- Medical Devices
- Prefilled Syringes Market
Prefilled Syringes Market Size, Share, and Growth Forecast, 2026 - 2033
Prefilled Syringes Market by Product Type (Conventional Prefilled Syringes, Safety Prefilled Syringes), Material (Glass, Polymer), Application (Diabetes, Rheumatoid Arthritis, Oncology, Vaccination, Cardiovascular Diseases (CVDs)), and Regional Analysis for 2026 - 2033
Prefilled Syringes Market Share and Trends Analysis
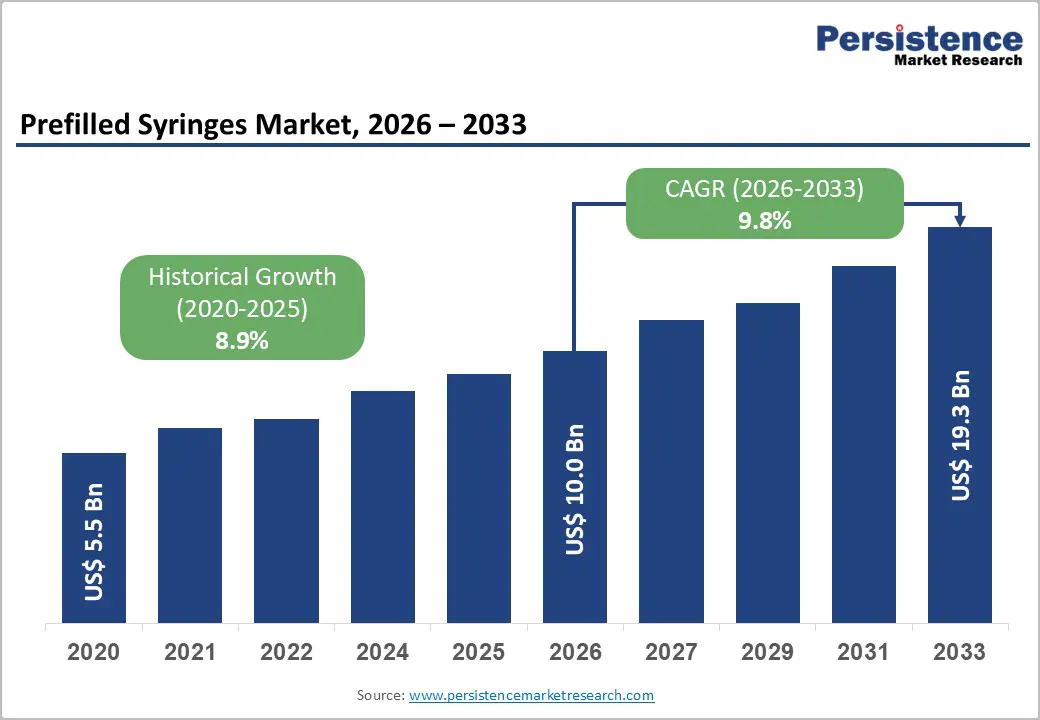
The global prefilled syringes market size is likely to be valued at US$ 10.0 billion in 2026, and is projected to reach US$ 19.3 billion by 2033, growing at a CAGR of 9.8% during the forecast period 2026–2033. The market is expanding steadily due to rising demand for biologic drugs, the growing prevalence of chronic diseases, and the increasing adoption of self-administered drug delivery systems. Biologic therapies such as monoclonal antibodies, hormones, and vaccines require accurate dosing and sterile delivery, making prefilled syringes a preferred option.
Pharmaceutical manufacturers increasingly adopt these systems to improve dosing accuracy, reduce medication preparation time, and minimize contamination risks. In addition, expanding global vaccination programs and the development of injectable treatments for diabetes, rheumatoid arthritis, and autoimmune diseases continue to support demand. Healthcare providers and regulatory authorities are also promoting ready-to-use injectable formulations to enhance patient safety and improve treatment efficiency across hospitals and home care settings.
