
- Biotechnology
- Microbiome Manufacturing Market
Microbiome Manufacturing Market Size, Share, and Growth Forecast, 2026 – 2033
Microbiome Manufacturing Market by Formulation (Solid, Liquid, Others), Primary Packaging (Blister Packs, Glass Bottles, Plastic Bottles, Pouches, Vials), Company Size (Small, Mid-sized, Large & Very Large), and Regional Analysis for 2026-2033
Microbiome Manufacturing Market Share and Trends Analysis
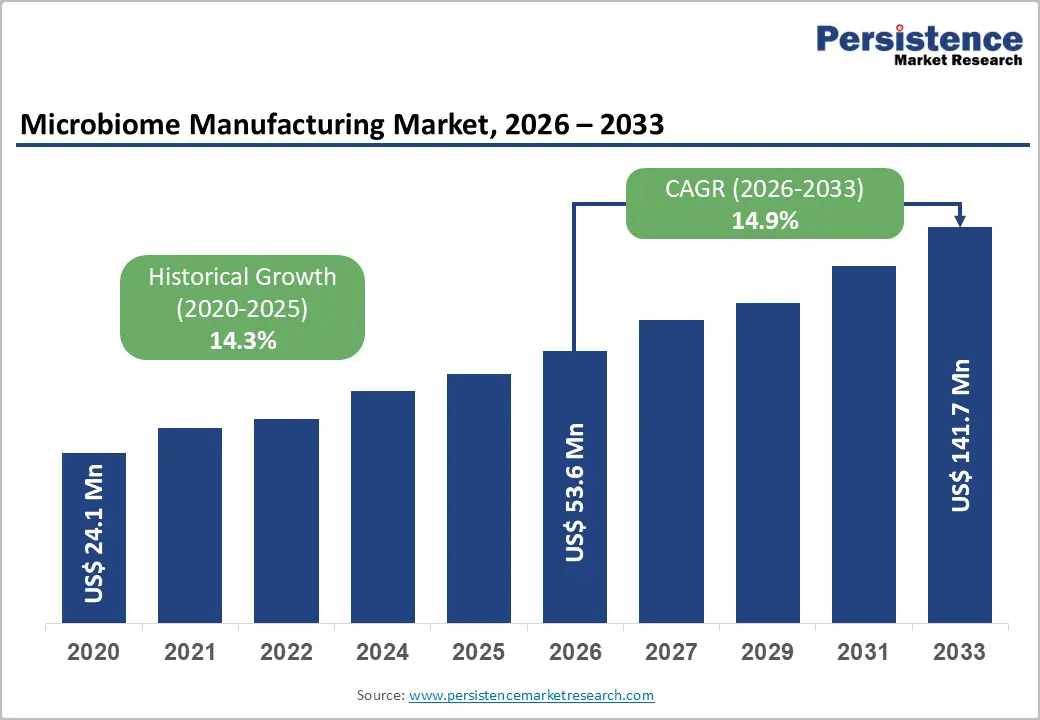
The global microbiome manufacturing market size is likely to be valued at US$ 53.6 million in 2026, and is projected to reach US$ 141.7 million by 2033, growing at a CAGR of 14.9% during the forecast period 2026−2033. Widening clinical recognition of microbiome-based therapies, increased adoption of personalized treatment approaches, integration of advanced manufacturing technologies, and expansion of healthcare infrastructure are factors favoring the market. Demand growth reflects demographic trends such as aging populations and rising prevalence of gastrointestinal, metabolic, and immune-related conditions, which create heightened clinical need for microbiome interventions.
Regulatory developments facilitating clinical trials and approvals encourage investment in scalable microbiome production platforms, while healthcare providers increasingly incorporate microbiome-based diagnostics and therapeutics into treatment protocols. Technological advancements, including automated culture systems, genomic sequencing integration, and high-throughput microbial cultivation, reduce production bottlenecks and enhance reproducibility, improving overall treatment efficacy. Expanding healthcare networks in emerging regions provide broader market access, supporting distribution of microbiome formulations in both clinical and preventive applications.
