
- Pharmaceuticals
- Pediatric Epilepsy Therapeutics Market
Pediatric Epilepsy Therapeutics Market Size, Share, and Growth Forecast, 2026 - 2033
Pediatric Epilepsy Therapeutics Market by Drug Type (Anti-Epileptic Drugs (AEDs), Gene Therapy, Others), Treatment Modality (Pharmacological Treatment, Others), End-user (Children's Hospitals, Specialty Clinics, Others), and Regional Analysis for 2026 - 2033
Pediatric Epilepsy Therapeutics Market Size and Trends Analysis
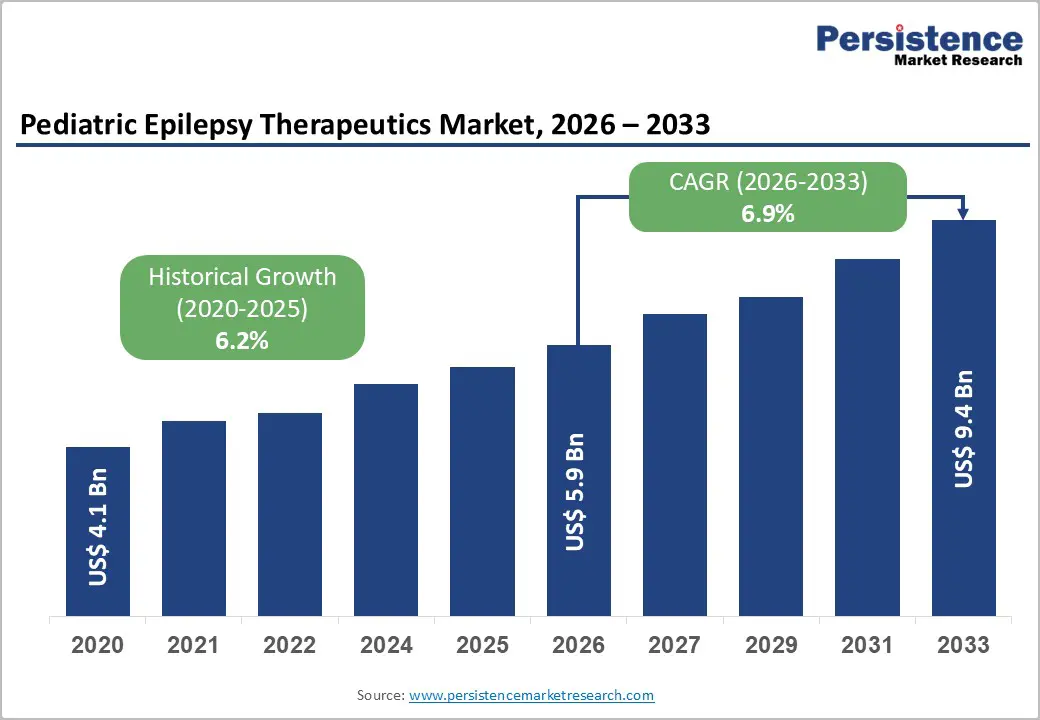
The global pediatric epilepsy therapeutics market size is likely to be valued at US$5.9 billion in 2026, and is expected to reach US$9.4 billion by 2033, growing at a CAGR of 6.9% during the forecast period from 2026 to 2033, driven by the rising prevalence of epilepsy in children, advancements in targeted therapies, and increasing focus on improving long-term neurological outcomes. Increasing adoption of novel anti-epileptic drugs and neurostimulation technologies for better seizure control remains a major driver of the pediatric epilepsy therapeutics market growth.
