- Executive Summary
- Global Pancreatic Elastase Testing Market Snapshot, 2026 and 2033
- Market Opportunity Assessment, 2026 - 2033, US$ Bn
- Key Market Trends
- Future Market Projections
- Premium Market Insights
- Industry Developments and Key Market Events
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Key Trends
- Macro-Economic Factors
- Global Sectorial Outlook
- Global GDP Growth Outlook
- COVID-19 Impact Analysis
- Forecast Factors - Relevance and Impact
- Value Added Insights
- Product Adoption Analysis
- Recent Product Launches
- Regulatory Landscape
- Value Chain Analysis
- Key Deals and Mergers
- PESTLE Analysis
- Porter’s Five Force Analysis
- Price Trend Analysis, 2020 - 2033
- Key Highlights
- Key Factors Impacting Technology Prices
- Pricing Analysis, By Technology Type
- Regional Prices and Technology Preferences
- Global Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Market Size (US$ Bn) and Y-o-Y Growth
- Absolute $ Opportunity
- Market Size (US$ Bn) Analysis and Forecast
- Historical Market Size (US$ Bn) Analysis, 2020-2025
- Market Size (US$ Bn) Analysis and Forecast, 2026-2033
- Global Pancreatic Elastase Testing Market Outlook: Product
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis and Volume (Units) Analysis, By Product, 2020 - 2025
- Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Attractiveness Analysis: Product
- Global Pancreatic Elastase Testing Market Outlook: Indication
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By Indication, 2020 - 2025
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Attractiveness Analysis: Indication
- Global Pancreatic Elastase Testing Market Outlook: Application
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By Application, 2020 - 2025
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Research & Clinical Studies
- Market Attractiveness Analysis: Application
- Global Pancreatic Elastase Testing Market Outlook: End User
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By End User, 2020 - 2025
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis: End User
- Key Highlights
- Global Pancreatic Elastase Testing Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Region, 2020 - 2025
- Market Size (US$ Bn) Analysis and Forecast, By Region, 2026 - 2033
- North America
- Europe
- East Asia
- South Asia and Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Product
- By Indication
- By Application
- By End User
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- U.S.
- Canada
- 1.1. Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Pyelonephritis
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis
- Europe Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Product
- By Indication
- By Application
- By End User
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- Germany
- France
- U.K.
- Italy
- Spain
- Russia
- Rest of Europe
- 1.1. Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Pyelonephritis
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis
- East Asia Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Product
- By Indication
- By Application
- By End User
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- China
- Japan
- South Korea
- 1.1. Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Pyelonephritis
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis
- South Asia & Oceania Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Product
- By Indication
- By Application
- By End User
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- India
- Southeast Asia
- ANZ
- Rest of South Asia & Oceania
- 1.1. Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Pyelonephritis
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis
- Latin America Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Product
- By Indication
- By Application
- By End User
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- Brazil
- Mexico
- Rest of Latin America
- 1.1. Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Pyelonephritis
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis
- Middle East & Africa Pancreatic Elastase Testing Market Outlook:
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Product
- By Indication
- By Application
- By End User
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- GCC Countries
- Egypt
- South Africa
- Northern Africa
- Rest of Middle East & Africa
- 1.1. Market Size (US$ Bn) Analysis and Volume (Units) Analysis and Forecast, By Product, 2026 - 2033
- Immunoassay kits
- ELISA Kits
- Chemiluminescence Immunoassay Kits
- Fluorescence Immunoassay Kits
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Indication, 2026 - 2033
- Chronic Pancreatitis
- Type 1 Diabetes
- Cystic Fibrosis
- Others
- Market Size (US$ Bn) Analysis and Forecast, By Application, 2026 - 2033
- Diagnosis of Pancreatic Disorders
- Monitoring Pancreatic Function
- Pyelonephritis
- Market Size (US$ Bn) Analysis and Forecast, By End User, 2026 - 2033
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Specialty Gastroenterology Clinics
- Academic and Research Institutes
- Market Attractiveness Analysis
- Competition Landscape
- Market Structure
- Competition Intensity Mapping By Market
- Competition Dashboard
- Company Profiles (Details - Overview, Recent Developments)
- DiaSorin S.p.A.
