- Biotechnology
- miRNA Sequencing and Assay Market
miRNA Sequencing and Assay Market Size, Share, Growth, and Regional Forecast, 2026 to 2033
Global miRNA Sequencing and Assay Market by Product (Instruments, Sequencing Consumables, Library Preparation Kits, and Bioinformatics Pipelines & Services), by Technology (Sequencing by Synthesis (SBS), Ion Semiconductor, Nanopore Sequencing, Single-Molecule Real-Time (SMRT), and Others), by Application (Oncology Diagnostics, Drug Discovery & Transcriptome Research, Polyglutamine Diseases, and Others) by End User, and Regional Analysis from 2026 to 2033
miRNA Sequencing and Assay Market Size and Share Analysis
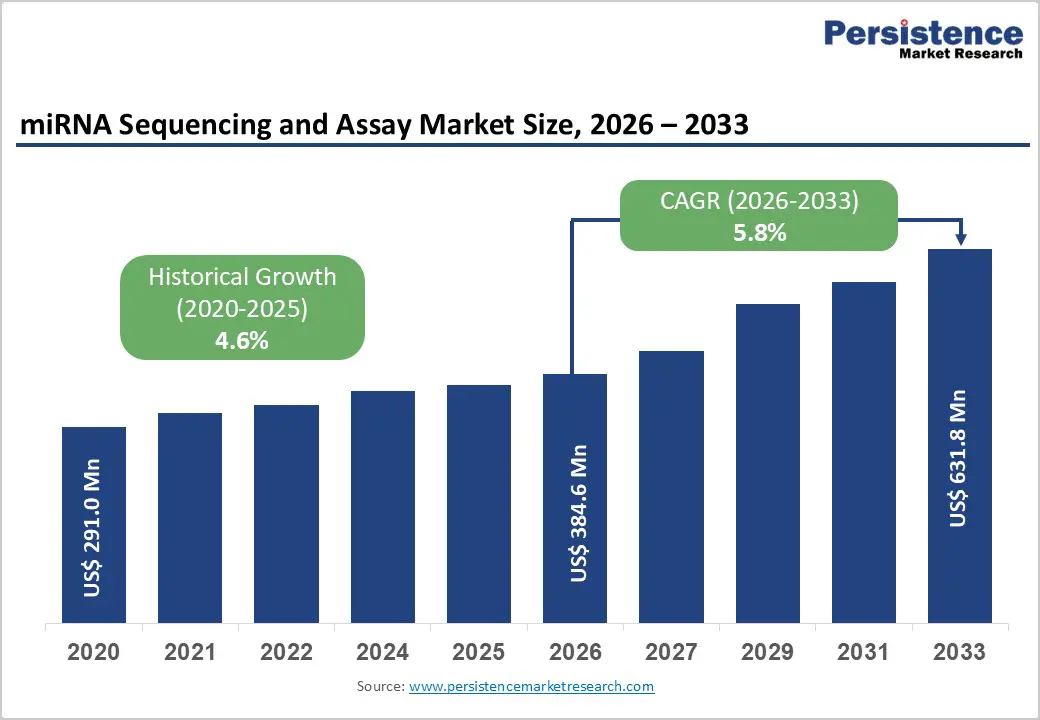
The global miRNA sequencing and assay market size is estimated to grow from US$ 384.6 Mn in 2026 to US$ 631.8 Mn by 2033. The market is projected to record a CAGR of 5.8% during the forecast period from 2026 to 2033.
Global demand for miRNA sequencing and assay technologies is increasing steadily, driven by the rising focus on precision medicine, growing use of molecular biomarkers in disease diagnosis, and expanding application of next-generation sequencing in translational research. Increasing adoption of miRNA profiling across oncology, neurology, cardiovascular research, and metabolic disease studies is supporting sustained market growth. Higher volumes of biomarker discovery programs, clinical research studies, and drug development activities, coupled with rising healthcare and life science R&D expenditure, are further accelerating demand. Continuous innovation in sequencing chemistries, library preparation workflows, and bioinformatics pipelines is improving sensitivity, reproducibility, and data accuracy. In addition, growing interest in liquid biopsy, non-invasive diagnostics, and digitally integrated genomics platforms is further propelling the global miRNA sequencing and assay market.