Key Industry Highlights:
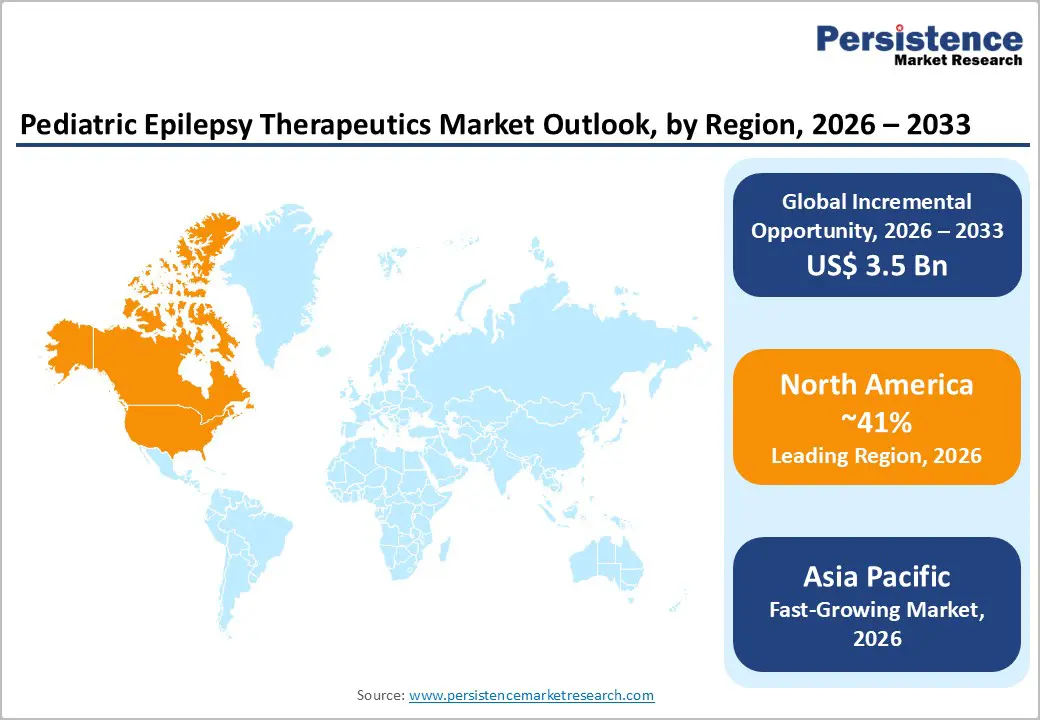
- Leading Region: North America, anticipated to account for a 41% market share in 2026, driven by a combination of high disease awareness, advanced healthcare infrastructure, and widespread access to innovative treatments.
- Fastest-growing Region: Asia Pacific, fueled by improving healthcare infrastructure, rising awareness, and growing pediatric epilepsy diagnosis in China and India.
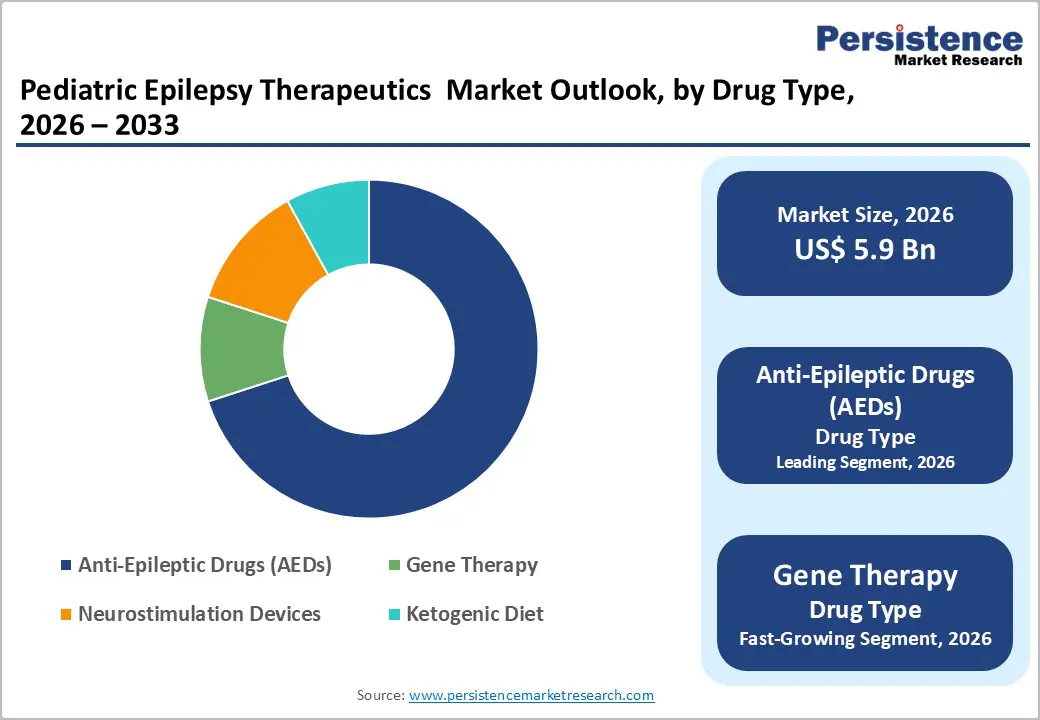
- Dominant Drug Type: Anti-epileptic drugs (AEDs), to hold approximately 62% of the market share, as they remain the first-line treatment.
- Leading Treatment Modality: Pharmacological treatments are set to dominate with 70% share in 2026, driven by proven efficacy, ease of use, and pediatric-friendly formulations such as Levetiracetam, Valproate, and Carbamazepine.
| Key Insights | Details |
|---|---|
| Pediatric Epilepsy Therapeutics Market Size (2026E) | US$5.9 Bn |
| Market Value Forecast (2033F) | US$9.4 Bn |
| Projected Growth CAGR (2026 - 2033) | 6.9% |
| Historical Market Growth (2020 - 2025) | 6.2% |
DRO Analysis
Driver Analysis - Rising Prevalence of Pediatric Epilepsy
A growing burden of neurological disorders in children is significantly increasing the demand for effective epilepsy treatments. Epilepsy is one of the most common chronic brain disorders in childhood, with a substantial proportion of cases beginning early in life. According to the Centers for Disease Control and Prevention, approximately 456,000 children (aged 0-17 years) in the U.S. have active epilepsy, highlighting the large pediatric patient pool requiring ongoing therapeutic management.
Globally, the condition also represents a major health burden. Data from large-scale epidemiological studies indicate that over 18 million children and adolescents were living with epilepsy worldwide in 2021, emphasizing the widespread nature of the disease. Epilepsy tends to occur more frequently