Key Industry Highlights
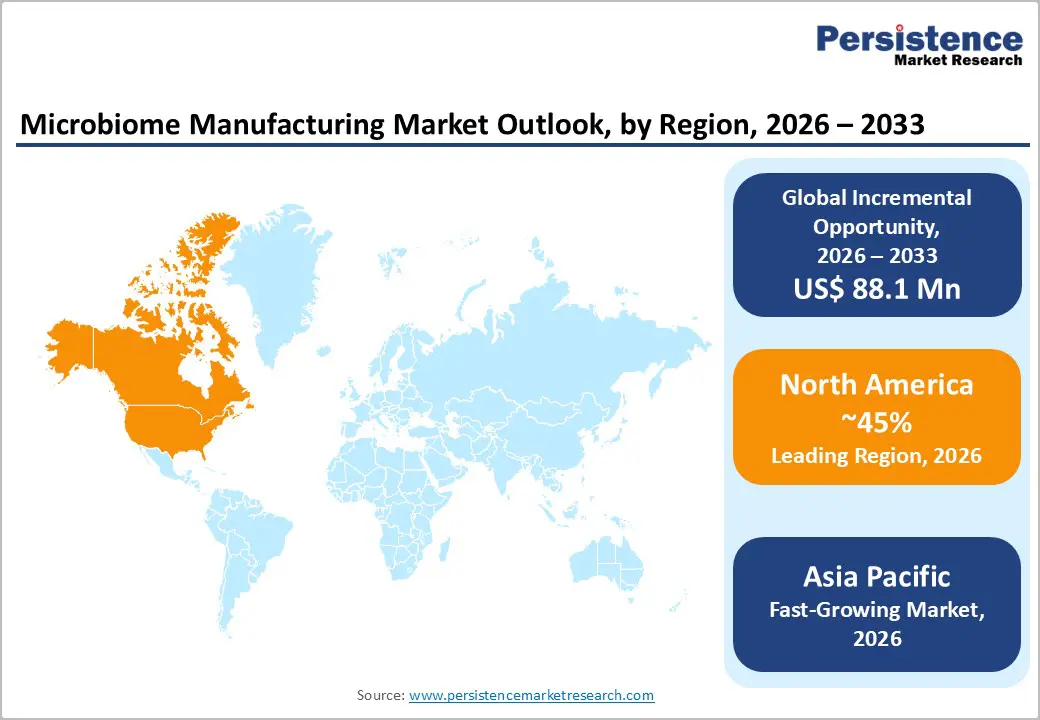
- Dominant Region: By 2026, North America is expected to lead with a 45% market share, driven by strong collaboration among manufacturers, research institutions, and contract research organizations (CROs).
- Fastest-growing Market: Asia Pacific is forecasted to be the fastest-growing market between 2026 and 2033, propelled by increasing frequency of clinical trials and improved biotech funding.
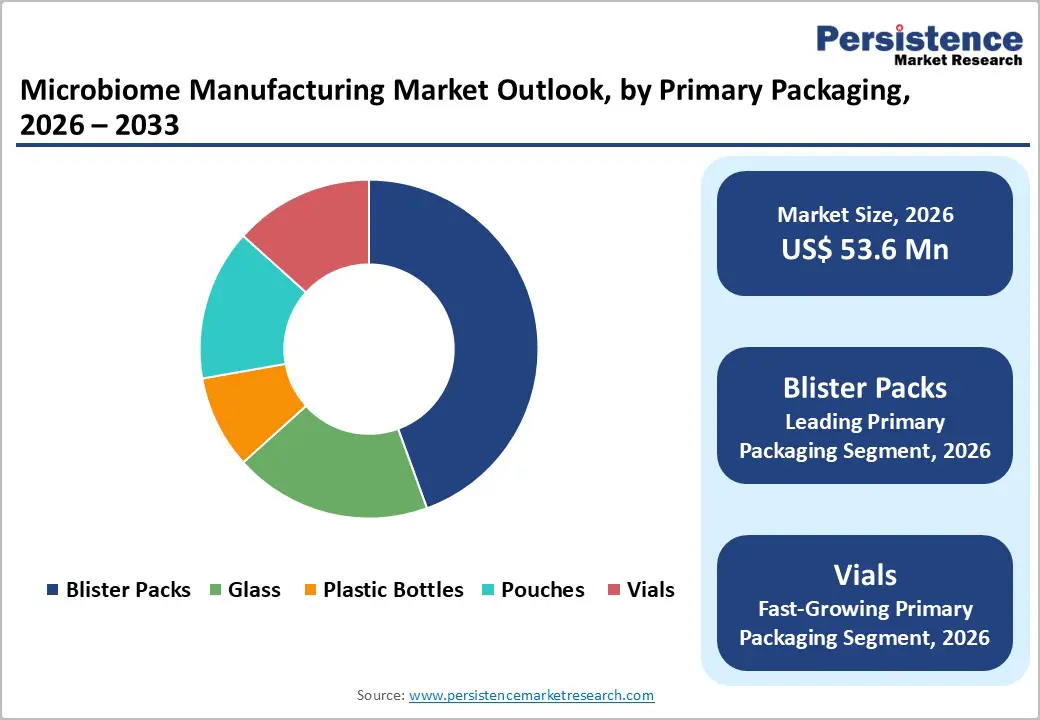
- Leading Primary Packaging: Blister packs are poised to dominate with over 40% market share in 2026, driven by convenience, dosage accuracy, and microbial stability.