Key Industry Highlights
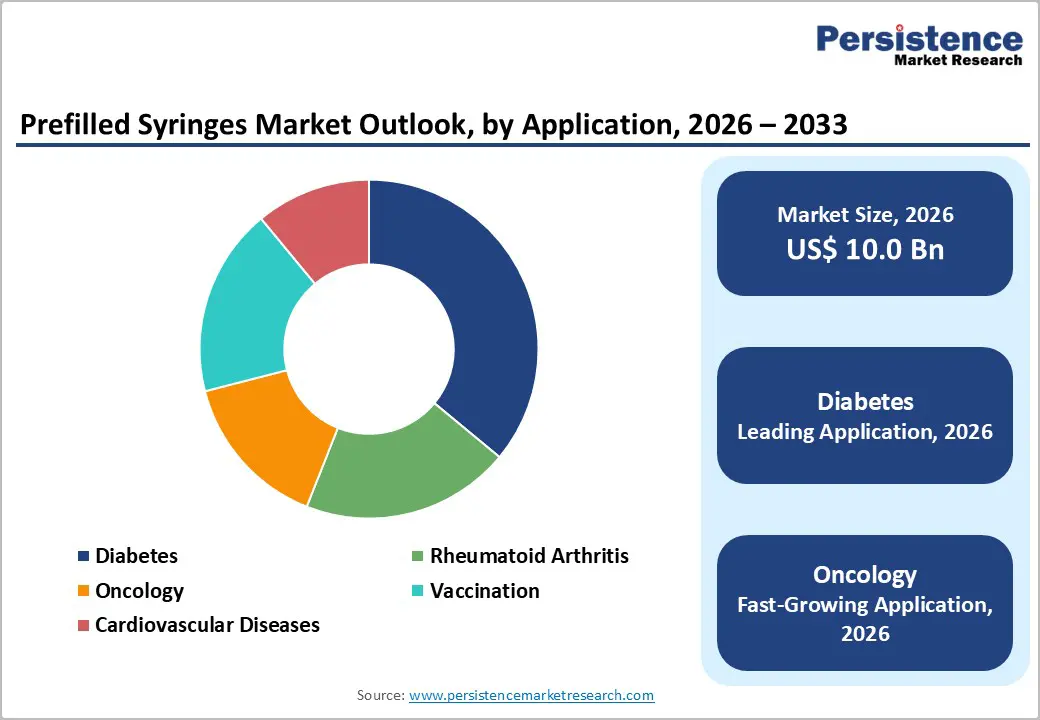
- Dominant Application: Diabetes is anticipated to hold roughly 36% revenue share in 2026, while oncology is likely to expand the fastest at 11.5% CAGR through 2033, supported by the rising adoption of injectable biologic cancer therapies.
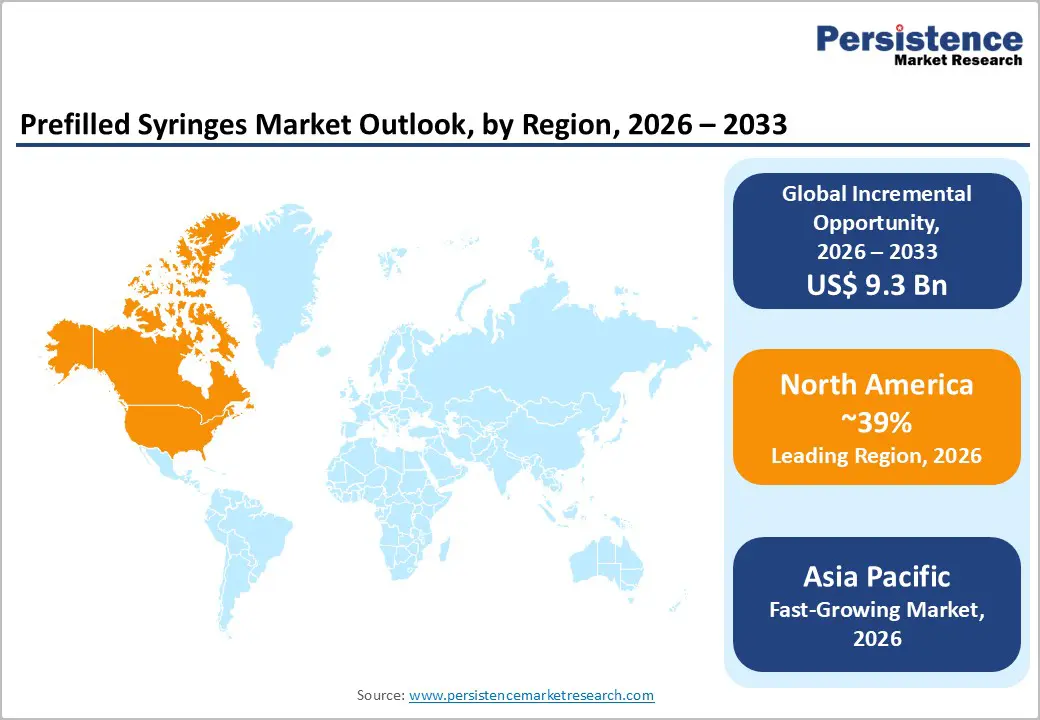
- Regional Leadership: North America is poised to dominate with an estimated 39% market share in 2026, while Asia Pacific is projected to register the fastest regional growth at 11.3% CAGR from 2026 to 2033, led by vaccine manufacturing expansion.