- Overview
- Segments and Product & End User
- Key Financials
- Market Developments
- Market Strategy
- ScheBo Biotech AG
- Immundiagnostik AG
- Invivo Healthcare Ltd
- DRG Instruments GmbH
- Verisana GmbH
- BÜHLMANN Laboratories AG
- CerTest Biotec S.L.
- R-Biopharm AG
- American Laboratory Products Company, Ltd. (ALPCO)
- Vitassay Healthcare, S.L.U.
- Eurospital S.p.A.
- Bioserv Diagnostics GmbH
- Eagle Biosciences, Inc.
- Alpha Laboratories Ltd
- Others
- DiaSorin S.p.A.
- Market Structure
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations
- Medical Devices
- Pancreatic Elastase Testing Market
Pancreatic Elastase Testing Market Size, Share, Growth, and Regional Forecast, 2026 to 2033
Pancreatic Elastase Testing Market by Product (Immunoassay kits, ELISA Kits, Chemiluminescence Immunoassay Kits, Fluorescence Immunoassay Kits, and Others), by Indication (Chronic Pancreatitis, Type 1 Diabetes, Cystic Fibrosis, and Others), Application (Diagnosis of Pancreatic Disorders, Monitoring Pancreatic Function, and Research & Clinical Studies) End User (Hospitals, Diagnostic Laboratories, Specialty Gastroenterology Clinics, and Academic and Research Institutes), and Regional Analysis from 2026 to 2033
Key Industry Highlights:
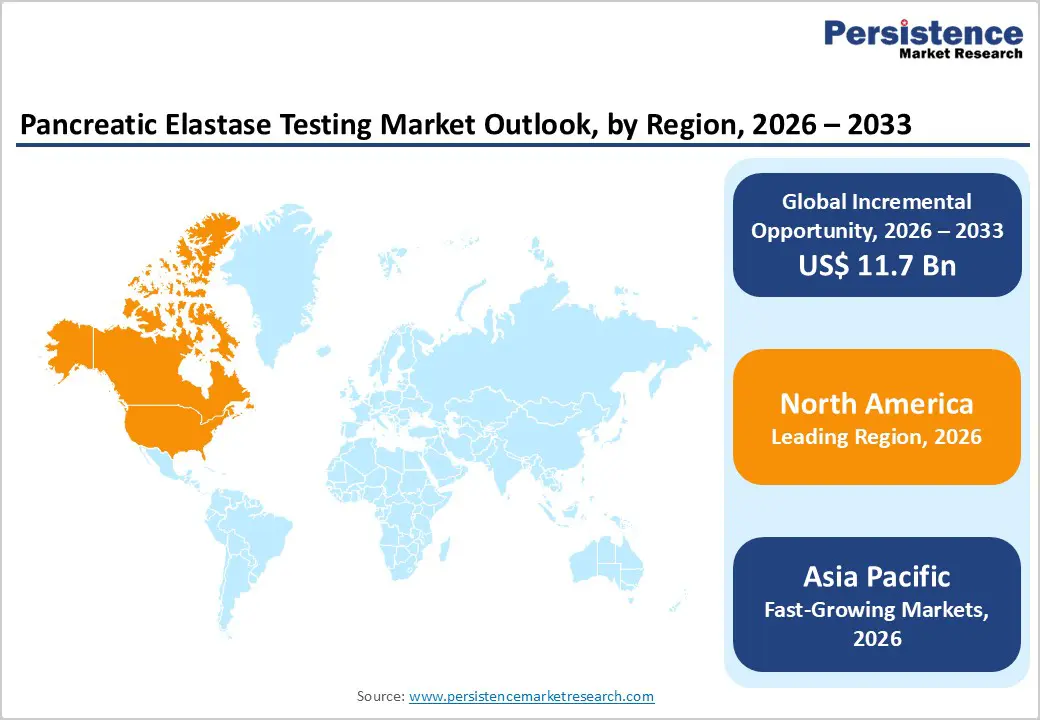
- Leading Region: North America accounts for 48.5% of global revenue, supported by sophisticated diagnostic laboratory networks, strong healthcare expenditure, advanced clinical testing infrastructure, and the presence of several established diagnostic technology providers.
