- Executive Summary
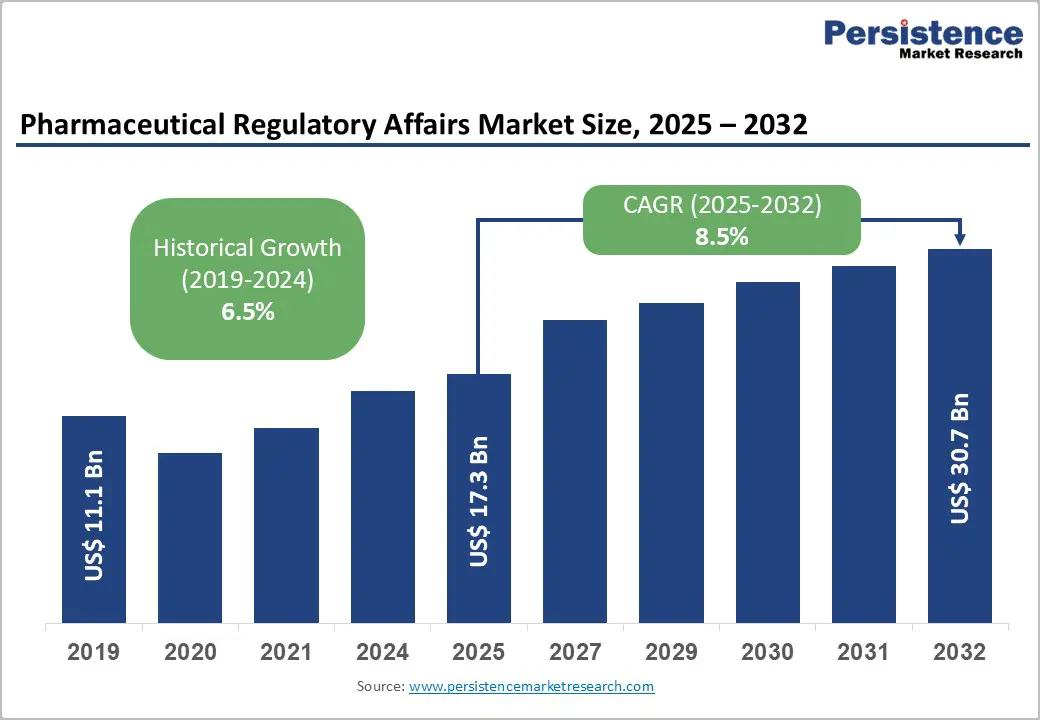
- Global Pharmaceutical Regulatory Affairs Market Snapshot, 2025 and 2032
- Market Opportunity Assessment, 2025 - 2032, US$ Bn
- Key Market Trends
- Future Market Projections
- Premium Market Insights
- Product Developments and Key Market Events
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Key Trends
- Macro-economic Factors
- Global Sectoral Outlook
- Global GDP Growth Outlook
- COVID-19 Impact Analysis
- Forecast Factors - Relevance and Impact
- Value Added Insights
- Tool Adoption Analysis
- Regulatory Landscape
- Value Chain Analysis
- PESTLE Analysis
- Porter’s Five Force Analysis
- Price Analysis, 2024A
- Key Highlights
- Key Factors Impacting Deployment Costs
- Pricing Analysis, By Service Type
- Global Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Market Volume (Units) Projections
- Market Size (US$ Bn) and Y-o-Y Growth
- Absolute $ Opportunity
- Market Size (US$ Bn) and Volume (Units) Analysis and Forecast
- Historical Market Size (US$ Bn) Analysis, 2019-2024
- Current Market Size (US$ Bn) Analysis and Forecast, 2025 - 2032
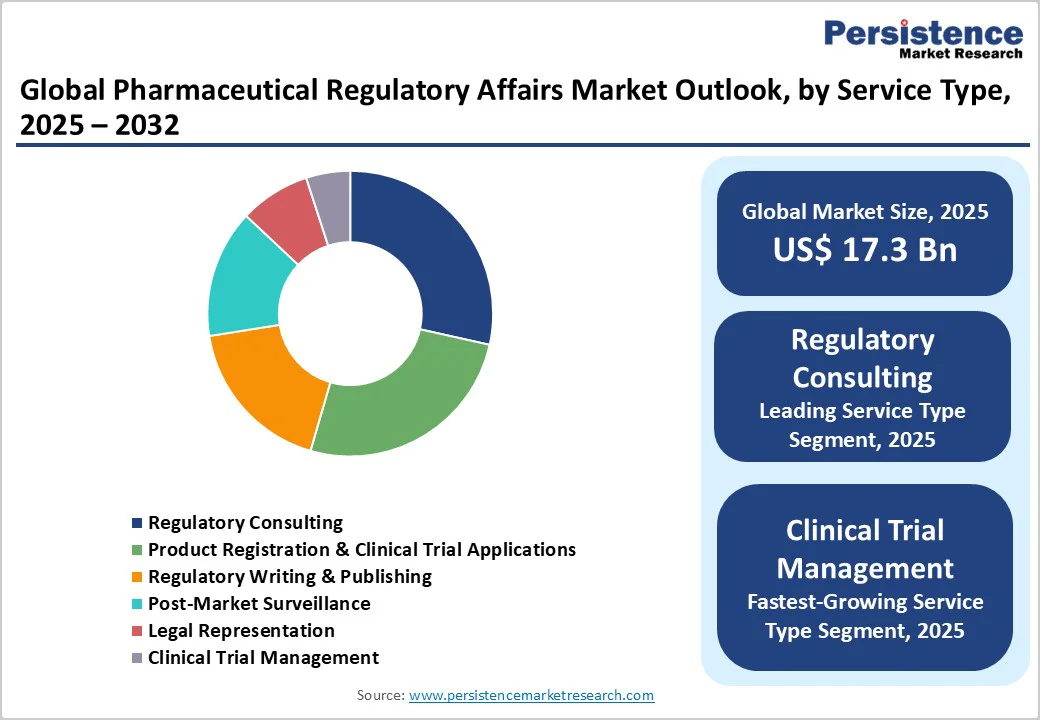
- Global Pharmaceutical Regulatory Affairs Market Outlook: Service Type
- Introduction / Key Findings
- Historical Market Size (US$ Bn) and Volume (Units) Analysis, By Service Type, 2019 - 2024
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Market Attractiveness Analysis: Service Type
- Global Pharmaceutical Regulatory Affairs Market Outlook: Product
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By Product, 2019 - 2024
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Market Attractiveness Analysis: Product
- Global Pharmaceutical Regulatory Affairs Market Outlook: Development Stage
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By Development Stage, 2019 - 2024
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025 - 2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis: Development Stage
- Key Highlights
- Global Pharmaceutical Regulatory Affairs Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Bn) and Volume (Units) Analysis, By Region, 2019 - 2024
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Region, 2025 - 2032
- North America
- Europe
- East Asia
- South Asia and Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2019 - 2024
- By Country
- By Service Type
- By Product
- By Development Stage
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2025 - 2032
- U.S.
- Canada
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025-2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis
- Europe Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2019 - 2024
- By Country
- By Service Type
- By Product
- Development Stage
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2025 - 2032
- Germany
- France
- U.K.
- Italy
- Spain
- Russia
- Türkiye
- Rest of Europe