- Dominant Product Type: Conventional prefilled syringes are set to command around 64% revenue share in 2026, while safety prefilled syringes are likely to grow the fastest through 2033, as regulatory mandates increasingly emphasize needlestick injury prevention.
- Leading Material: Glass is expected to lead with about 71% share in 2026, while polymer prefilled syringes are projected to grow the fastest, driven by enhanced durability and improved compatibility with advanced biologic formulations.
- Competitive Environment: Market competition is driven by capacity expansions, partnerships with biologic drug manufacturers, and investments in advanced polymer syringe technologies.
| Key Insights | Details |
|---|---|
|
Prefilled Syringes Market Size (2026E) |
US$ 10.0 Bn |
|
Market Value Forecast (2033F) |
US$ 19.3 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
9.8% |
|
Historical Market Growth (CAGR 2020 to 2025) |
8.9% |
DRO Analysis
Rising Prevalence of Chronic Diseases and Increasing Injectable Therapies
The global rise in chronic diseases is a primary driver of the prefilled syringes market. According to the World Health Organization (WHO), chronic diseases account for approximately 74% of all global deaths, with conditions such as diabetes, cardiovascular diseases, and autoimmune disorders requiring long-term injectable treatment. The International Diabetes Federation (IDF) reported that 537 million adults were living with diabetes in 2021, projected to reach 643 million by 2030, increasing the demand for insulin delivery systems. These conditions require frequent and accurate drug administration, making reliable drug delivery devices essential in modern healthcare.
Prefilled syringes simplify drug administration by providing accurate dosing and improved patient convenience, particularly for insulin, biologics, and monoclonal antibodies. The shift toward home-based care and self-injection therapies further supports demand as healthcare systems aim to reduce hospital visits and treatment costs. These devices also reduce preparation steps for healthcare professionals, improving treatment efficiency. Reflecting this trend, Becton, Dickinson and Company announced a US$110 million investment in 2026 to expand production of glass prefillable syringes in the United States, strengthening supply chains for biologic and GLP-1 injectable therapies and supporting the growing global demand for advanced drug delivery systems.
Expansion of Biologic Drugs and Global Vaccination Programs
The growing biologics market significantly drives the adoption of prefilled syringes. According to the U.S. Food and Drug Administration (FDA), biologics represent over 40% of newly approved drugs in recent years, many of which require injectable delivery systems. Biologic drugs such as monoclonal antibodies, vaccines, and hormone therapies require precise dosing and protection from contamination, making prefilled syringes a preferred delivery format. Pharmaceutical manufacturers are increasingly integrating these devices into drug-device combination products to enhance usability and ensure consistent drug administration.
Global vaccination initiatives have also increased the demand for ready-to-use injection systems. According to UNICEF and Gavi, the Vaccine Alliance, more than 5 billion vaccine doses are administered worldwide each year, requiring safe and efficient delivery methods. Industry developments further highlight this trend. In 2025, the U.S. FDA approved a new injectable formulation of Merck’s cancer therapy Keytruda, enabling faster subcutaneous administration than the traditional intravenous infusion, supporting the broader shift toward injectable drug delivery formats. Additionally, pharmaceutical manufacturers are investing in syringe production capacity and advanced fill-finish technologies to meet growing demand for vaccines and biologics, underscoring the importance of prefilled syringes in modern immunization and therapeutic delivery systems.
