- Biotechnology
- Oligonucleotide Therapeutics Market
Oligonucleotide Therapeutics Market Size, Share, and Growth Forecast, 2025 - 2032
Oligonucleotide Therapeutics Market by Treatment Type (Antisense, Ribozymes, Aptamers, miRNA, CpG/Immunostimulatory, RNAi), Application (Infectious Diseases, Oncology, Neurodegenerative Disorders, Cardiovascular Diseases, Kidney Diseases, Others), and Regional Analysis for 2025 - 2032
Oligonucleotide Therapeutics Market Size and Trend Analysis
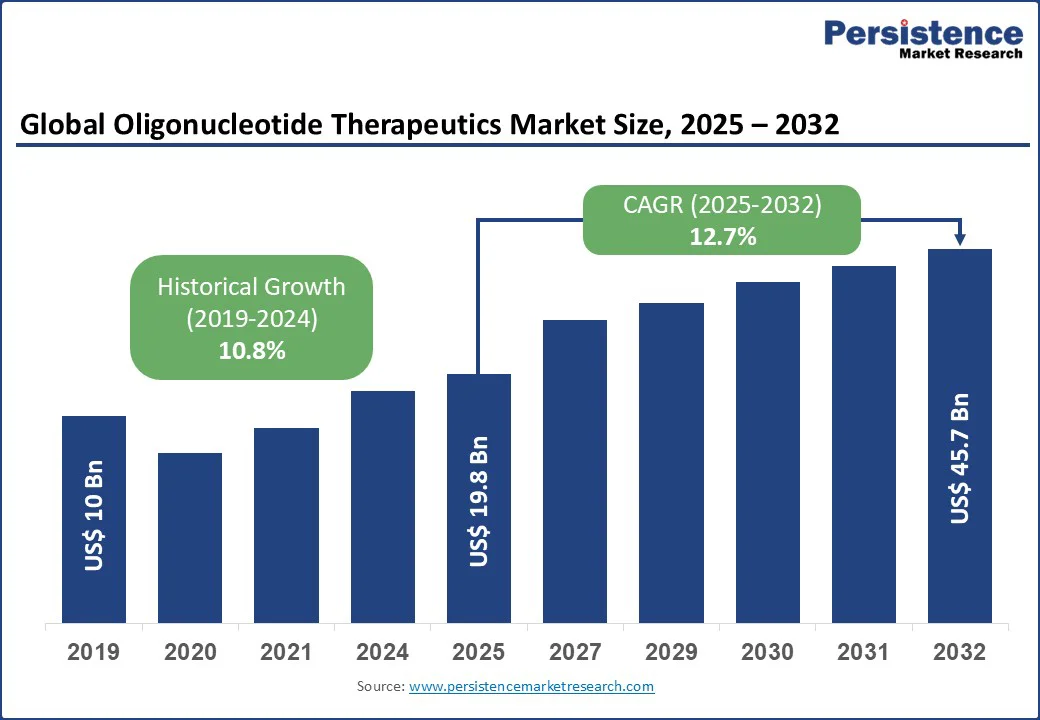
The global oligonucleotide therapeutics market size is likely to be value at US$ 19.8 Bn in 2025 and reach US$ 45.7 Bn by 2032, growing at a CAGR of 12.7% during the forecast period from 2025 to 2032 driven by increasing demand for precision medicine and advancements in genetic research.