in early childhood, with incidence rates peaking during the first years of life due to factors such as birth complications, genetic abnormalities, and infections. In developing countries like India, the burden remains notable. A systematic analysis shows that around 0.8% of children and adolescents are affected by epilepsy, with certain regions reporting even higher prevalence rates. This rising and geographically widespread prevalence is driving continuous demand for anti-epileptic drugs, advanced therapies, and long-term disease management solutions, thereby acting as a key growth driver for the pediatric epilepsy therapeutics market.
High Unmet Medical Need in Drug-Resistant Epilepsy
A substantial proportion of children with epilepsy continue to experience seizures despite receiving appropriate anti-seizure medications, creating a critical gap in effective treatment outcomes. This condition, often referred to as drug-resistant epilepsy, significantly increases the complexity of disease management and the need for alternative therapeutic approaches. According to the World Health Organization, up to 30% of people with epilepsy do not respond adequately to currently available medications, indicating a large population requiring additional care strategies.
In pediatric cases, the challenge is even more pronounced due to developing brains, varied epilepsy syndromes, and limited availability of age-specific therapies. The National Institute of Neurological Disorders and Stroke notes that children with drug-resistant epilepsy are at higher risk of developmental delays, cognitive impairment, and reduced quality of life. Persistent seizures also increase the likelihood of hospitalization and long-term neurological complications. This treatment gap is driving demand for innovative solutions, including combination drug regimens, neuromodulation techniques such as vagus nerve stimulation, ketogenic dietary therapies, and emerging gene-based treatments.