High Manufacturing and Material Costs
Prefilled syringes require specialized manufacturing processes, including high-precision filling technology, sterile packaging, and compatibility testing, which increases production costs compared with traditional vial-and-syringe systems. Glass syringes must meet stringent pharmaceutical-grade standards and sterilization requirements, further increasing manufacturing complexity. These requirements demand advanced production infrastructure and strict quality control procedures. As a result, manufacturers must invest heavily in automated filling systems, cleanroom environments, and specialized inspection technologies. The high level of technical precision involved in syringe manufacturing significantly raises operational expenses for pharmaceutical packaging companies.
In 2025, supply chain pressures and rising raw material costs continued to affect pharmaceutical packaging manufacturers. The cost of borosilicate glass and medical-grade polymers increased due to energy price fluctuations and logistics constraints, affecting syringe production costs. In response to rising demand and supply challenges, specialty glass manufacturer SCHOTT expanded its syringe glass tubing production in India in 2025 to strengthen pharmaceutical packaging supply chains and address shortages in high-precision syringe materials. Smaller pharmaceutical manufacturers may therefore face financial barriers in transitioning to prefilled syringe formats, particularly for low-volume injectable drugs, limiting adoption among emerging pharmaceutical firms.
Regulatory Complexity and Drug-Device Compatibility Challenges
Prefilled syringes are regulated as combination drug-device products, requiring compliance with both pharmaceutical and medical device regulations. Agencies such as the U.S. FDA and the European Medicines Agency (EMA) require extensive testing to ensure drug stability, material compatibility, and sterility. Manufacturers must conduct detailed validation studies covering container closure integrity, extractables and leachables testing, and product stability. These regulatory requirements extend development timelines and require significant documentation and compliance efforts before product approval.
Compatibility issues between biologic drugs and syringe materials, such as silicone oil interaction, protein aggregation, and leachables from polymer components, can delay product approvals. Certain biologic formulations are highly sensitive to container materials, making packaging selection a critical step in drug development. Recent industry observations have highlighted issues such as glass delamination in high-pH biologic drugs, which can release microscopic particles into injectable medicines and trigger product recalls if compatibility testing fails. As a result, pharmaceutical companies often need to redesign syringe materials or conduct additional stability studies, increasing development timelines and regulatory complexity.
Expansion of Self-Injection Therapies and Home Healthcare
The growing preference for home-based treatment and self-administration of injectable drugs creates a major opportunity for the prefilled syringes market. According to the Organization for Economic Co-operation and Development (OECD), healthcare systems are increasingly shifting chronic disease management from hospitals to outpatient and home settings to reduce treatment costs. Prefilled syringes allow patients to safely administer medications without complex preparation, ensuring accurate dosing and convenience for therapies such as insulin, monoclonal antibodies, and hormone treatments. They also help reduce the burden on healthcare professionals by minimizing time spent on preparation and administration.
In 2025, pharmaceutical companies expanded self-injection formats for biologic therapies targeting rheumatoid arthritis, psoriasis, and multiple sclerosis. For example, the U.S. FDA approved a new subcutaneous version of Roche’s Actemra, allowing home administration of the therapy, supporting the trend toward patient-centered care. The global home healthcare market is projected to exceed US$ 600 billion by 2030, underscoring strong demand for user-friendly drug-delivery devices. Prefilled syringes facilitate self-administration, reduce hospital visits, improve adherence, enhance patient experience, and empower patients to manage chronic conditions more independently.
Emerging Markets and Technological Innovation in Syringes
Rapid healthcare expansion in emerging economies presents a significant growth opportunity for prefilled syringes. Countries in the Asia-Pacific, Latin America, and the Middle East are investing heavily in healthcare infrastructure and pharmaceutical manufacturing. In 2025, India’s Department of Biotechnology (DBT) announced funding to expand domestic vaccine fill-and-finish capacity, while China accelerated investments in local biologics production to strengthen public health programs. These initiatives have increased demand for high-volume syringe packaging solutions and ready-to-use injectables, helping emerging markets catch up with advanced healthcare systems.
Technological innovation in syringe design further enhances market potential. In 2025, several manufacturers launched advanced polymer and safety syringe platforms supporting high-viscosity biologic drugs, large-volume injections, and needle-safety mechanisms. For example, BD introduced a new self-injection pen with an integrated safety shield, enabling safer home administration of biologics. These innovations reduce drug interaction risks, improve break resistance, enhance patient and healthcare worker safety, and support broader adoption of injectable therapies across hospitals, clinics, and home care, opening new revenue streams for manufacturers.