- Fastest-Growing Region: Asia Pacific is experiencing the fastest market growth, driven by expanding healthcare infrastructure, increasing investment in hospital and laboratory development, rising clinical awareness of pancreatic disorders, and a large patient base requiring gastrointestinal diagnostics.
- Leading Product Segment: Immunoassay kits hold the leading share as they are widely utilized in hospitals and reference laboratories to quantify fecal pancreatic elastase levels for evaluating pancreatic exocrine function.
- Fastest-Growing Product Segment: ELISA kits are expanding at a notable pace due to their strong analytical reliability, compatibility with automated laboratory systems, and increasing adoption in clinical diagnostic workflows.
- Leading Indication Segment: Chronic pancreatitis represents the largest share of testing demand since pancreatic elastase measurement is frequently used to assess enzyme deficiency and confirm exocrine pancreatic insufficiency in affected patients.
- Fastest-Growing Indication Segment: Type 1 diabetes is emerging as a high-growth segment because individuals with long-standing diabetes often develop pancreatic enzyme dysfunction, prompting greater use of elastase testing for early detection of digestive complications.
| Key Insights | Details |
|---|---|
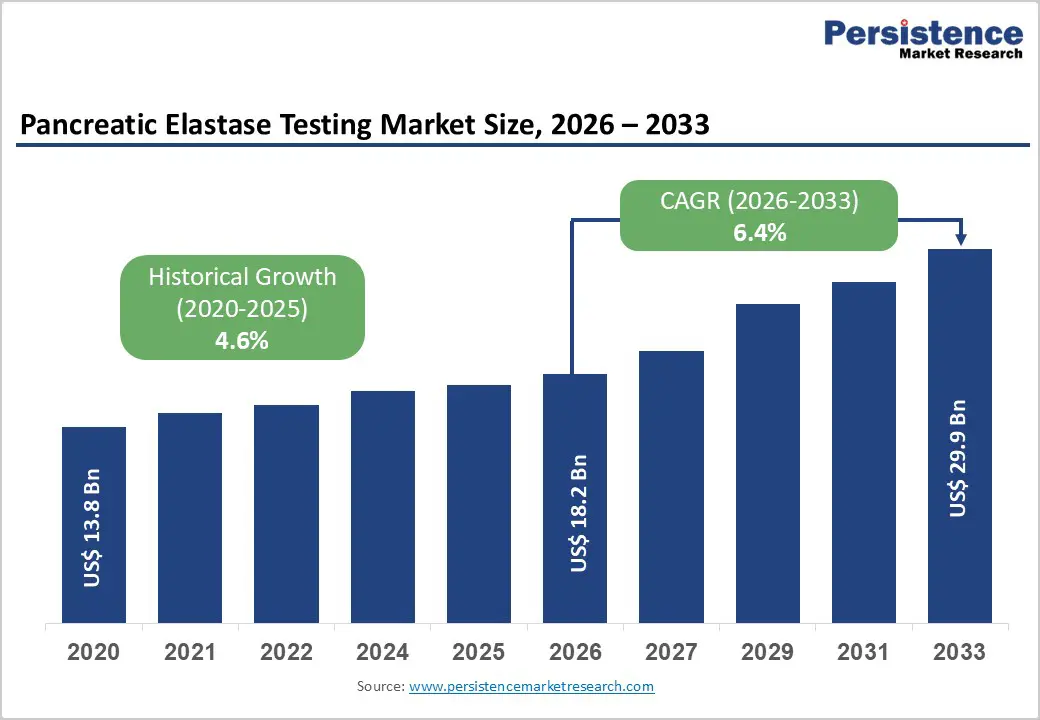
| Pancreatic Elastase Testing Market Size (2026E) | US$ 18.2 Bn |
| Market Value Forecast (2033F) | US$ 29.9 Bn |
| Projected Growth (CAGR 2026 to 2033) | 6.4% |
| Historical Market Growth (CAGR 2020 to 2025) | 4.6% |
Market Dynamics
Driver - Growing Incidence of Pancreatic Disorders and Rising Need for Non-Invasive Functional Diagnostics
The increasing prevalence of pancreatic disorders is playing a significant role in strengthening demand for pancreatic function testing worldwide. Conditions such as chronic pancreatitis, cystic fibrosis, pancreatic cancer, and long-standing diabetes frequently impair the pancreas’ ability to produce digestive enzymes, leading to exocrine pancreatic insufficiency. Patients experiencing persistent gastrointestinal symptoms including steatorrhea, diarrhea, malabsorption, and unexplained weight loss often require diagnostic evaluation of pancreatic enzyme activity. As clinical awareness of pancreatic insufficiency improves among gastroenterologists and primary care physicians, the use of fecal pancreatic elastase testing has expanded as a reliable method for evaluating pancreatic function.