- Fastest-growing Primary Packaging: Vials are estimated to be the fastest-growing segment from 2026 to 2033, fueled by liquid formulation compatibility, precise dosing, and automated dispensing.
- February 2026: Liverpool City Region’s Microbiome Innovation Hub was shortlisted for a BioNow 2025 Award, reflecting its contribution to advancing UK microbiome research collaboration and early-stage biotech commercialization infrastructure.
| Key Insights | Details |
|---|---|
| Microbiome Manufacturing Market Size (2026E) | US$ 53.6 Mn |
| Market Value Forecast (2033F) | US$ 141.7 Mn |
| Projected Growth (CAGR 2026 to 2033) | 14.9% |
| Historical Market Growth (CAGR 2020 to 2025) | 14.3% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Increasing Pipeline of Live Biotherapeutic Products
The strength of the live biotherapeutic products (LBPs) development pipeline directly influences demand for specialized manufacturing. LBPs are defined by the U.S. Food and Drug Administration (FDA) as biological products containing live organisms intended to prevent, treat or cure disease, and developers must include detailed chemistry, manufacturing and controls (CMC) documentation in investigational new drug (IND) submissions to support clinical work. This regulatory framework gives developers clear quality and process requirements that inform manufacturing strategy well before late-stage trials, enabling earlier planning of capacity for production scale-up and compliance with stringent quality standards. The existence of established guidance reduces uncertainty about regulatory expectations, which encourages sponsors and manufacturing partners to invest in facility upgrades, anaerobic fermentation capabilities and robust quality systems tailored for LBPs.
A broad and advancing clinical pipeline signals potential future volumes of work that justify investment in complex production capabilities. Industry sources indicate that hundreds of microbiome-based therapies, including LBPs, are under investigation globally, with numerous clinical trials focused on indications such as gastrointestinal disorders and chronic inflammatory conditions. This breadth of investigational programs suggests that manufacturing organizations might support multiple product candidates rather than a single asset, spreading fixed costs across several pipelines and lowering risk. The presence of multiple development programs also encourages collaboration between sponsors and specialized contract manufacturers with expertise in handling live organisms under regulatory quality systems, strengthening partnerships and accelerating technology transfer.
Rising Clinical Awareness and Adoption
Clinical validation of microbiome-directed interventions has led healthcare providers to integrate these therapies into clinical decision-making, which creates stable demand that supports expanded manufacturing operations. Data from the U.S. FDA in 2025 show that an FDA-approved microbiome-based product for preventing recurrent Clostridioides difficile infection achieved a recurrence rate of 12.4% in treated patients versus 39.8% with placebo, affirming clinical benefit and safety in a regulated setting. Clinical decision-makers reference such data when defining treatment pathways, which enhances the confidence of hospital formulary committees and payers in budgeting for these products.