Restraint Analysis - Adverse Effects of Anti-Epileptic Drugs (AEDs)
Treatment with anti-seizure medications in children is often associated with a range of side effects that can impact both physical and cognitive development. Commonly used drugs such as Valproate and Carbamazepine may cause drowsiness, dizziness, gastrointestinal issues, and behavioral changes. In pediatric patients, these effects can interfere with learning ability, attention span, and overall school performance.
Long-term use also raises concerns about growth disturbances, hormonal imbalances, and potential effects on brain development. According to the Centers for Disease Control and Prevention, managing side effects is a critical part of epilepsy care, especially in children who require prolonged therapy. Some medications carry risks of severe reactions such as skin disorders or liver toxicity, necessitating continuous monitoring.
Limited Efficacy in Drug-Resistant Epilepsy
A considerable number of pediatric patients continue to experience uncontrolled seizures even after trying multiple anti-seizure medications, highlighting the limitations of current therapeutic approaches. This condition, clinically recognized as drug-resistant epilepsy, is typically defined when seizures persist despite the use of two or more appropriately chosen and tolerated drugs.
Children affected by this condition often face frequent seizures, which can impair cognitive development, behavior, and overall quality of life. According to the World Health Organization, a significant share of epilepsy patients does not achieve complete seizure control with existing medications, underscoring the need for alternative solutions. The variability in underlying causes, including genetic mutations and structural brain abnormalities, further complicates treatment response.
Opportunity Analysis - Development of Targeted and Precision Therapies
Advances in genetic research and molecular diagnostics are transforming the way epilepsy is treated in children by enabling therapies tailored to the underlying cause of the disorder rather than just controlling symptoms. Many pediatric epilepsy syndromes, such as Dravet Syndrome, are now known to result from specific gene mutations, allowing clinicians to adopt more personalized treatment strategies.
Precision approaches involve identifying the exact genetic or biochemical pathway responsible for seizures and selecting therapies that directly target those mechanisms. For instance, sodium channel blockers may be avoided in certain genetic conditions where they can worsen seizures, while newer treatments are being designed to modulate specific neural pathways. According to the National Institute of Neurological Disorders and Stroke, ongoing genomics research is improving diagnosis accuracy and helping match patients with more effective therapies. Advancements in biomarkers and diagnostic tools are supporting earlier and more precise intervention.
Growing Adoption of Digital Health and Remote Monitoring
Increasing integration of technology into healthcare is transforming the management of pediatric epilepsy by enabling continuous and real-time monitoring of patients outside clinical settings. Digital tools such as wearable seizure-detection devices, mobile applications, and cloud-based platforms help accurately track seizure frequency, duration, and triggers. Applications like Seizure Tracker enable caregivers and physicians to maintain detailed logs, which improve treatment decisions and medication adjustments.
Remote monitoring is particularly beneficial for children who require long-term supervision, as it reduces the need for frequent hospital visits while ensuring timely medical intervention. According to the Centers for Disease Control and Prevention, effective epilepsy management depends heavily on consistent monitoring and adherence to treatment plans, which digital solutions help facilitate. Wearable technologies can also provide alerts during seizure episodes, enhancing patient safety and caregiver responsiveness. Telemedicine platforms enable virtual consultations with specialists, improving access to pediatric neurologists in remote or underserved areas.
Category-wise Analysis
Drug Type Insights
Anti-epileptic drugs (AEDs) are anticipated to dominate, accounting for 62% of the market share in 2026, due to their proven effectiveness in controlling a wide range of seizure types and ease of administration. Commonly prescribed drugs such as Levetiracetam and Valproate are widely used as first-line therapies, offering relatively quick symptom control. Their availability in multiple formulations suitable for children further supports adherence. Levetiracetam oral solution is commonly used in pediatric epilepsy treatment as a first-line option because of its broad-spectrum effectiveness and strong safety profile in children. Marketed by UCB Pharma under the brand name Keppra, it is widely prescribed for the management of partial-onset seizures in infants and young patients, with its liquid formulation enabling accurate and convenient dosing.
