – Relevance and Impact
- COVID-19 Impact Assessment
- PESTLE Analysis
- Porter's Five Forces Analysis
- Geopolitical Tensions: Market Impact
- Regulatory and Technology Landscape
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Price Trend Analysis, 2019 – 2032
- Region-wise Price Analysis
- Price by Segments
- Price Impact Factors
- Global Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Global Zinc Chloride Market Outlook: Grade
- Introduction/Key Findings
- Historical Market Size (US$ Mn) and Volume (Units) Analysis by Grade, 2019-2024
- Current Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- Market Attractiveness Analysis: Grade
- Global Zinc Chloride Market Outlook: Form
- Introduction/Key Findings
- Historical Market Size (US$ Mn) and Volume (Units) Analysis by Form, 2019-2024
- Current Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- Market Attractiveness Analysis: Form
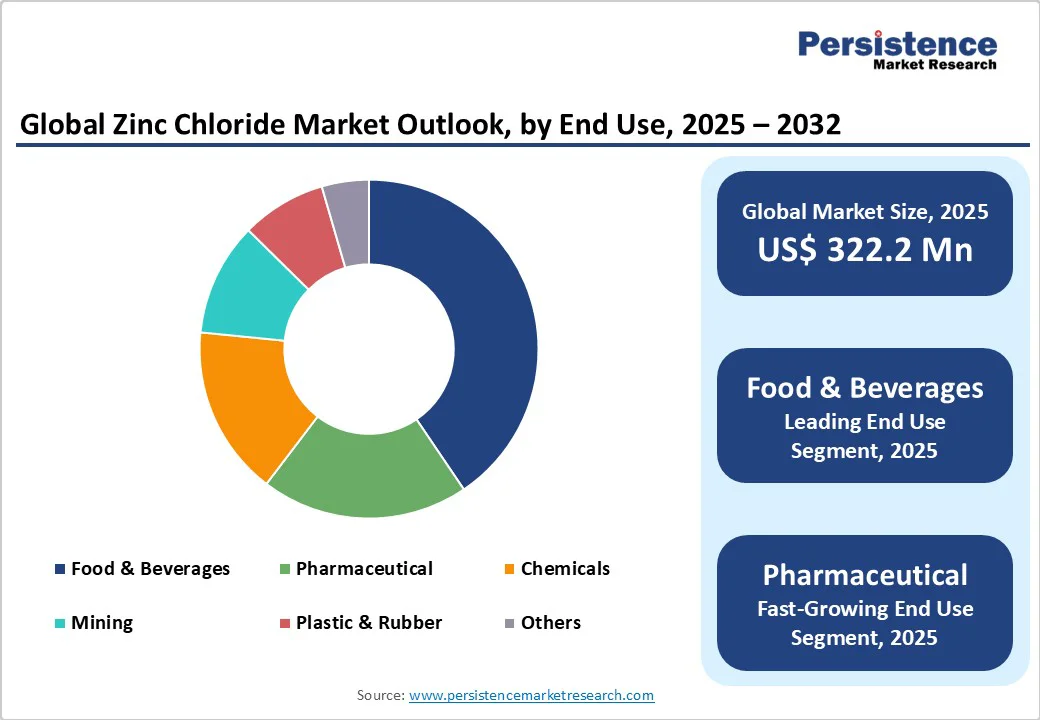
- Global Zinc Chloride Market Outlook: End-Use
- Introduction/Key Findings
- Historical Market Size (US$ Mn) and Volume (Units) Analysis by End-Use, 2019-2024
- Current Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- Market Attractiveness Analysis: End-Use
- Global Zinc Chloride Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Mn) and Volume (Units) Analysis by Region, 2019-2024
- Current Market Size (US$ Mn) and Volume (Units) Forecast, by Region, 2025-2032
- North America
- Europe
- East Asia
- South Asia & Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Pricing Analysis
- North America Market Size (US$ Mn) and Volume (Units) Forecast, by Country, 2025-2032
- U.S.
- Canada
- North America Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- North America Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- North America Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- Europe Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Pricing Analysis
- Europe Market Size (US$ Mn) and Volume (Units) Forecast, by Country, 2025-2032
- Germany
- Italy
- France
- U.K.
- Spain
- Russia
- Rest of Europe
- Europe Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- Europe Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- Europe Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- East Asia Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Pricing Analysis
- East Asia Market Size (US$ Mn) and Volume (Units) Forecast, by Country, 2025-2032
- China
- Japan
- South Korea
- East Asia Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- East Asia Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- East Asia Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- South Asia & Oceania Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Pricing Analysis
- South Asia & Oceania Market Size (US$ Mn) and Volume (Units) Forecast, by Country, 2025-2032
- India
- Southeast Asia
- ANZ
- Rest of SAO
- South Asia & Oceania Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- South Asia & Oceania Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- South Asia & Oceania Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- Latin America Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Pricing Analysis
- Latin America Market Size (US$ Mn) and Volume (Units) Forecast, by Country, 2025-2032
- Brazil
- Mexico
- Rest of LATAM
- Latin America Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- Latin America Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- Latin America Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- Middle East & Africa Zinc Chloride Market Outlook: Historical (2019 – 2024) and Forecast (2025 – 2032)
- Key Highlights
- Pricing Analysis
- Middle East & Africa Market Size (US$ Mn) and Volume (Units) Forecast, by Country, 2025-2032
- GCC Countries
- South Africa
- Northern Africa
- Rest of MEA
- Middle East & Africa Market Size (US$ Mn) and Volume (Units) Forecast, by Grade, 2025-2032
- Battery Grade
- Industrial Grade
- Reagent/High-Purity Grade
- Middle East & Africa Market Size (US$ Mn) and Volume (Units) Forecast, by Form, 2025-2032
- Liquid (Aqueous Solution)
- Powder (Anhydrous)
- Granules and Pellets
- Agrochemicals
- Chemical Synthesis
- Others
- Middle East & Africa Market Size (US$ Mn) and Volume (Units) Forecast, by End-Use, 2025-2032
- Metal Surface Treatment
- Catalyst and Reactant
- Textile Processing
- Wood Preservation
- Deodorizing and Disinfection
- Others
- Competition Landscape
- Market Share Analysis, 2024
- Market Structure
- Competition Intensity Mapping
- Competition Dashboard
- Company Profiles
- Apex Chemicals Corporation
- Company Overview
- Product Portfolio/Offerings
- Key Financials
- SWOT Analysis
- Company Strategy and Key Developments
- Eurocontal SA
- Weifang Dongfangsheng Chemical Co., Ltd
- PT Indo Lysaght
- Flaurea Chemicals
- Vijaychem Industries
- Eurocontal SA
- PT. Indo Lysaght
- Global Chemical Co. Ltd
- TIB Chemical AG
- Lipmes
- Haihua Industry
- Pan-Continental Chemical Co., Ltd
- Yanggu Zhongtian Zinc Industry Co., Ltd.
- Apex Chemicals Corporation
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations

Loading page data
Please wait a moment