Structured regulatory guidance and clinician education promote standardized use of microbiome therapies across healthcare systems, aligning clinical practice with scalable production needs. When professional societies and regulatory bodies endorse clinical guidelines based on government-validated evidence, health systems adjust procurement plans to support anticipated utilization. Manufacturers interpret this alignment as a clear demand signal, which reduces commercialization risk and accelerates capital allocation to production infrastructure. Consistent clinical adoption also drives long-term contracts with healthcare providers, which improves revenue forecasting and supports strategic decisions for technology platforms and distribution networks.
Complex and Evolving Regulatory Landscape
The regulatory environment for microbiome related products requires multiple, evolving compliance pathways that significantly challenge manufacturers and extend product development timelines. A core reason for this is the absence of well defined, globally harmonized frameworks that clearly delineate how microbiome products such as live biotherapeutic preparations, microbial consortia, or fecal microbiota components should be regulated. Different jurisdictions set varying criteria for what constitutes a food ingredient, a dietary supplement, a medical therapy, or a biological product, forcing manufacturers to engage separately with agencies like the U.S. FDA or the European Union (EU)’s regulatory bodies and adapt datasets and dossiers for each regulatory class and region.
Major regulatory updates are underway that add layers of complexity to compliance planning. For example, the EU’s Regulation on standards of quality and safety for substances of human origin (SoHO) adopted in 2024 and effective with transition through 2027 will extend oversight to human microbiome materials used in manufacturing and clinical applications, requiring additional quality and safety obligations beyond existing pharmaceutical legislation. These evolving requirements raise costs and risk for manufacturers, who must continuously monitor and respond to shifting regulatory boundaries, engage in extensive pre submission interactions with authorities, and maintain flexible development strategies that can accommodate distinct regional regulations.
Challenges in Maintaining Microbial Viability and Stability during Manufacturing
Microbial populations used in commercial biological products are inherently sensitive to physical and chemical stressors during production, transport, and storage. Cell membranes, proteins, and metabolic pathways of living microorganisms can be disrupted by common manufacturing conditions such as temperature shifts, dehydration, oxygen exposure, freezing and thawing cycles, and mechanical agitation. These stresses directly reduce the number of viable cells in a batch, which in turn can diminish the potency of the final product and its ability to deliver the intended biological effect. For example, freeze-thaw cycles at −80°C have been shown to decrease viability of microbes used in fecal microbiota transplant products when compared to fresh material, illustrating how process parameters influence survival rates in real-world settings.
Maintaining viability and stability also carries regulatory and quality implications. Government agencies such as the U.S. FDA closely examine how manufacturing processes impact the live microbial content of products that rely on biological activity for efficacy. If a production process substantially alters the viable microbial composition, the product may fail to meet regulatory expectations for quality and performance. Robust analytical methods are required to monitor live cell counts and stability in-process and post-production, and these methods are technically challenging to implement for complex microbial communities that vary by strain and growth characteristics.
Adoption of AI-Driven and Automated Manufacturing Platforms
AI and automated manufacturing platforms fundamentally re-engineer production processes through data-driven predictability and real-time optimization. These technologies convert raw process data into actionable decisions at speeds unattainable through manual intervention, improving yield consistency and throughput while reducing variability. Automated systems equipped with machine learning can continuously monitor equipment health and forecast maintenance needs, preventing unplanned downtime and maintaining steady operational flows essential for delicate biological workflows. For complex bioprocesses that are sensitive to environmental shifts and require tight control of growth conditions, this transformation results in more reliable product quality and reproducibility. Predictive algorithms also enhance resource utilization by minimizing waste of expensive inputs and ensuring quality compliance through automated inspection and corrective actions.
The operational agility provided by AI and automation aligns with broader industrial priorities that governments have articulated for the future of manufacturing. Official guidance from the National Institute of Standards and Technology (NIST) highlights that small manufacturers will increasingly deploy robots and automated systems to streamline repetitive tasks, improve accuracy, and speed up production in 2025 as a strategic response to labor and quality challenges. By embedding AI at the core of production platforms, organizations can scale biologics manufacturing with greater precision and lower cycle time, reduce dependence on specialized manual labor, and adapt rapidly to shifts in demand. This capability is particularly valuable in microbiome manufacturing, where scalability and regulatory compliance are key business imperatives.