Key Industry Highlights
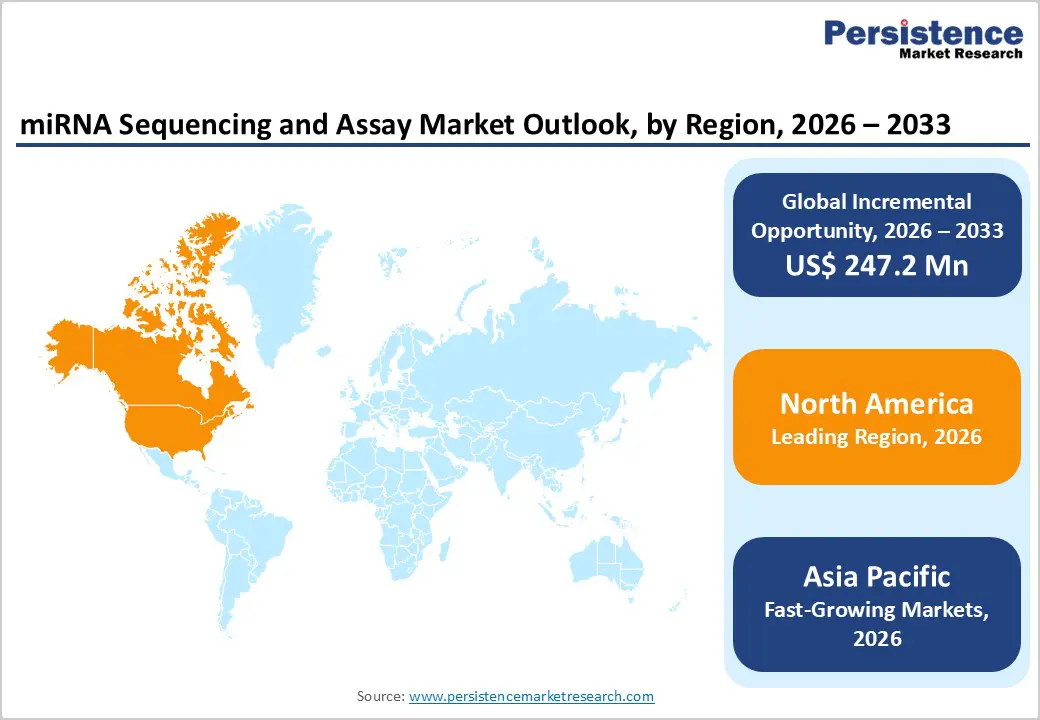
- Leading Region: North America holds the largest share at 46.7%, supported by strong genomics research infrastructure, high adoption of next-generation sequencing technologies, robust funding for cancer and precision medicine research, and the strong presence of leading sequencing and life science companies.
- Fastest-Growing Region: Asia Pacific is expanding fastest due to rising investments in genomics infrastructure, growing cancer and chronic disease burden, increasing clinical research activity, and government-led precision medicine initiatives.