Category-wise Analysis
Product type Insights
Conventional prefilled syringes currently hold the largest market revenue share at roughly 64% in 2026, driven by widespread use in hospitals, vaccination programs, and large-volume pharmaceutical applications. They are commonly used for insulin, vaccines, anticoagulants, and other injectable drugs, offering reliable dosing, workflow efficiency, and cost-effectiveness. Their established regulatory acceptance makes them the preferred choice for large-scale public health campaigns. For example, in March 2025, the U.S. CDC distributed conventional prefilled syringes nationwide as part of the expanded adult immunization schedule, including influenza and tetanus boosters, reducing preparation time and contamination risks during peak operations.
Safety prefilled syringes are expected to record the fastest growth, with a 10.6% CAGR from 2026 to 2033, driven by regulatory emphasis on preventing needlestick injuries among healthcare workers. These syringes feature automatic needle shielding or retractable mechanisms that activate immediately after injection, minimizing occupational hazards and reducing bloodborne pathogen exposure. In October 2025, the New York State Department of Health mandated the use of safety prefilled syringes for anticoagulant and biologic injections in state hospitals, aligning with OSHA guidelines. This policy adoption accelerated the deployment of safety syringes in institutional settings and underscores the regulatory drivers behind their market growth.
Application Insights
Diabetes is set to remain the largest application, holding around 36% of the prefilled syringes market revenue share in 2026, due to the global prevalence of insulin-dependent patients and reliance on injectable therapies. Prefilled insulin syringes improve dosing accuracy, reduce preparation errors, and support chronic disease management, particularly in home and outpatient settings. In November 2025, the U.S. Centers for Medicare & Medicaid Services (CMS) revised coverage policies to improve access to prefilled insulin delivery devices, enabling broader use among Medicare beneficiaries. This policy change reinforced the adoption of prefilled syringes in diabetes care, improving patient outcomes and treatment adherence.
Oncology is projected to be the fastest-growing application, with an 11.5% CAGR from 2026 to 2033, driven by the rise of injectable cancer therapies and monoclonal antibody treatments that require precise, sterile delivery. Prefilled syringes support subcutaneous administration outside traditional infusion centers, improving patient convenience and hospital efficiency. In July 2025, Dana-Farber Cancer Institute implemented prefilled subcutaneous administration protocols for specific outpatient biologic therapies, reducing infusion times and increasing treatment capacity. This development highlights how oncology adoption of prefilled syringes enhances care efficiency while supporting patient-centric treatment models.
Regional Insights
North America Prefilled Syringes Market Trends
North America is expected to dominate with an estimated 39% of the prefilled syringes market share in 2026, supported by strong pharmaceutical manufacturing capabilities and early adoption of advanced drug delivery systems. The region benefits from robust regulatory oversight by the U.S. FDA, which emphasizes safety, sterility, and self-administration options for combination drug-device products. Chronic disease prevalence, including diabetes and autoimmune conditions, continues to drive demand for reliable, user-friendly injectable systems that support outpatient care and home-based therapies.
A notable industry development in April 2025 saw the FDA approve the prefilled syringe version of argenx’s VYVGART Hytrulo for self-injection, allowing patients with generalized myasthenia gravis and chronic inflammatory demyelinating polyneuropathy (CIDP) to administer subcutaneous treatment at home. This regulatory action reflects broader trends toward patient empowerment through self-injectable formats and convenience-oriented care pathways. Regional innovation ecosystems, combined with high healthcare expenditure and established biologics pipelines, continue to strengthen North America’s leadership position in the global market.
Europe Prefilled Syringes Market Trends
Europe ranks as the second-largest regional market, with strong pharmaceutical manufacturing infrastructure and harmonized regulatory frameworks across EU member states facilitating market access for prefilled syringes. Countries such as Germany, the United Kingdom, France, and Spain play pivotal roles in regional demand, supported by advanced biotechnology research, vaccination infrastructure, and integrated healthcare systems. The EMA also ensures stringent quality, safety, and performance standards for drug-device combination products, encouraging adoption of ready-to-use injectable delivery formats.
A key 2025 development in Europe was SGD Pharma’s acquisition of Alphial S.r.l. in October 2025, expanding capacity in tubular glass products and ready-to-use packaging formats, including vials and syringe components, to better serve pharmaceutical manufacturers. This acquisition enhances European capabilities for high-precision glass packaging, which is essential for maintaining stability and sterility in injectable drugs. The region is also seeing sustainability-oriented investments in cleaner manufacturing sites and energy-efficient glass production, supporting long-term competitiveness in biologics and vaccine delivery.