Government Support Driving R&D and Advanced Bioprocessing Investments
Government commitment to research and development (R&D) and advanced bioprocessing investments strengthens innovation, accelerates technology maturation, and reduces commercial risk for manufacturers. Public funding and incentives lower the cost barriers for expensive early-stage research and pilot-scale production infrastructure, enabling organizations to explore complex biological systems and scale production processes that firms might otherwise defer due to uncertain returns. For example, Canada’s Strategic Innovation Fund allocated significant contributions to build a new biomanufacturing and R&D facility in 2025, supporting the development of cutting-edge therapeutics and vaccines and creating new skilled jobs. This type of government program signals long-term stability and commitment, which encourages private capital to co-invest in technologies that require extended development timelines and regulatory navigation.
State involvement aligns innovation agendas with broader social priorities such as health security, supply chain resilience, and economic competitiveness. By funding collaborative hubs and research networks, governments promote knowledge sharing across universities, startups, and established manufacturers, shortening the timeline from discovery to scalable production. Public backing helps address market inefficiencies that arise when early discoveries remain confined to laboratories due to cost barriers for scale-up. By enabling translation of foundational research into industrial application, government programs ensure emerging technologies progress through validation and commercialization phases, supporting both technological leadership and domestic production capacity that can serve global demand.
Category-wise Analysis
North America Microbiome Manufacturing Market Trends
By 2026, North America is expected to lead with an estimated 45% of the microbiome manufacturing share, supported by advanced healthcare infrastructure, high clinical awareness, and established biotechnology ecosystems. The United States and Canada host leading manufacturers, research institutions, and clinical trial networks that accelerate adoption of microbiome-based therapeutics. Extensive collaboration between academic centers, biotechnology companies, and contract manufacturing organizations enhances innovation across the product lifecycle. State and private funding initiatives support early-stage R&D, while clinical programs provide real-world validation of efficacy. This integrated ecosystem strengthens capacity to scale production and rapidly translate discoveries into commercial solutions.
Regulatory frameworks from the U.S. FDA provide clear guidance on biologics and microbial therapeutics, facilitating timely approval of novel formulations. Structured approval processes reduce uncertainty and risk for manufacturers, enabling faster market entry and higher confidence in compliance. Companies can leverage regulatory incentives for breakthrough therapies and orphan indications to streamline clinical validation. Strong intellectual property protections and guidance on quality control, sterility, and safety standards encourage investment in advanced manufacturing technologies. Combined with access to a skilled workforce and specialized facilities, regulatory clarity positions the market for scalable, high-quality microbiome production capable of meeting domestic and international demand.
Europe Microbiome Manufacturing Market Trends
Europe exhibits strong momentum in microbiome manufacturing, driven by scientific depth, advanced bioprocessing infrastructure, and collaborative innovation. Extensive academic research and clinical trial activity generate a steady pipeline of validated microbiome applications, enabling efficient translation from discovery to production. Established biotechnology networks support integration of novel microbial therapeutics into manufacturing processes, while specialized facilities with advanced fermentation, anaerobic processing, and cold-chain systems ensure product stability and quality. Investment in automation, digital monitoring, and quality assurance enhances production efficiency and consistency, allowing complex microbiome-based therapies to reach scalable manufacturing levels. Collaborative efforts between research institutions and industrial partners facilitate knowledge transfer, process optimization, and faster adoption of emerging technologies across the value chain.
Support mechanisms and multi-stakeholder initiatives strengthen commercial and operational capacity. Harmonized regulatory frameworks provide clear guidance on approvals, quality standards, and safety protocols, fostering confidence among manufacturers and investors. Publicly funded programs and innovation partnerships accelerate development of novel microbial modalities, promote technology transfer, and expand access to specialized manufacturing hubs. Industry alliances integrate research, policy, and production initiatives, enabling streamlined scale-up and cross-border collaboration. Adoption of advanced analytical tools, next-generation sequencing, and bioinformatics enhances process control and compliance, improving efficiency and reducing risk.