Key Industry Highlights
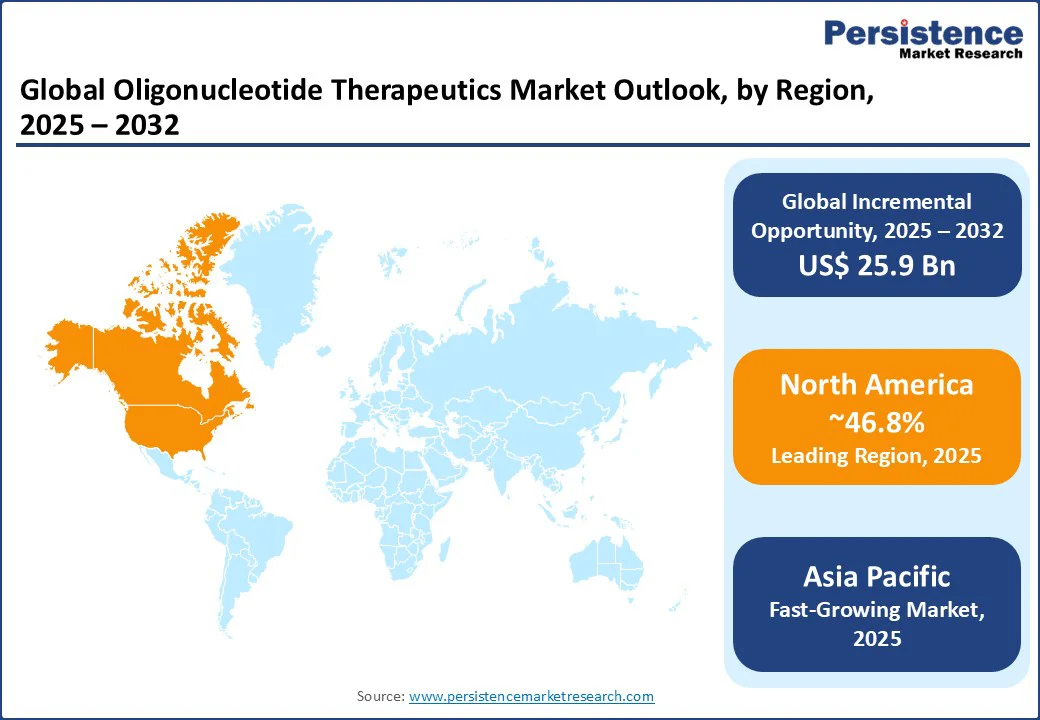
- Leading Region: North America holds 46.8% market share in 2025, propelled by advanced biotechnology infrastructure, high R&D investments, and strong regulatory support in the U.S. and Canada.
- Fastest-growing Region: Asia Pacific is the fastest-growing region, driven by increasing biotech investments and the growing prevalence of chronic diseases in countries such as China and India.
- Investment Plans: Statista lists “human genome funding by the NIH” with an estimated value of around USD 5 billion in fiscal year 2023, boosting demand for oligonucleotide therapeutics in clinical applications.
- Dominant Treatment Type: Antisense oligonucleotides account for nearly 56.8% of the oligonucleotide therapeutics market share, due to their specificity and efficacy in targeting genetic disorders and cancers.
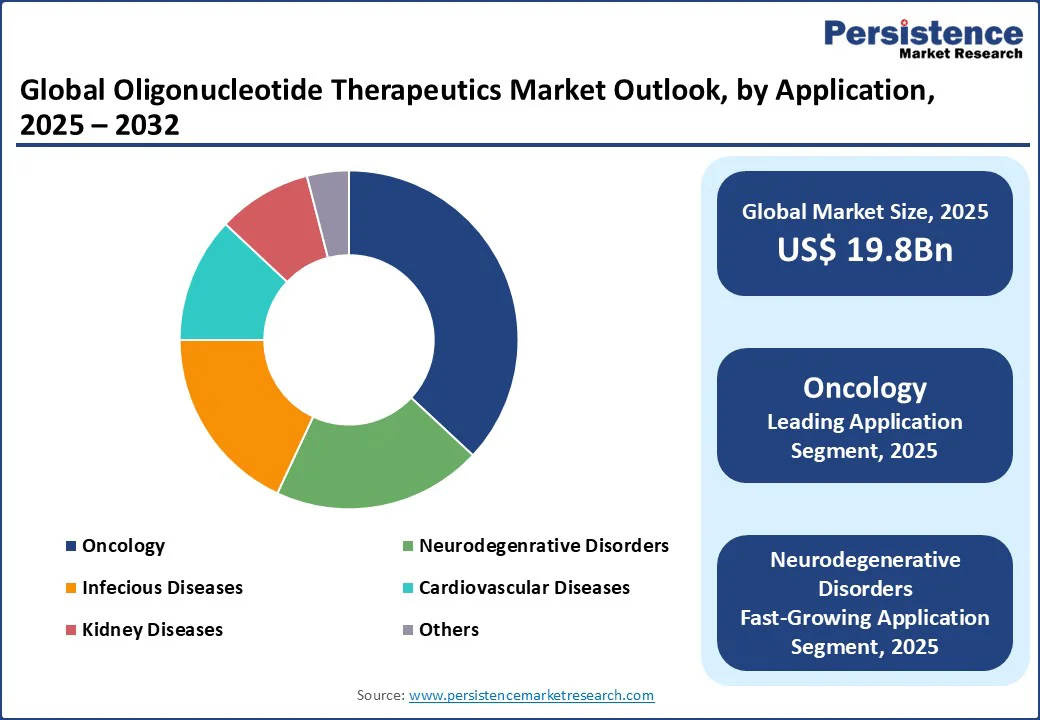
- Leading Application: Oncology contributes over 36.9% of market revenue, driven by the rising incidence of cancer and demand for targeted therapies.
|
Key Insights |
Details |
|
Oligonucleotide Therapeutics Market Size (2025E) |
US$ 19.8Bn |
|
Market Value Forecast (2032F) |
US$ 45.7Bn |
|
Projected Growth (CAGR 2025 to 2032) |
12.7% |
|
Historical Market Growth (CAGR 2019 to 2024) |
10.8% |
Market Dynamics
Driver - Growing Demand for Precision Medicine and Targeted Therapies Fuels Market Expansion
The global oligonucleotide therapeutics market is rapidly expanding, driven by the increasing demand for precision medicine and targeted therapies. Oligonucleotide-based drugs—particularly antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs)—enable highly specific modulation of gene expression, offering transformative potential for treating complex conditions such as genetic disorders, cancer, and neurodegenerative diseases. The World Health Organization projects that chronic diseases will account for 74% of global deaths by 2025, underscoring the urgent need for more precise and personalized treatment options.