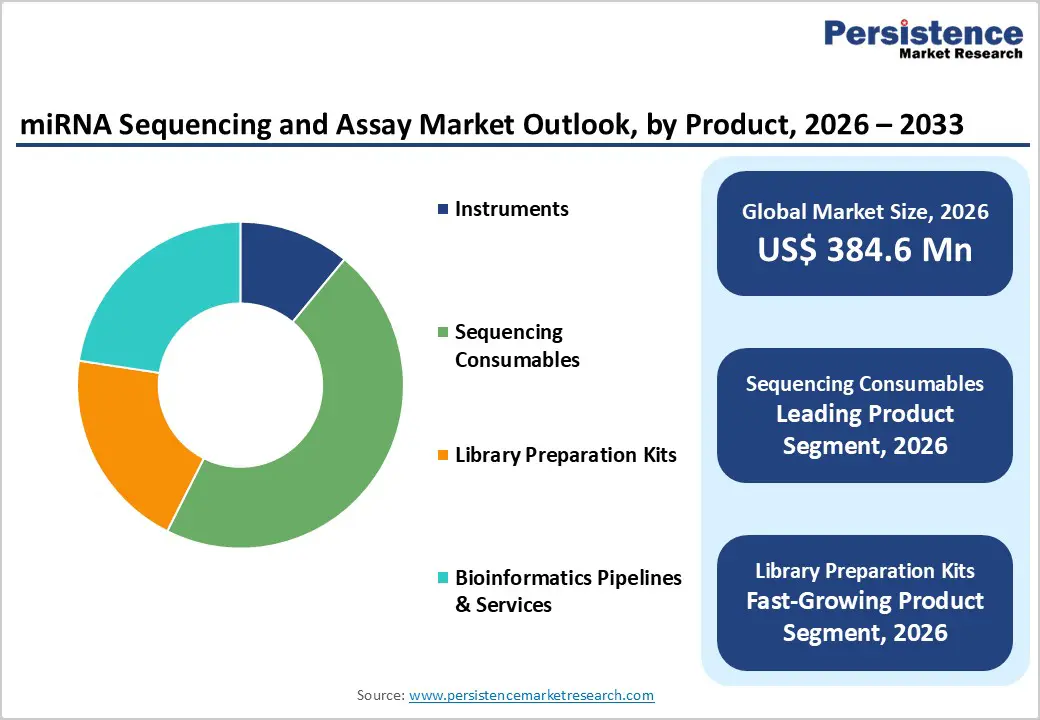
- Leading Product Segment: Sequencing consumables dominate the market due to their recurring use across miRNA sequencing runs, biomarker validation studies, and large-scale transcriptome research projects.
- Fastest-Growing Product Segment: Library preparation kits are expanding rapidly as demand increases for high-sensitivity, low-input miRNA analysis in liquid biopsy and clinical research applications.Leading Application Segment: Oncology diagnostics remains the top application, driven by extensive use of miRNA biomarkers for cancer detection, prognosis assessment, and treatment response monitoring.
- Fastest-Growing Application Segment: Drug discovery & transcriptome research is scaling quickly as pharmaceutical and biotechnology companies increasingly use miRNA profiling to support target identification, mechanism-of-action studies, and companion diagnostic development.
| Report Attribute | Details |
|---|---|
|
miRNA Sequencing and Assay Market Size (2026E) |
US$ 384.6 Mn |
|
Market Value Forecast (2033F) |
US$ 631.8 Mn |
|
Projected Growth (CAGR 2026 to 2033) |
5.8% |
|
Historical Market Growth (CAGR 2020 to 2025) |
4.6% |
Market Dynamics
Driver – Growing Adoption of Precision Medicine and Expanding Role of miRNA in Disease Diagnostics
The primary growth driver is the increasing integration of miRNA sequencing and assay technologies into precision medicine, biomarker discovery, and disease diagnostics. miRNAs play a critical regulatory role in gene expression and are increasingly recognized as stable, sensitive biomarkers for cancer, cardiovascular diseases, neurological disorders, and metabolic conditions. Rising global disease burden, coupled with the need for early-stage detection and personalized treatment strategies, is accelerating demand for accurate miRNA profiling tools. Advances in next-generation sequencing platforms have significantly improved sensitivity, throughput, and reproducibility, enabling reliable detection of low-abundance miRNAs from limited samples such as plasma, serum, and other biofluids.
Additionally, expanding translational research and large-scale genomics programs funded by governments and private institutions are driving sustained utilization of miRNA sequencing workflows. Pharmaceutical and biotechnology companies are increasingly leveraging miRNA analysis to support drug target identification, mechanism-of-action studies, and companion diagnostics. The growing body of clinical evidence linking miRNA expression patterns to disease prognosis and therapeutic response continues to strengthen adoption across both research and clinical settings, reinforcing long-term market momentum.
