- Clothing, Footwear, & Accessories
- Orthopedic Shoes Market
Orthopedic Shoes Market Size, Share, and Growth Forecast, 2026 – 2033
Orthopedic Shoes Market by Product Type (Standard Shoes, Custom-made Shoes, Diabetic Shoes, Arch support Shoes, Orthopedic Insoles, Others), Application (Medical Treatment Use, Preventive Use, Post-surgery Use, Rehabilitation Support), Distribution Channel (Online Sales, Specialty Footwear Stores, Hospitals, Orthotic Centers), and Regional Analysis for 2026 - 2033
Orthopedic Shoes Market Share and Trends Analysis
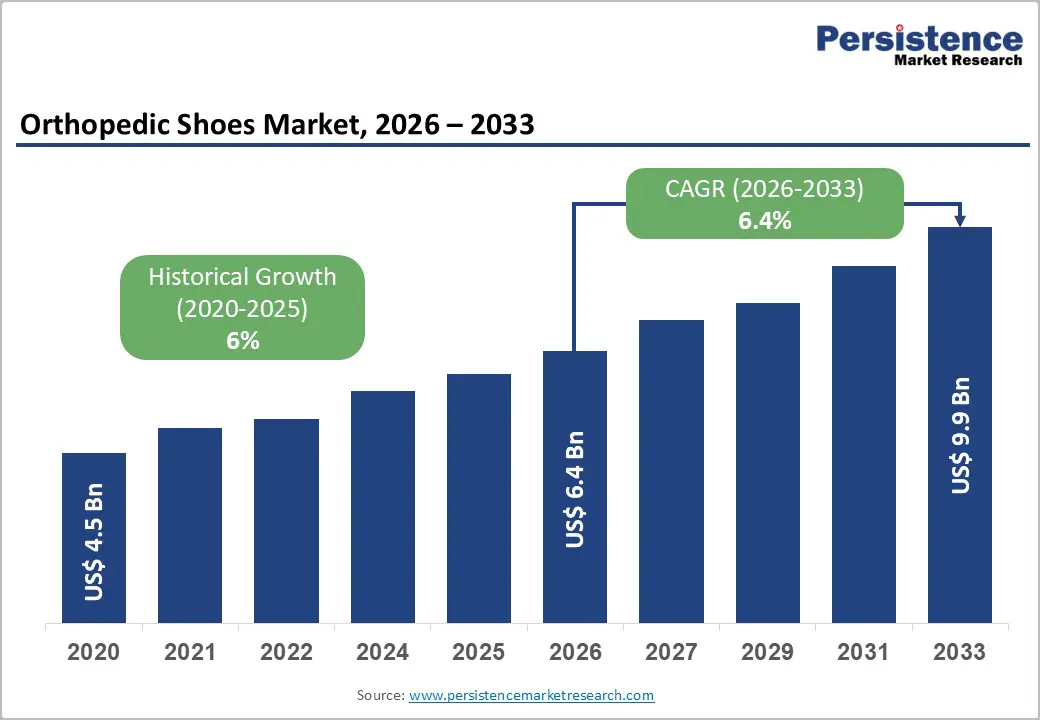
The global orthopedic shoes market size is likely to be valued at US$ 6.4 billion in 2026, and is projected to reach US$ 9.9 billion by 2033, growing at a CAGR of 6.4% during the forecast period 2026–2033.
The growth in the market is primarily driven by the rising prevalence of diabetes and musculoskeletal disorders, both of which significantly increase the need for therapeutic and preventive foot care solutions. According to international health agencies, diabetes-related foot complications and arthritis cases continue to rise, creating sustained demand for diabetic shoes and medically approved footwear. Aging demographics further reinforce this trend, as older adults are more susceptible to mobility limitations and balance disorders.
Regulatory recognition of orthopedic footwear as medical-grade devices in major markets strengthens reimbursement frameworks and physician-led prescriptions. Furthermore, technological advancements in digital foot scanning, 3D customization, and expanding e-commerce platforms are improving product accessibility and consumer adoption across developed and emerging economies.
Key Industry Highlights
- Dominant Product Type: Diabetic shoes are set to command around 35% revenue share in 2026, while custom-made shoes are likely to grow the fastest through 2033, driven by digital fitting technologies.