Asia Pacific Microbiome Manufacturing Market Trends
Asia Pacific is forecasted to be the fastest-growing market for microbiome manufacturing between 2026 and 2033, stimulated by rising healthcare infrastructure, growing clinical awareness, and government initiatives supporting biotechnology. Expanding clinical trial networks targeting gastrointestinal, metabolic, and immune-related conditions are generating robust data that informs product development and enhances regulatory confidence. Increasing funding for biotech startups and translational research supports development of specialized bioprocessing facilities, enabling rapid scale-up of microbial therapeutics. Rising adoption of preventive healthcare solutions and awareness of microbiome applications among populations further amplifies demand. Strategic partnerships between domestic companies and multinational manufacturers drive technology transfer and access to advanced bioprocessing platforms, accelerating commercialization and operational efficiency. Focused investment in automation, digital monitoring, and quality control ensures production aligns with international compliance standards while meeting growing throughput requirements.
Government-backed incentives and ecosystem initiatives underpin accelerated capacity expansion and competitive positioning. Regulatory frameworks have been streamlined to provide clarity on approvals for biologics and microbial products, reducing time-to-market risk and supporting large-scale production planning. Development of biotech clusters, incubators, and specialized manufacturing hubs attracts contract manufacturing collaborations that integrate global expertise with local capabilities. Workforce development through collaborations between industry and academic institutions addresses technical skill gaps, enabling adoption of cutting-edge fermentation, bioinformatics, and analytical technologies. Competitive operational costs, combined with high R&D intensity and infrastructure growth, attract further investment and optimize capacity utilization.
Competitive Landscape
The global microbiome manufacturing market exhibits a moderately consolidated structure, with large and very large companies holding leading positions while mid-sized and small players contribute through niche innovations. Key players such as Seres Therapeutics, Vedanta Biosciences, Ferring, MaaT Pharma, Enterome, and BIOSE INDUSTRIE maintain significant influence through advanced manufacturing infrastructure, strong clinical expertise, and strategic collaborations. These companies leverage robust production capacity, integrated bioprocess technologies, and quality-assured manufacturing practices to deliver scalable microbial therapeutics across multiple applications. Their extensive networks allow effective supply chain management and market reach, enhancing adoption among healthcare providers and clinical programs.
Fragmented competition exists among emerging manufacturers, which drives continuous innovation in process development, formulation, and quality control. Leading companies benefit from economies of scale, enabling cost efficiencies and rapid adoption of automation, digital monitoring, and process optimization technologies. Smaller firms differentiate through agility, faster development cycles, and specialized expertise, often collaborating with academic institutions or contract manufacturers to scale production. Partnerships between established players and niche innovators create a dynamic ecosystem where technological advances and clinical validation progress simultaneously.
Key Industry Developments
- In February 2026, Biomica and Lishan Biotech signed an exclusive worldwide licensing agreement for a microbiome-based cancer therapeutic BMC128, enabling Lishan Biotech to lead global development, manufacturing, and commercialization of the therapy.
- In January 2026, MaaT Pharma announced that the first patient had been randomized in the IMMUNOLIFE Phase 2 study, investigating whether modulation of the gut microbiome with MaaT033 could help overcome resistance to immune checkpoint inhibitors in advanced non-small cell lung cancer patients with antibiotic-induced dysbiosis.
- In October 2025, EnteroBiotix completed a new manufacturing facility dedicated to producing gut microbiome-based therapies, strengthening its ability to scale commercial production and support broader clinical development efforts in microbiome health. The expanded capacity is aimed at improving supply reliability for therapeutic products targeting chronic gastrointestinal conditions.
Companies Covered in Microbiome Manufacturing Market
- Seres Therapeutics
- Vedanta Biosciences, Inc. A
- Ferring.
- MaaT Pharma.
- Enterome
- BIOSE INDUSTRIE
- Finch Therapeutics Group, Inc.
- Prokarium
- Lallemand Inc.
Frequently Asked Questions
The global microbiome manufacturing market is projected to reach US$ 53.6 million in 2026.
Rising demand for microbiome-based therapeutics, increased R&D investment, and advanced bioprocessing technologies are driving the market.
The market is poised to witness a CAGR of 14.9% from 2026 to 2033.
Government support for R&D, expansion of advanced bioprocessing infrastructure, and strategic partnerships are creating key market opportunities.
Some of the key market players include Seres Therapeutics, Vedanta Biosciences, Inc., Ferring., MaaT Pharma., Enterome, and BIOSE INDUSTRIE.