In the U.S., the FDA had approved 21 RNA oligonucleotide therapeutics by 2024, including landmark drugs such as Spinraza for spinal muscular atrophy, further validating this class of treatments. Industry leaders such as Ionis Pharmaceuticals reported increased sales of ASO-based therapies, reflecting strong commercial uptake. Coupled with government investments in genomic research and national precision medicine initiatives, the market is well-positioned for sustained growth, with projections extending robustly through 2032 and beyond.
Restraint - Delivery Challenges and High Development Costs Hinder Market Growth
The oligonucleotide therapeutics market faces notable challenges that hinder its full potential despite its promising therapeutic benefits. One of the primary obstacles is the inefficiency of in vivo delivery, which remains a significant hurdle for many candidates. Furthermore, stringent regulatory frameworks in major markets such as Europe and North America contribute to longer development timelines and increased costs, disproportionately affecting smaller biotech firms with limited resources.
Competition from established small-molecule drugs adds another layer of pressure, particularly in cost-sensitive regions where pricing is a critical factor. Additionally, concerns around potential toxicity and challenges in scaling up manufacturing processes further complicate market growth. Together, these factors create a complex environment that restrains widespread adoption and slows down the commercial expansion of oligonucleotide therapeutics.
Opportunity - Rising Investments in Rare Disease Research and Gene Therapies
The growing emphasis on rare disease research and gene therapies is fueling robust opportunities in the oligonucleotide therapeutics market. Oligonucleotides play a pivotal role in treating rare genetic disorders such as Duchenne muscular dystrophy and Huntington’s disease by precisely targeting disease-causing gene mutations. This precision has transformed treatment paradigms, offering hope for previously untreatable conditions. Investor confidence in this sector is exemplified by Tune Therapeutics $175 million Series B funding in January 2025 to advance its gene-tuning therapy for chronic hepatitis B, underscoring the promise of epigenetic and gene-editing approaches.