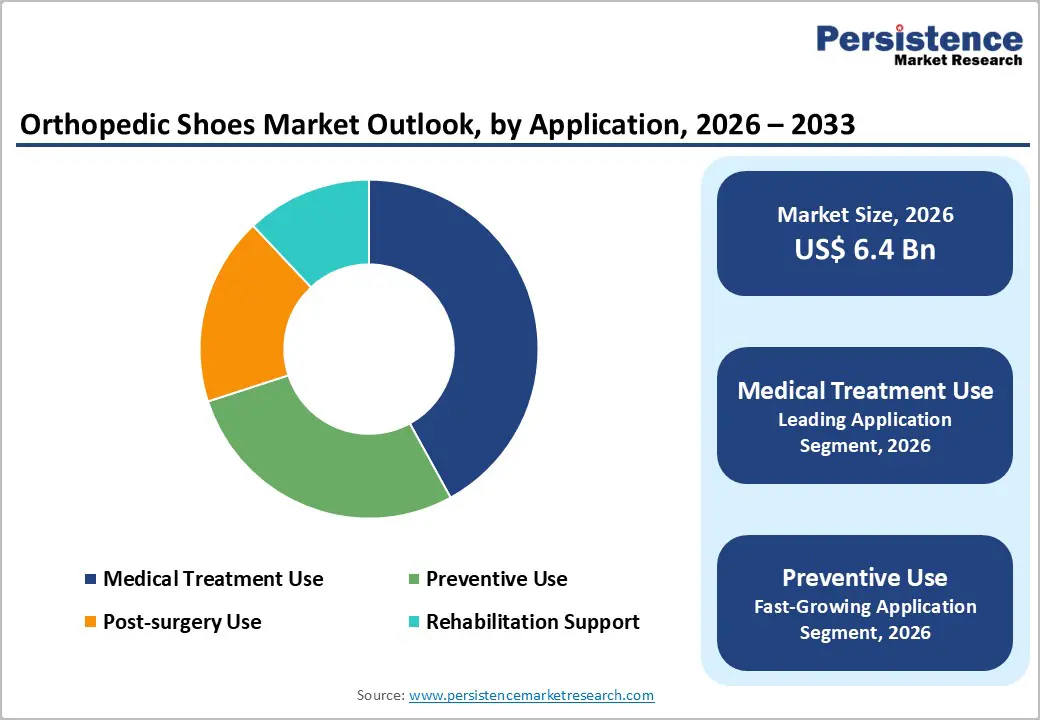
- Leading Application: Medical treatment use is projected to hold about 42% share in 2026, while preventive use is expected to expand at the fastest rate of roughly 7.2% CAGR during 2026–2033, supported by rising foot health awareness.
- Dominant Distribution Channel: Specialty footwear stores are anticipated to account for nearly 39% share in 2026, while online sales are forecast to grow the fastest through 2033, driven by AI-enabled direct-to-consumer (D2C) platforms.
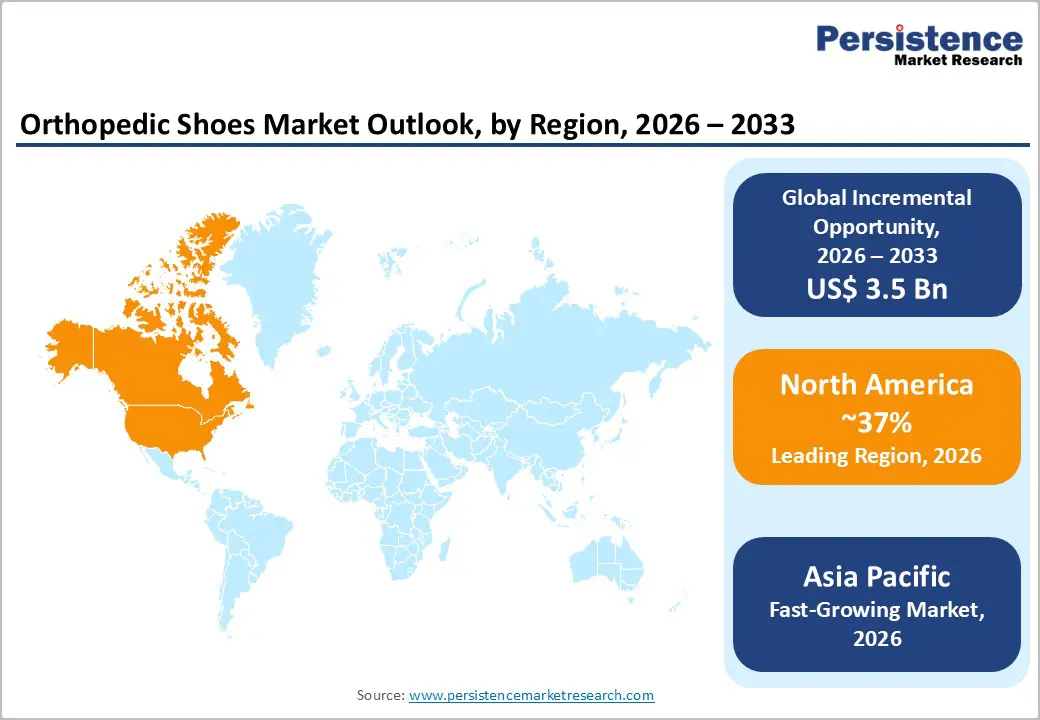
- Regional Leadership: North America is poised to lead with approximately 37% share in 2026, while Asia Pacific is projected to register the highest 2026-2033 growth at 7.9% CAGR, fueled by demographic and income changes.