Gene therapy represents the fastest-growing segment, driven by rapid progress in genetic research, which is accelerating the adoption of gene-based treatments for pediatric epilepsy, particularly for rare and severe conditions with known genetic causes. Unlike conventional therapies that mainly control symptoms, gene therapy aims to address the root cause by correcting or modifying faulty genes. Disorders such as Dravet Syndrome are key targets for these emerging approaches. Encoded Therapeutics is advancing ETX101, a potential one-time gene regulation therapy aimed at modifying the course of Dravet Syndrome. As the company’s lead program, it is specifically developed to target the root genetic cause mutations in the SCN1A gene responsible for this severe developmental and epileptic encephalopathy.
Treatment Modality Insights
Pharmacological treatment is projected to dominate, with 70% of the share in 2026, fueled by its proven effectiveness across various seizure types and ease of administration. Medications such as Levetiracetam, Valproate, and Carbamazepine are widely used as first-line therapies, offering reliable seizure control and multiple pediatric-friendly formulations. Their established safety profiles and adaptability to different age groups ensure high adherence and long-term use. Valproate (sodium valproate) syrup, marketed by Abbott Laboratories under the brand name Depakene, is commonly prescribed for pediatric patients to manage both generalized and partial-onset seizures. Its liquid formulation enables precise dosing for infants and young children, making it particularly well-suited for long-term treatment regimens.
Neurostimulation is the fastest-growing modality, supported by a rapidly expanding treatment approach in pediatric epilepsy, particularly for children with drug-resistant seizures. Techniques such as vagus nerve stimulation (VNS) and responsive neurostimulation directly modulate neural activity to reduce seizure frequency without relying solely on medications. Technological improvements in device design, battery life, and programmable stimulation settings have enhanced safety, precision, and patient comfort. Vagus Nerve Stimulation (VNS) Therapy, created and marketed by LivaNova, is approved for pediatric patients with drug-resistant epilepsy. The VNS Therapy System delivers controlled electrical impulses to the vagus nerve, which helps reduce the frequency of seizures in children who are unresponsive to conventional medications.
Regional Insights
North America Pediatric Epilepsy Therapeutics Market Trends
North America is projected to dominate, accounting for 41% of the share in 2026, driven by a combination of high disease awareness, advanced healthcare infrastructure, and widespread access to innovative treatments. The region has a significant pediatric epilepsy patient population, with the Centers for Disease Control and Prevention reporting approximately 456,000 children living with active epilepsy in the U.S. Early diagnosis, facilitated by well-established screening programs and access to pediatric neurologists, allows timely initiation of therapies, improving clinical outcomes and quality of life.
Pharmacological treatments, including widely prescribed drugs such as Levetiracetam and Valproate, continue to dominate the market due to their efficacy and availability in child-friendly formulations. Emerging modalities such as neurostimulation and gene therapy are gaining traction, particularly for children with drug-resistant or genetically defined epilepsy syndromes. The region also benefits from strong R&D investment, regulatory support for orphan and rare disease therapies, and the adoption of digital health tools such as wearable seizure monitors and remote-tracking apps. These factors collectively enhance patient management, adherence, and long-term outcomes.
Europe Pediatric Epilepsy Therapeutics Market Trends
The European market is expanding steadily due to increasing awareness, improved healthcare access, and growing adoption of advanced treatment options. European countries benefit from well-established healthcare systems that provide early diagnosis and specialized care through pediatric neurology centers. Early identification of epilepsy in children enables the timely initiation of anti-epileptic drugs (AEDs) and other therapeutic interventions, improving seizure control and reducing long-term developmental complications.