Leading companies such as Alnylam Pharmaceuticals continue to innovate with RNA interference (RNAi) therapies tailored to rare diseases, aligning closely with global health priorities. Moreover, government initiatives such as the EU’s Horizon Europe program provide crucial financial incentives and regulatory support, accelerating research and development in precision medicine. Together, these factors position the oligonucleotide therapeutics market for significant growth and innovation through 2032, addressing evolving clinical needs worldwide.
Category-wise Insights
By Treatment Type
- Antisense oligonucleotides (ASOs) hold the largest market share, approximately 56.8% in 2025, due to their high specificity and efficacy in targeting disease-causing genes. Widely used in oncology and genetic disorders, ASOs modulate gene expression with reduced toxicity, as seen in drugs such as Spinraza. Companies such as Ionis Pharmaceuticals and Biogen lead with extensive ASO portfolios, catering to demand in North America and Europe for precision therapies.
- RNA interference (RNAi) is the fastest-growing segment, driven by its potential in silencing specific genes for conditions such as neurodegenerative disorders and cancers. Its adoption is rising due to advancements in delivery systems, such as nanoparticle-based technologies, with brands such as Alnylam Pharmaceuticals expanding offerings in the Asia Pacific and North America, supported by growing clinical trial success.
By Application
- The oncology segment accounts for over 36.9% of market revenue in 2025, driven by the rising global cancer burden and demand for targeted therapies. Oligonucleotide therapeutics, particularly ASOs and siRNAs, are critical for treating cancers such as leukemia and breast cancer by targeting specific oncogenes. Major players such as Alnylam Pharmaceuticals and Pfizer supply advanced therapies for clinical applications, with the U.S. and Europe leading in adoption due to robust oncology research.
- The neurodegenerative disorders segment is the fastest-growing application, propelled by increasing investments in Alzheimer’s and Parkinson’s research. Oligonucleotides are used to modulate protein expression, with companies such as Ionis Pharmaceuticals innovating for high-efficacy therapies. Growth in North America and the Asia Pacific, driven by aging populations and clinical advancements, supports this segment’s rapid expansion.
Regional Insights
North America Oligonucleotide Therapeutics Market Trends
North America holds a leading position in the global oligonucleotide therapeutics market, commanding approximately 46.8% of the total market share. This dominance is driven by a well-established biotechnology infrastructure, substantial R&D investments, and a strong pipeline of innovative therapies. In the United States, the bioscience industry generated an economic output of $3.2 trillion in 2023, according to the Biotechnology Innovation Organization, with oligonucleotide technologies playing a critical role in advancing precision medicine and genetic therapies.
Canada also contributes significantly, with organizations such as Genome Canada supporting cutting-edge genomics research and precision health initiatives. This regional ecosystem fosters high adoption of targeted oligonucleotide-based therapies, especially in treating oncology and neurodegenerative disorders. Industry leaders such as Alnylam Pharmaceuticals and Biogen have anchored their global R&D strategies in North America, leveraging scientific talent and regulatory support. Additionally, rising consumer demand for high-efficacy, personalized treatments continues to reinforce North America’s leadership in the oligonucleotide therapeutics space.
Asia Pacific Oligonucleotide Therapeutics Market Trends
The Asia Pacific region is emerging as the fastest-growing market for oligonucleotide therapeutics, fueled by robust biotechnology investments and a growing burden of chronic and genetic diseases. In China, public funding for biotechnology research reached CNY 20 billion in 2023, according to government-backed reports, reflecting the country’s strategic focus on becoming a global biotech leader. China’s commitment to cutting-edge therapies, including gene and RNA-based treatments, is accelerating innovation and clinical adoption. Meanwhile, India’s expanding pharmaceutical and biotech sectors, supported by initiatives such as the Biotechnology Industry Research Assistance Council (BIRAC), are enhancing domestic research capabilities and fostering the development of precision therapies for oncology and rare diseases.