- Competitive Environment: Manufacturers’ strategies emphasize AI-based customization, manufacturing expansion, and digital channel investments to strengthen market positioning.
| Report Attribute | Details |
|---|---|
|
Orthopedic Shoes Market Size (2026E) |
US$ 6.4 Bn |
|
Market Value Forecast (2033F) |
US$ 9.9 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
6.4% |
|
Historical Market Growth (CAGR 2020 to 2025) |
6.0% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Rising Disease Burden and Aging Demographics Driving Clinical Demand
The increasing burden of diabetes and orthopedic conditions significantly drives demand for orthopedic shoes, diabetic shoes, and custom orthopedic footwear. According to the International Diabetes Federation (IDF), over 537 million adults worldwide live with diabetes, with numbers projected to rise further by 2030. The World Health Organization (WHO) reports that 1.71 billion people are affected by musculoskeletal conditions, making them a leading cause of disability globally. Diabetes-related complications such as neuropathy and foot ulcers increase the need for therapeutic footwear to prevent severe outcomes, including amputations. Healthcare authorities including the U.S. Centers for Disease Control and Prevention (CDC) emphasize regular foot examinations and appropriate footwear as preventive measures. These clinical guidelines and rising chronic disease prevalence continue to strengthen demand across therapeutic and preventive orthopedic footwear segments.
In April 2025, a major orthopedic footwear and limb salvage project was launched in Barbados to prevent amputations from diabetic foot disease, highlighting government support for therapeutic footwear. Public health campaigns and healthcare conferences emphasized limb preservation and early intervention, reinforcing clinical adoption. Population aging further expands the market for medical and arch support shoes, as the UN DESA projects the global 65+ population will double by 2050, increasing risks of arthritis, plantar fasciitis, and balance disorders. Healthcare systems in North America and Europe prioritize fall prevention and mobility maintenance, integrating orthopedic shoes into rehabilitation and long-term care. These demographic and policy trends create recurring demand via prescription channels and reimbursement frameworks, strengthening orthopedic footwear’s role in preventive health and providing a stable revenue base.
Technological Innovation and Expanding Access Channels
Advancements in technology are transforming how orthopedic footwear is designed, produced, and distributed, supporting market expansion. 3D scanning, CAD-based orthotic design, and pressure-mapping technologies improve precision and fit accuracy for custom-made orthopedic shoes, reducing lead times and enhancing patient outcomes. These tools enable manufacturers to address individual biomechanical needs with greater clinical efficacy. The healthcare innovation forums highlighted digital platforms that enable clinician-driven ordering and nationwide delivery of custom therapeutic footwear through mobile applications. This integration expands access without requiring in-person visits. These advancements improve workflow efficiency and patient access to personalized medical-grade footwear.
Industry adoption of additive manufacturing (3D printing) enhances cost optimization and localized production. As reported in research publications and news, smart sensor integration and telehealth platforms are contributing to next-generation orthopedic solutions that combine fit, analytics, and comfort, particularly in diabetic foot care. These technological advancements, including remote assessment tools and online fitting services, are supporting direct-to-consumer growth and expanding reach into emerging and underserved markets. By reducing barriers to personalization and improving clinical outcomes, technology is strengthening product differentiation and supporting premium pricing strategies in both developed and emerging economies.