Pharmacological treatments remain the primary approach, with medications such as Levetiracetam and Valproate widely prescribed due to their proven efficacy and safety in pediatric patients. Emerging therapies, including neurostimulation techniques such as vagus nerve stimulation and gene therapies targeting rare epilepsy syndromes, are gaining adoption, particularly for children with drug-resistant or genetically driven epilepsy. Europe also sees increased integration of digital health technologies, including wearable seizure-monitoring devices and telemedicine platforms, that support continuous patient monitoring, caregiver engagement, and remote consultations.
Asia Pacific Pediatric Epilepsy Therapeutics Market Trends
Asia Pacific is likely to be the fastest-growing region supported by increasing disease awareness, improving healthcare infrastructure, and rising adoption of advanced therapies. Large populations in countries such as India, China, and Japan contribute to a substantial pediatric epilepsy patient base. Limited awareness in rural areas is gradually being addressed through public health initiatives, screening programs, and education campaigns, enabling earlier diagnosis and timely intervention for children.
Pharmacological treatments remain the cornerstone of therapy, with drugs such as Levetiracetam and Valproate widely used due to their efficacy, safety, and availability in child-friendly formulations. Emerging modalities such as neurostimulation and gene therapies are also slowly gaining traction, especially for children with drug-resistant or genetically defined epilepsy. Healthcare improvements, including the expansion of pediatric neurology centers, better access to specialists, and government support for rare disease therapies, are facilitating market growth. Adoption of digital health tools, such as wearable seizure monitors and mobile applications for remote tracking, further enhances disease management and treatment adherence.
Competitive Landscape
The global pediatric epilepsy therapeutics market is moderately consolidated, dominated by a mix of pharmaceutical and medical device companies that emphasize innovation and advanced treatment options. Market competition primarily revolves around clinical efficacy, safety in children, and the ability to address drug-resistant epilepsy, which remains a significant unmet need.
Leading companies, including Pfizer, Novartis, GW Pharmaceuticals, Medtronic, Eisai Co., Ltd., and UCB Pharma, are actively developing safer and more effective anti-epileptic drugs. In addition, they are investing in cutting-edge gene therapies and neurostimulation technologies to target pediatric patients with drug-resistant conditions. Expansion into emerging markets, partnerships with pediatric neurology centers, and collaboration on clinical research are key strategies these players use to enhance patient outcomes, broaden access, and maintain competitive advantage in this specialized therapeutic area.
Key Industry Developments:
- In March 2026, researchers at Northwestern University Feinberg School of Medicine and Ann & Robert H. Lurie Children’s Hospital of Chicago completed Phase 1/2a trials of zorevunersen, a gene-regulation therapy for Dravet Syndrome. The therapy was safe, reduced seizures, and improved motor and communication skills in children aged 2-18. Targeting the SCN1A gene, it addressed the disease’s root cause.
Companies Covered in Pediatric Epilepsy Therapeutics Market
- Pfizer
- Novartis
- GW Pharmaceuticals
- Medtronic
- Eisai Co., Ltd.
- UCB Pharma
Frequently Asked Questions
The global pediatric epilepsy therapeutics market is projected to reach US$5.9 billion in 2026.
The pediatric epilepsy therapeutics market is largely fueled by the increasing incidence of epilepsy among children and ongoing innovations in anti-epileptic drugs and neurostimulation treatments.
The pediatric epilepsy therapeutics market is poised to witness a CAGR of 6.9% from 2026 to 2033.
Major opportunities in the pediatric epilepsy therapeutics market lie in advancing gene therapies, broadening the use of neurostimulation technologies, and expanding home healthcare and ambulatory care services for long-term epilepsy management.
Key players in the pediatric epilepsy therapeutics market include Pfizer, Novartis, Eisai Co., Ltd., UCB Pharma, and Medtronic.