Additionally, accelerating demand is the growing preference for non-invasive diagnostic approaches in gastrointestinal disease management. Fecal elastase testing provides a convenient and patient-friendly alternative to invasive pancreatic function assessments that require complex procedures. Hospitals and diagnostic laboratories increasingly utilize immunoassay-based elastase tests because they deliver accurate quantitative measurement of enzyme activity while supporting standardized laboratory workflows. With the rising number of patients undergoing evaluation for chronic digestive disorders and malabsorption syndromes, pancreatic elastase testing is becoming an important diagnostic tool in gastroenterology practice, supporting continued market expansion.
Restraints - Limited Clinical Awareness and Operational Challenges in Specialized Diagnostic Testing
Although pancreatic elastase testing offers valuable insights into pancreatic function, limited awareness regarding exocrine pancreatic insufficiency remains a key barrier in some healthcare systems. Digestive symptoms associated with pancreatic insufficiency are often nonspecific and may overlap with other gastrointestinal conditions such as irritable bowel syndrome or inflammatory bowel diseases. As a result, physicians may initially pursue alternative diagnostic pathways before considering pancreatic enzyme testing. In healthcare environments where awareness of pancreatic insufficiency screening remains relatively low, the number of elastase tests performed may remain limited despite the clinical benefits of early detection.
Laboratory operational factors can also influence testing adoption across different regions. Performing pancreatic elastase analysis requires immunoassay-based diagnostic kits, standardized laboratory protocols, and skilled laboratory personnel to ensure reliable sample processing and interpretation. Smaller clinics and healthcare facilities with limited laboratory infrastructure may rely primarily on general gastrointestinal testing rather than investing in specialized pancreatic diagnostics. Additionally, variations in stool sample consistency can occasionally affect test accuracy, requiring repeat testing or complementary diagnostic evaluation. These practical challenges, along with cost considerations and differences in diagnostic practices across healthcare systems, may slow broader adoption of pancreatic elastase testing services in certain markets.
Opportunity - Expansion of Advanced Immunoassay Technologies and Growing Focus on Digestive Disease Diagnostics
Technological progress in immunoassay-based diagnostic platforms is creating new opportunities for the broader adoption of pancreatic elastase testing. Modern laboratory analyzers and reagent kits are designed to provide improved analytical sensitivity, faster processing times, and greater compatibility with automated clinical laboratory systems. These advancements allow diagnostic laboratories to handle higher testing volumes while maintaining consistent accuracy and quality control. Continuous improvements in assay design, reagent stability, and automation integration are making pancreatic elastase testing more efficient and accessible for hospital laboratories and reference diagnostic centers worldwide.
Moreover, the growing focus on comprehensive digestive health diagnostics is expanding the clinical role of pancreatic enzyme testing. Gastroenterology specialists increasingly emphasize early identification of exocrine pancreatic insufficiency in patients with chronic digestive symptoms or malabsorption conditions. Healthcare providers are also integrating pancreatic elastase measurement into broader gastrointestinal diagnostic panels used to evaluate complex digestive disorders. Expanding healthcare infrastructure in emerging economies, along with the rapid growth of private diagnostic laboratory networks, is further improving access to specialized biochemical testing. As awareness of pancreatic insufficiency continues to rise and laboratory technologies advance, the demand for pancreatic elastase diagnostic solutions is expected to increase steadily in the coming years.