Restraints – High Workflow Costs, Data Complexity, and Limited Clinical Standardization
The adoption is constrained by the high overall cost of miRNA sequencing and assay workflows. Expenses associated with sequencing consumables, specialized library preparation kits, advanced instruments, and bioinformatics infrastructure can be prohibitive, particularly for smaller laboratories and institutions in resource-limited regions. In addition, miRNA analysis generates complex datasets that require skilled personnel and robust computational capabilities for accurate interpretation, creating operational and talent-related challenges. Lack of standardized protocols for sample preparation, normalization, and data analysis further limits comparability of results across laboratories and clinical studies. Regulatory uncertainty surrounding clinical validation of miRNA-based diagnostics also slows translation from research to routine clinical use. Variability in reimbursement coverage for molecular diagnostics, especially for exploratory or preventive applications, restricts uptake in cost-sensitive healthcare systems. Furthermore, technical challenges such as sequencing bias, low input RNA variability, and cross-platform inconsistencies can impact data reliability. Collectively, cost pressures, analytical complexity, and limited standardization act as key barriers moderating the pace of broader market adoption.
Opportunity – Expansion of Liquid Biopsy, AI-Enabled Analytics, and Clinical Translation of miRNA Biomarkers
Significant growth opportunities are emerging from the expanding use of miRNA sequencing in liquid biopsy and non-invasive diagnostics. Circulating miRNAs derived from blood and other biofluids offer substantial potential for early cancer detection, disease monitoring, and treatment response assessment. Increasing clinical acceptance of liquid biopsy approaches is driving demand for highly sensitive and reproducible miRNA assays. Parallel advancements in artificial intelligence and machine learning are enhancing interpretation of complex miRNA expression datasets, enabling faster biomarker validation and improved clinical decision-making. Integration of sequencing platforms with cloud-based analytics is further lowering barriers for data management and collaborative research.
Additionally, ongoing clinical trials evaluating miRNA signatures as prognostic and predictive biomarkers are accelerating translation into regulated diagnostic applications. Emerging markets present untapped opportunities as governments invest in genomics infrastructure and precision medicine initiatives. As standardization improves and costs gradually decline, miRNA sequencing is expected to move beyond research into routine clinical workflows, positioning advanced assays and integrated analytical solutions as a key long-term growth avenue for industry participants.
Category-wise Analysis
By Product, Sequencing Consumables Lead Due to Recurring Usage Across High-Throughput miRNA Profiling Workflows
The sequencing consumables segment is projected to dominate the global miRNA sequencing and assay market in 2026, accounting for a revenue share of 46.4%. This leadership is primarily driven by the repetitive and high-volume use of reagents, enzymes, flow cells, buffers, and indexing primers across miRNA sequencing and expression analysis workflows. Unlike instruments, consumables are required for every sequencing run, ensuring consistent demand across research and clinical settings. Increasing adoption of miRNA profiling in oncology diagnostics, biomarker discovery, and transcriptome research is significantly boosting consumables consumption. In addition, growing sample volumes from population-scale genomics studies and multi-center research collaborations are reinforcing demand. The rise of liquid biopsy-based miRNA analysis, coupled with expanding use of next-generation sequencing platforms, further accelerates consumable usage. Continuous innovation in chemistry formulations, improved sensitivity for low-input samples, and compatibility with multiple sequencing platforms continues to support sustained dominance of this segment.
By Application, Sequencing by Synthesis (SBS) Leads Due to High Accuracy and Broad Platform Adoption
The sequencing by synthesis (SBS) segment is projected to dominate the global miRNA sequencing and assay market in 2026, accounting for a revenue share of 62.5%. This dominance is driven by the widespread adoption of SBS-based platforms in miRNA sequencing due to their high accuracy, scalability, and ability to generate reproducible data for small RNA analysis. SBS technology is extensively used in oncology research, transcriptome profiling, and biomarker validation studies, where precise quantification of miRNA expression is critical. Its compatibility with high-throughput workflows enables large-scale studies across academic institutions, pharmaceutical companies, and clinical research organizations. In addition, SBS platforms support multiplexing, cost efficiency at scale, and seamless integration with established bioinformatics pipelines. Continuous improvements in read quality, reduced error rates, and enhanced chemistry for short RNA sequencing further strengthen adoption. Strong vendor ecosystems and standardized protocols also reinforce SBS as the preferred technology across global miRNA research initiatives.