High Product Costs and Limited Reimbursement Coverage
Custom orthopedic footwear involves higher manufacturing costs due to individualized design, specialized labor, and medical-grade materials, making these products more expensive than standard footwear. In markets without universal reimbursement, high out-of-pocket expenses can limit adoption, while Medicare in the U.S. and some European healthcare systems provide conditional coverage requiring strict clinical documentation. These administrative requirements can delay procurement and reduce flexibility for providers and patients. Emerging economies face stronger price sensitivity due to low insurance penetration, restricting demand for premium or fully customized solutions and concentrating sales in specialty stores and orthotic centers.
Operational and reimbursement pressures are increasing. In 2025, the U.S. Centers for Medicare & Medicaid Services (CMS) required annual accreditation for durable medical equipment, prosthetics, and orthotics suppliers, raising documentation and compliance burdens. Scrutiny continued into 2026, with auditors demanding detailed medical necessity records and fabrication tracking for orthotic claims. These requirements extend time-to-payment, increase operational complexity, and limit smaller manufacturers’ ability to invest in innovation, slowing market expansion and constraining volume growth in price-conscious segments.
Regulatory Compliance and Product Classification Complexities
Orthopedic shoes are often classified as medical devices in major markets, including under the U.S. Food and Drug Administration (FDA) and European Union Medical Device Regulation (EU MDR). Compliance requires comprehensive quality management systems, clinical evaluation documentation, and ongoing post-market surveillance. Regulatory approval can be time-intensive, particularly for therapeutic or corrective devices, and manufacturers must ensure traceability, labeling accuracy, and conformity before entering or expanding in regulated markets. These requirements increase operational costs and extend time-to-market, especially for small and mid-sized companies.
Evolving regulatory updates and program changes further complicate compliance. Updates to competitive bidding programs and stricter documentation audits have increased administrative burdens, adding uncertainty to pricing and reimbursement. Capital investments for certification audits, technical documentation, and regulatory consultants are often necessary, creating barriers for new entrants. These complexities can slow innovation, delay product launches, and contribute to moderate industry consolidation, as larger, financially stronger firms are better positioned to manage compliance and maintain market access.
Opportunity Analysis - Expansion in Emerging Asia Pacific Markets
Rapid urbanization, rising healthcare awareness, and growing disposable incomes in China, India, and ASEAN nations are creating significant growth opportunities for orthopedic footwear. Increasing diabetes prevalence in the region, coupled with expanding middle-class purchasing power, supports adoption of preventive and therapeutic foot care solutions. Local manufacturing advantages reduce production costs, enabling competitive pricing strategies across both institutional and retail channels.
Government investment in primary healthcare infrastructure is enhancing distribution opportunities through hospitals, clinics, and orthotic centers. Programs targeting diabetes prevention and foot health are increasing public awareness of medically approved footwear, which mirrors initiatives such as Barbados’ 2025 limb salvage project, showing the effectiveness of government-backed preventive footwear programs. Partnerships with local distributors and hospital networks further facilitate market penetration. These developments make Asia Pacific a strategic growth hub, offering both volume and value potential for global orthopedic footwear manufacturers.
Growth in Preventive Foot Care and Direct-to-Consumer Channels
The preventive use segment is growing as consumers increasingly adopt comfort-oriented, ergonomic footwear for posture correction, plantar fasciitis management, and everyday foot health. Fitness-conscious and aging populations are integrating arch support shoes and orthopedic insoles into their lifestyle, broadening the customer base and reducing reliance on strictly prescription-based channels. Growing awareness of preventive foot care in workplaces, gyms, and senior care communities further supports this trend.
U.S. Medicare telehealth coverage was extended through 2027, enabling remote foot health assessments and virtual orthotic prescriptions, while Special Diabetes Programs funding continued through 2026, supporting preventive care and early adoption of therapeutic footwear. Advanced digital tools, including smart insoles and virtual fitting platforms, allow direct-to-consumer engagement, expanding reach without heavy retail investment and supporting recurring purchases. These developments altogether strengthen the e-commerce scalability, personalized care delivery, and long-term adoption of preventive orthopedic footwear, while creating opportunities for integrated health solutions.