Category-wise Analysis
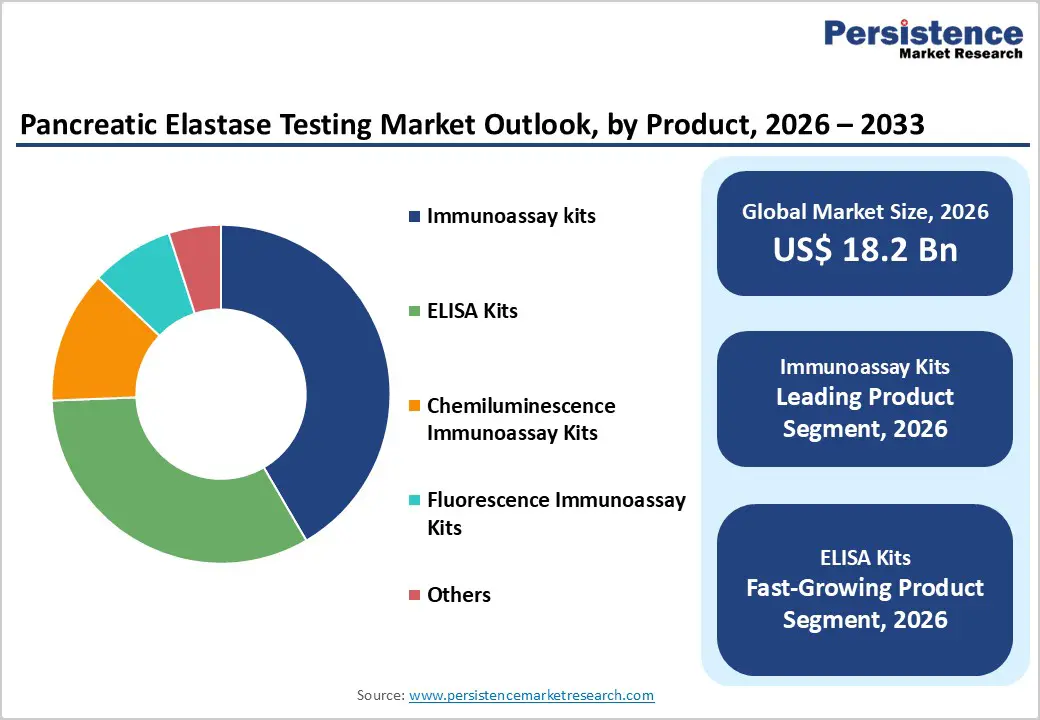
By Product, Immunoassay Kits Dominate Product Category Owing to High Sensitivity and Routine Laboratory Adoption
Immunoassay kits are projected to contribute 41.6% of global revenue in 2026, positioning them as the most widely utilized product category within pancreatic elastase testing. Their strong market presence is primarily linked to their ability to deliver reliable quantitative measurement of fecal pancreatic elastase, a key biomarker used to evaluate pancreatic exocrine function. Clinical laboratories widely prefer immunoassay-based platforms because they provide strong analytical sensitivity while supporting standardized diagnostic workflows. Many hospital laboratories and reference diagnostic centers incorporate these assays into routine gastrointestinal diagnostic panels when evaluating suspected exocrine pancreatic insufficiency.
Continuous improvements in assay chemistry, reagent stability, and automated analyzer compatibility have further strengthened their role in high-throughput laboratory environments. In addition, the growing need for non-invasive diagnostic methods for pancreatic function assessment has encouraged wider adoption of fecal elastase testing kits globally. Increasing prevalence of chronic pancreatic disorders and greater awareness among gastroenterologists regarding early detection of pancreatic insufficiency are also expanding the volume of diagnostic tests performed. As healthcare providers continue prioritizing accurate biochemical markers for pancreatic evaluation, immunoassay kits are expected to maintain their leadership in the global pancreatic elastase testing market.
By Application Insights
The diagnosis of pancreatic disorders segment is expected to account for 58.7% of the market in 2026, making it the largest application area for pancreatic elastase testing. Fecal pancreatic elastase measurement has become a widely accepted non-invasive diagnostic approach for identifying exocrine pancreatic insufficiency associated with chronic pancreatitis, cystic fibrosis, and other pancreatic dysfunctions. Physicians frequently recommend this test when patients present with symptoms such as chronic diarrhea, malabsorption, unexplained weight loss, or nutrient deficiencies. Diagnostic laboratories utilize elastase assays to evaluate pancreatic enzyme secretion capacity and assist clinicians in confirming pancreatic insufficiency. Increased awareness among healthcare professionals regarding early pancreatic dysfunction detection has significantly raised testing frequency. Additionally, advances in immunoassay-based diagnostics allow laboratories to perform these tests efficiently with minimal sample preparation and reliable reproducibility.