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025-2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis
- East Asia Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2019 - 2024
- By Country
- By Service Type
- By Product
- By Development Stage
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2025 - 2032
- China
- Japan
- South Korea
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025-2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis
- South Asia & Oceania Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2019 - 2024
- By Country
- By Service Type
- By Product
- By Development Stage
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2025 - 2032
- India
- Southeast Asia
- ANZ
- Rest of South Asia & Oceania
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025-2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis
- Latin America Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2019 - 2024
- By Country
- By Service Type
- By Product
- By Development Stage
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2025 - 2032
- Brazil
- Mexico
- Rest of Latin America
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025-2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis
- Middle East & Africa Pharmaceutical Regulatory Affairs Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2019 - 2024
- By Country
- By Service Type
- By Product
- By Development Stage
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2025 - 2032
- GCC Countries
- Egypt
- South Africa
- Northern Africa
- Rest of Middle East & Africa
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Service Type, 2025 - 2032
- Regulatory Consulting
- Legal Representation
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Applications
- Post-Market Surveillance
- Clinical Trial Management
- Current Market Size (US$ Bn) Analysis and Forecast, By Product, 2025 - 2032
- Drugs
- Biologics
- Medical Devices
- Current Market Size (US$ Bn) Analysis and Forecast, By Development Stage, 2025-2032
- Pre-Clinical
- Clinical Trials
- Post-Approval/Post-Market Authorization (PMA)
- Market Attractiveness Analysis
- Competition Landscape
- Market Share Analysis, 2024
- Market Structure
- Competition Intensity Mapping By Market
- Competition Dashboard
- Company Profiles (Details - Overview, Financials, Strategy, Recent Developments)
- IQVIA Holdings Inc.
- Overview
- Segments and Deployments
- Key Financials
- Market Developments
- Market Strategy
- Parexel International Corporation
- ICON plc
- Labcorp Drug Development
- Syneos Health, Inc.
- WuXi AppTec
- Charles River Laboratories International, Inc.
- PRA Health Sciences
- Medpace Holdings, Inc.
- Freyr Solutions
- Pharmalex GmbH
- Genpact Limited
- Pharmexon
- Criterium, Inc.
- Clarivate Analytics
- IQVIA Holdings Inc.
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations

Loading page data
Please wait a moment