Category-wise Analysis
Product Type Insights
Diabetic shoes are expected to remain the leading product type, estimated to hold about 32% of the orthopedic footwear market revenues in 2026, owing to their established clinical adoption and structured reimbursement support in major healthcare systems. These products are widely prescribed for managing diabetic foot complications, driving recurring demand through hospitals and orthotic centers. In 2025, Insollz secured a national supply partnership with the U.K.’s National Health Service (NHS) to provide its clinical insoles through Ortho Europe catalogues, expanding access to therapeutic insole designs across NHS orthotic clinics. This reinforces clinical relevance and distribution strength in institutional channels.
Custom made shoes are anticipated to be the fastest growing product type, projected to register a 9.1% CAGR through 2033 as personalization and precision become key differentiators in orthopedic footwear. In Singapore, Sengkang General Hospital’s in house 3D printing of custom insoles reduced turnaround to one week, demonstrating how clinical adoption of digital fabrication enhances accessibility, quality, and value in customized foot solutions. Rising consumer preference for personalized comfort and performance, along with digital foot scanning and printing technologies showcased at events such as OTWorld 2026, further accelerates adoption.
Application Insights
The medical treatment use application segment is likely to lead with an estimated 41% of the orthopedic shoes market share in 2026, driven by physician prescriptions and reimbursement pathways for diabetic foot care, post surgery support, and rehabilitation programs. Hospitals and orthotic centers remain the primary procurement channels, with structured clinical protocols emphasizing therapeutic footwear within treatment regimens. In 2025, European orthopedic footwear companies expanded distribution agreements with hospital networks, enhancing availability of medically certified shoes and strengthening integration into formal care programs.
Preventive use is poised to be the fastest growing application, projected to achieve an 8.7% CAGR through 2033, as rising foot health awareness and ergonomic priorities expand demand into lifestyle and wellness segments. Consumer emphasis on posture correction, plantar fasciitis prevention, and daily comfort drives uptake of orthopedic insoles and arch support shoes outside clinical settings. Trade shows such as OTWorld 2026 in Leipzig, emphasizing innovations in diabetic foot syndrome care and preventive technologies, are promoting knowledge transfer and adoption of preventive solutions among clinicians and end users alike. Digital tools and e-commerce platforms are further enhancing accessibility, enabling personalized preventive products to reach broader populations.
Regional Insights
North America Orthopedic Shoes Market Trends
North America is expected to be the leading regional market for orthopedic shoes in 2026, with the U.S. expected to hold approximately 37% share in 2026, supported by rising chronic conditions, aging population, and strong healthcare infrastructure. Preventive care integration, including physician-led screening and clinical foot assessments, drives consistent demand for therapeutic and custom footwear. Hospitals, rehabilitation centers, and specialty orthotic clinics serve as primary procurement channels. Medicare reimbursement for eligible patients strengthens accessibility, while awareness campaigns educate consumers on diabetic foot complications and musculoskeletal health. The combination of institutional support and public awareness creates a stable and recurring market base.
In late 2025, Podimetrics released a provider insights report highlighting best practices to prevent diabetic foot ulcers and reduce amputations, emphasizing remote temperature monitoring and coordinated clinical collaboration. Simultaneously, NIH-funded trials are evaluating sensor-enabled insoles and remote monitoring platforms to detect early signs of foot complications. These developments illustrate North America’s leadership in integrating digital health technologies with orthopedic care. The convergence of clinical guidance, digital tools, and preventive strategies is strengthening adoption across hospitals, orthotic centers, and direct-to-consumer platforms.
Europe Orthopedic Shoes Market Trends
Europe remains a mature and stable market for orthopedic footwear, with Germany, the U.K., France, and Spain, supported by structured public healthcare systems, regulatory clarity, and growing clinical focus on foot health. Regulatory harmonization under the EU Medical Device Regulation ensures high product quality and patient safety, underpinning broad clinician confidence and institutional adoption across member states. National health services and insurance frameworks in Germany and the U.K. offer structured reimbursement pathways for medically necessary footwear, further strengthening uptake among diabetic and musculoskeletal patient groups.
Support the market movements, in early 2026, IDF Europe launched policy actions aimed at advancing integrated diabetes care, calling for a shift from fragmented care approaches to coordinated, person centered systems that align preventive care, early detection, and therapeutic support. This policy momentum underscores a broader public health emphasis on continuous, integrated management of diabetes and related complications, including foot care. In parallel, professional networks such as the European Wound Management Association (EWMA) are conducting diabetic foot care surveys and collaborative programs to map care gaps and improve prevention, treatment, and rehabilitation outcomes across Europe. These initiatives reinforce clinical best practices and strengthen demand for orthopedic solutions that support comprehensive diabetes care.