Growing incidence of gastrointestinal diseases, increasing diagnostic referrals to gastroenterology specialists, and expansion of clinical laboratory services across healthcare systems are further strengthening demand. As clinical emphasis on early and accurate pancreatic disease identification continues to increase, diagnostic testing for pancreatic disorders will remain the dominant application within the pancreatic elastase testing market.
By End-user Insights
Hospitals are anticipated to generate 51.7% of total market revenue in 2026, confirming their position as the leading end-user segment in the pancreatic elastase testing market. Hospital-based clinical laboratories play a critical role in evaluating patients with suspected gastrointestinal and pancreatic disorders. These facilities typically maintain advanced laboratory infrastructure capable of performing biochemical assays, immunoassay testing, and comprehensive digestive disease diagnostics. Gastroenterology departments frequently rely on fecal elastase testing to assess pancreatic enzyme activity in patients experiencing chronic digestive symptoms or suspected malabsorption conditions.
The presence of trained clinical laboratory professionals and automated diagnostic analyzers enables hospitals to manage high testing volumes efficiently while maintaining consistent quality standards. Hospitals also treat patients with complex pancreatic conditions such as chronic pancreatitis and cystic fibrosis, which often require repeated monitoring of pancreatic function. Rising hospital admissions associated with digestive disorders and increasing referrals to specialized gastroenterology units are further contributing to diagnostic demand. Integration of laboratory data with hospital electronic medical record systems also enhances diagnostic decision-making and treatment planning, reinforcing hospitals’ leading role in pancreatic elastase testing services globally.
Regional Insights
North America Pancreatic Elastase Testing Market Trends
North America is expected to account for 48.5% of global market revenue in 2026, maintaining its leadership position in the pancreatic elastase testing industry. The United States represents the largest contributor, supported by a well-developed healthcare system and an extensive network of hospital laboratories and diagnostic service providers. Gastroenterologists across the region routinely utilize fecal elastase testing as a non-invasive method to evaluate pancreatic exocrine function, particularly in patients suspected of having chronic pancreatitis or digestive malabsorption disorders.
The region benefits from strong diagnostic awareness among healthcare professionals, which encourages early clinical evaluation of pancreatic dysfunction. Advanced laboratory infrastructure allows healthcare facilities to integrate immunoassay-based diagnostic platforms that deliver reliable and rapid testing outcomes. In addition, the presence of leading diagnostic companies and continuous technological innovation in biochemical testing assays contribute to sustained market expansion. Favorable reimbursement policies and strong healthcare spending further enable hospitals and diagnostic laboratories to adopt advanced laboratory testing solutions. Growing incidence of digestive disorders, combined with increasing clinical emphasis on accurate pancreatic function assessment, continues to stimulate diagnostic demand across the region. These factors collectively ensure North America remains the largest and most technologically advanced market for pancreatic elastase testing worldwide.
Europe Pancreatic Elastase Testing Market Trends
Europe represents a mature yet steadily evolving market for pancreatic elastase diagnostics, supported by comprehensive public healthcare systems and well-organized clinical laboratory infrastructure. Countries such as Germany, the United Kingdom, France, Italy, and Spain conduct extensive gastrointestinal diagnostic evaluations through hospital laboratories and specialized diagnostic centers. Healthcare providers across the region place strong emphasis on early identification of pancreatic insufficiency, particularly among patients suffering from chronic digestive conditions and cystic fibrosis. Established clinical guidelines for pancreatic disease management encourage physicians to use fecal elastase testing as part of routine diagnostic evaluation. Europe also benefits from centralized laboratory networks that ensure consistent testing quality and standardized diagnostic procedures across healthcare systems.
The region’s aging population contributes significantly to diagnostic demand, as pancreatic insufficiency and digestive disorders are more common among older individuals. Technological progress within clinical laboratories is gradually enhancing testing efficiency, with greater adoption of automated immunoassay analyzers and advanced biochemical testing platforms. Additionally, regulatory harmonization across the European Union and ongoing investments in healthcare modernization support stable market expansion. Although growth remains moderate compared with emerging regions, consistent diagnostic demand and continuous laboratory upgrades ensure steady development of the pancreatic elastase testing market across Europe.