Asia Pacific Prefilled Syringes Market Trends
Asia Pacific is expected to register the fastest regional growth, with an estimated CAGR of 11.3% through 2033, driven by rapid expansion of healthcare infrastructure, rising pharmaceutical manufacturing, and supportive government healthcare initiatives. China, Japan, and India are key contributors: China continues scaling vaccine and biologic drug output, Japan leads in advanced pharmaceutical technology adoption, and India plays a significant role in large-scale vaccine production and generic injectable therapies. These trends are increasing demand for automated fill-finish and packaging solutions tailored to prefilled syringes.
In April 2025, the U.S. FDA’s approval of a self-injectable prefilled syringe for a chronic disease therapeutic highlighted broader global interest in self-administration formats; although a U.S. example, this success influences the Asia Pacific market through regulatory harmonization and international product rollout strategies. Furthermore, governments across the region have been accelerating mass immunization programs and public healthcare investments, prioritizing operational simplicity and vaccine access, which heighten preference for prefilled syringe presentations to streamline point-of-care delivery. These policy and production shifts position Asia Pacific for rapid adoption of advanced injectable technologies.
Competitive Landscape
The global prefilled syringes market structure is moderately consolidated, with leading players such as BD (Becton Dickinson), Gerresheimer, Terumo, West Pharmaceutical Services, and SCHOTT AG collectively controlling over half of the global revenue share. These established companies leverage extensive relationships with pharmaceutical manufacturers, strong regulatory compliance expertise, and vertically integrated production capabilities spanning glass, polymer, and combination drug-device systems. They continue to invest heavily in R&D to maintain technological leadership in safety syringe mechanisms, polymer innovations, high-viscosity biologic delivery, and automated fill-finish solutions.
Regional and niche competitors, including Nipro Corporation, Ypsomed, and NABI, focus on specialized product segments and geographic strongholds, often targeting emerging markets or specific biologic therapies. Barriers such as stringent regulatory approvals, complex drug-device compatibility testing, and high production costs limit new entrants, yet digitalization trends and modular syringe platforms are enabling smaller or software-enabled firms to participate in tracking, traceability, and connected delivery systems. Market consolidation is expected to increase gradually as global leaders acquire smaller vendors to expand geographically and technologically, while strategic partnerships with biotech companies foster innovation in advanced injectable therapies.
Key Industry Developments
- In August 2025, BD invested over US$ 35 million at its Nebraska facility to expand prefilled flush syringe manufacturing, enhancing capacity to support U.S. healthcare systems and ensuring timely supply for hospitals and outpatient care. This expansion strengthens BD’s position in high-demand injectable delivery devices.
- In June 2025, Pii completed a US$ 3.6 million upgrade of its automated fill and inspection lines for prefilled syringes, increasing production efficiency and reliability for pharmaceutical partners. The enhancement addresses growing demand for ready-to-use injectable therapies.
- In April 2025, the U.S. FDA approved a prefilled syringe version of Argenx’s Vyvgart for patients with generalized myasthenia gravis and CIDP, enabling safe at-home self-administration. This regulatory milestone supports patient convenience and expands the adoption of self-injection therapies.
Companies Covered in Prefilled Syringes Market
- Becton, Dickinson and Company
- Gerresheimer AG
- SCHOTT Pharma
- West Pharmaceutical Services Inc.
- Nipro Corporation
- Terumo Corporation
- Stevanato Group
- Baxter International Inc.
- Catalent Inc.
- Weigao Group
- Ypsomed AG
- Medtronic plc
- Daikyo Seiko Ltd.
- AptarGroup Inc.
Frequently Asked Questions
The global prefilled syringes market is projected to reach US$ 10.0 billion in 2026.
Rising demand for biologic drugs, chronic disease prevalence, and growing adoption of self-injection therapies are key market drivers.
The market is poised to witness a CAGR of 9.8% from 2026 to 2033.
Expansion of self-injection therapies, home healthcare, and emerging market healthcare infrastructure present major opportunities.
BD, Gerresheimer, Terumo, West Pharmaceutical Services, and SCHOTT AG are some of the leading market companies.