Asia Pacific Orthopedic Shoes Market Trends
Asia Pacific is anticipated to be the fastest growing regional market for orthopedic shoes, projected to record a CAGR of 7.9% from 2026 to 2033, propelled by expanding diabetic and aging populations, rising incomes, and improving healthcare access. Emerging public health initiatives and cross border clinical forums are supporting broader adoption of diabetic foot care and orthopedic solutions in the region. The 21st Asia Pacific Association for Diabetic Limb Problems (APADLP) Conference, held in Bangkok in November 2025, brought together regional clinicians and policymakers to share clinical insights and best practices aimed at improving diabetic limb care outcomes and enhancing awareness of preventive interventions.
Regional collaboration continues to strengthen professional networks and clinical knowledge transfer: preparations for the World Union of Wound Healing Societies (WUWHS) Congress 2026 in Kuala Lumpur position Asia Pacific as a hub for global wound care leadership, advancing evidence based approaches to diabetic foot prevention and treatment. Participation by clinicians, researchers, and industry partners at this congress, widely recognized in wound care and diabetic limb health circles, supports adoption of advanced rehabilitation and preventive practices. Combined with increasing digital healthcare adoption and expanding distribution through e-commerce and clinical channels, these developments provide Asia Pacific with strong momentum for future orthopedic footwear growth.
Competitive Landscape
The global orthopedic shoes market structure is moderately consolidated, with leading players such as Dr. Comfort, Aetrex Worldwide, Vionic Group, Bauerfeind AG, and OrthoStep controlling over half of the revenue share in 2026. These companies leverage strong hospital and orthotic center networks, regulatory compliance, and clinical validation, while investing in R&D for digital fitting, 3D-printed insoles, sensor-enabled monitoring, and ergonomic designs.
Regional and niche players, including Pedag GmbH, Apex Foot Health, and Schein Orthopedic, focus on specialized segments and geographic strongholds. Regulatory complexity and custom manufacturing barriers limit new entrants, but e-commerce and online customization enable smaller firms to grow. Market consolidation is expected through acquisitions, partnerships, and digital platform collaborations, while technology-driven personalization supports scalable, preventive, and therapeutic footwear adoption.
Key Industry Developments
- In January 2026, Anodyne, a Wisconsin-based therapeutic footwear manufacturer, completed a buyout of Dia-Foot, a Florida-based diabetic shoe company. This acquisition consolidates Anodyne’s retail presence in the specialized diabetic and orthopedic footwear market, enhancing product offerings and in-house laboratory capabilities for foot health solutions.
- In November 2025, Acumed finalized the acquisition of TECHFIT’s proprietary digital surgical planning and patient-specific reconstruction tools. This strategic move strengthens Acumed’s capabilities in customized orthopedic and reconstructive solutions, enabling improved surgical precision and reduced operative times globally.
- In April 2025, Zimmer Biomet completed a US$ 1.2 billion acquisition of Paragon 28, expanding its foot and ankle orthopedic portfolio. Paragon 28 becomes a wholly-owned subsidiary, allowing Zimmer Biomet to capitalize on the high-growth foot and ankle market while integrating Paragon’s specialized product lines into its global distribution network.
Companies Covered in Orthopedic Shoes Market
- Ottobock SE & Co. KGaA
- Aetrex Worldwide Inc.
- Dr. Comfort
- Drew Shoe Corporation
- Mephisto
- Birkenstock Holding plc
- New Balance Athletics Inc.
- Orthofeet Inc.
- Propet USA Inc.
- Finn Comfort
- Vionic Group LLC
- Apex Foot Health Industries
- Hush Puppies
Frequently Asked Questions
The global orthopedic shoes market is projected to reach US$ 6.4 billion in 2026.
Heightening diabetes prevalence worldwide, aging population, and increased focus on preventive foot care are driving the market.
The market is poised to witness a CAGR of 6.4% from 2026 to 2033.
Growth in emerging Asia Pacific markets, preventive foot care adoption, and online distribution channels are key opportunities.
Dr. Comfort, Aetrex Worldwide, Vionic Group, Bauerfeind AG, and OrthoStep are among the leading players.