Asia Pacific Pancreatic Elastase Testing Market Trends
Asia Pacific is projected to experience the fastest growth, registering a CAGR of approximately 8.6% between 2026 and 2033, driven by rapid healthcare development and increasing diagnostic awareness. Large populations in countries such as China, India, Japan, and South Korea generate considerable demand for gastrointestinal diagnostic services. As awareness of pancreatic disorders improves among healthcare professionals, physicians are increasingly recommending fecal elastase testing to evaluate patients presenting with symptoms of malabsorption or chronic digestive discomfort. Governments across the region are investing heavily in healthcare infrastructure, including hospital expansion, clinical laboratory development, and diagnostic technology upgrades.
These investments are significantly improving access to advanced diagnostic services in both urban and semi-urban healthcare facilities. The growing presence of private diagnostic chains and independent pathology laboratories is also expanding testing capacity across major cities. Rising healthcare expenditure and expanding health insurance coverage further enable more patients to undergo specialized diagnostic testing. In addition, international diagnostic companies are strengthening partnerships with regional healthcare providers to improve product availability and laboratory training. With healthcare systems continuing to modernize and clinical awareness of pancreatic disorders steadily increasing, Asia Pacific is expected to emerge as one of the most dynamic growth regions in the global pancreatic elastase testing market.
Competitive Landscape
The global pancreatic elastase testing market is highly competitive, with strong participation from companies such as DiaSorin S.p.A., ScheBo Biotech AG, Immundiagnostik AG, Invivo Healthcare Ltd., DRG Instruments GmbH, and Verisana GmbH. These players leverage extensive in-vitro diagnostic portfolios, strong relationships with hospitals and diagnostic laboratories, advanced immunoassay technologies, and well-established global distribution networks.
Competitive strategies focus on developing highly sensitive ELISA and immunoassay-based pancreatic elastase tests, improving diagnostic accuracy, and enhancing laboratory workflow efficiency. Companies are investing in assay innovation, automation integration, clinical collaborations, and expansion into emerging healthcare markets, which is intensifying competition and supporting sustained market growth globally.
Key Industrial Developments
- In July 2025, QuidelOrtho Corporation and BÜHLMANN Laboratories AG announced the availability of the BÜHLMANN fCAL turbo and fPELA turbo assays on VITROS Systems, enabling particle-enhanced turbidimetric immunoassay testing for fecal calprotectin and fecal pancreatic elastase to support the diagnosis of inflammatory bowel disease and pancreatic insufficiency.
- In February 2023, Microba Life Sciences Limited announced the launch of its next-generation microbiome testing platform MetaXplore™, introduced to healthcare professionals in Australia under the new brand Co-Biome™. The platform enables advanced microbiome analysis to support improved clinical insights into digestive health and related conditions.
Companies Covered in Pancreatic Elastase Testing Market
- DiaSorin S.p.A.
- ScheBo Biotech AG
- Immundiagnostik AG
- Invivo Healthcare Ltd
- DRG Instruments GmbH
- Verisana GmbH
- BÜHLMANN Laboratories AG
- CerTest Biotec S.L.
- R-Biopharm AG
- American Laboratory Products Company, Ltd. (ALPCO)
- Vitassay Healthcare, S.L.U.
- Eurospital S.p.A.
- Bioserv Diagnostics GmbH
- Eagle Biosciences, Inc.
- Alpha Laboratories Ltd
- Others
Frequently Asked Questions
What is the global pancreatic elastase testing market in 2026?
Rising prevalence of pancreatic disorders such as chronic pancreatitis, cystic fibrosis, and exocrine pancreatic insufficiency is increasing demand for pancreatic elastase diagnostic testing.
The global pancreatic elastase testing market is poised to witness a CAGR of 6.4% between 2026 and 2033.
Advancements in non-invasive and point-of-care pancreatic elastase testing technologies are creating new opportunities for faster and decentralized pancreatic function diagnostics.
DiaSorin S.p.A., ScheBo Biotech AG, Immundiagnostik AG, Invivo Healthcare Ltd., DRG Instruments GmbH, and Verisana GmbH are some of the key players in the pancreatic elastase testing market.