The region also benefits from a rapidly expanding clinical trial ecosystem, with regulatory bodies streamlining approvals to attract global R&D investments. Combined with healthcare reforms aimed at improving access and affordability, the Asia Pacific is positioned for sustained high growth in oligonucleotide therapeutics through 2032 and beyond.
Europe Oligonucleotide Therapeutics Market Trends
Europe stands as the second fastest-growing region in the oligonucleotide therapeutics market, fueled by a combination of advanced biotechnology infrastructure, regulatory rigor, and rising clinical demand. Countries such as Germany and the UK are leading innovation in RNA-based therapeutics, particularly in oncology and neurodegenerative disorders. According to *The Economic Footprint of the Biotechnology Industry in the European Union*, the sector contributed €38.1 billion in gross value added (GVA) in 2022, underscoring its significant role in the continent’s economy and healthcare innovation.
Germany’s strong pharmaceutical landscape, anchored by companies such as Merck KGaA, is a major consumer of antisense oligonucleotide (ASO) therapies. Meanwhile, the EU’s Horizon Europe program actively supports precision medicine initiatives, incentivizing research into advanced oligonucleotide-based treatments. Europe’s focus on regulatory compliance, patient safety, and high therapeutic standards positions it as a critical hub for clinical trials and commercialization. These factors collectively support the region’s accelerating growth in oligonucleotide therapeutics through 2032.
Competitive Landscape
The global oligonucleotide therapeutics market is highly competitive, characterized by a fragmented landscape with numerous domestic and international players. Leading companies such as Ionis Pharmaceuticals, Alnylam Pharmaceuticals, and Biogen dominate through extensive product portfolios and global R&D networks. Regional players, such as InteRNA Technologies B.V., focus on localized offerings in Europe.
Companies are investing in advanced delivery technologies and AI-driven drug design to enhance market share, driven by demand for high-efficacy therapies in oncology and rare diseases.
Key Industry Developments
- February 2025: Biogen and Stoke Therapeutics are collaborating to develop and commercialize zorevunersen, an investigational antisense oligonucleotide (ASO) for Dravet syndrome, a severe genetic epilepsy. Zorevunersen aims to increase NaV1.1 protein production from the healthy SCN1A gene, a potential first-in-class disease-modifying approach. Stoke leads development and retains rights in North America, while Biogen holds rest-of-world commercialization rights. The collaboration includes an upfront payment, shared development costs, and milestone payments for Stoke.
- December 2024: The U.S. Food and Drug Administration (FDA) approved Tryngolza (olezarsen) as the first-ever treatment for adults with familial chylomicronemia syndrome (FCS), a rare genetic disorder characterized by extremely high triglyceride levels. This approval marked Ionis Pharmaceuticals' first independent drug launch.
Companies Covered in Oligonucleotide Therapeutics Market
- PCI Biotech Holding
- SomaGenics Inc
- Hepion Pharmaceuticals
- Alnylam Pharmaceuticals
- Regulus Therapeutics Inc
- Ionis Pharmaceuticals
- Gilead Sciences, Inc
- Santaris
- InteRNA Technologies B.V.
- Mirage
- Biogen
- Merck KGaA
- Others
Frequently Asked Questions
The Oligonucleotide Therapeutics market is projected to reach US$19.8 Bn in 2025.
Growing demand for precision medicine and targeted therapies, and expanding applications in rare disease research are the key market drivers.
The Oligonucleotide Therapeutics market is poised to witness a CAGR of 12.7% from 2025 to 2032.
The rising investments in rare disease research and gene therapies are the key market opportunities.
Ionis Pharmaceuticals, Alnylam Pharmaceuticals, Biogen, and Merck KGaA are key market players.