By End User, Academic & Research Institutes Lead Due to High Research Intensity and Grant-Funded Genomics Programs
The academic & research institutes segment is projected to dominate the global miRNA sequencing and assay market in 2026, accounting for a revenue share of 41.2%. This dominance is largely driven by the extensive use of miRNA sequencing and assay technologies in basic research, translational studies, and disease mechanism exploration. Universities and research institutes conduct a high volume of miRNA-focused projects related to cancer biology, neurological disorders, cardiovascular diseases, and rare genetic conditions. Strong access to public and private research funding enables continuous investment in sequencing consumables, library preparation kits, and bioinformatics tools. These institutions also play a central role in method development, biomarker discovery, and validation studies, which require repeated sequencing runs. Collaboration with pharmaceutical companies and CROs further increases assay utilization. In addition, the presence of skilled genomics researchers and established sequencing infrastructure supports sustained demand. Expanding global emphasis on precision medicine and systems biology continues to reinforce leadership of this segment.
Region-wise Insights
North America miRNA Sequencing and Assay Market Trends
North America is expected to dominate the global miRNA sequencing and assay market with a value share of 46.7% in 2026, led primarily by the United States. The region benefits from a highly mature genomics ecosystem, strong federal and private research funding, and early adoption of next-generation sequencing technologies. Academic institutions, cancer research centers, and pharmaceutical companies across North America extensively utilize miRNA sequencing for biomarker discovery, oncology diagnostics, and drug development. High prevalence of cancer and chronic diseases has accelerated demand for advanced molecular profiling tools, including miRNA-based assays.
The region also demonstrates strong integration of sequencing platforms with advanced bioinformatics and cloud-based data analysis solutions. Favorable reimbursement environments for molecular diagnostics, combined with robust clinical trial activity, further support market expansion. Additionally, the strong presence of leading sequencing technology providers and continuous innovation in library preparation and assay sensitivity reinforce market leadership. Ongoing investments in precision medicine, liquid biopsy research, and translational genomics ensure sustained growth across North America.
Europe miRNA Sequencing and Assay Market Trends
The Europe miRNA sequencing and assay market is expected to grow steadily, supported by increasing investments in genomics research, an aging population, and rising incidence of cancer and neurological disorders. Countries such as Germany, the U.K., France, Italy, and the Nordic nations maintain strong public healthcare and research infrastructures, enabling broad adoption of miRNA sequencing in both academic and clinical research settings. European research institutions actively participate in large-scale consortium-based genomics projects, driving sustained demand for sequencing consumables and assay kits.
Growing emphasis on early disease detection and personalized medicine is expanding the use of miRNA biomarkers in oncology and rare disease research. In addition, stringent regulatory standards are encouraging adoption of high-quality, validated sequencing workflows. Advances in bioinformatics standardization and cross-border data-sharing initiatives further enhance market penetration. Increasing focus on translational research, coupled with expanding biotech activity and supportive innovation policies, continues to strengthen Europe’s position in the global miRNA sequencing and assay landscape.
Asia Pacific miRNA Sequencing and Assay Market Trends
The Asia Pacific miRNA sequencing and assay market is expected to register a relatively higher CAGR of around 7.9% between 2026 and 2033, driven by rapid expansion of genomics infrastructure and increasing research output. Large patient populations, rising cancer incidence, and growing awareness of molecular diagnostics are fueling adoption across China, India, Japan, and South Korea. Governments across the region are investing heavily in precision medicine programs, national genomics initiatives, and modernization of research laboratories.
Expanding academic research capabilities and increasing collaborations with global pharmaceutical companies are accelerating demand for miRNA sequencing technologies. Cost-sensitive markets are also encouraging adoption of scalable and locally manufactured consumables and assay solutions. In addition, rising clinical trial activity and outsourcing of genomics research to regional CROs are strengthening market growth. Growing integration of sequencing platforms with digital bioinformatics tools and increasing availability of skilled genomics professionals further support long-term expansion across the Asia Pacific region.
Market Competitive Landscape
The global miRNA sequencing and assay market is highly competitive, with strong participation from companies such as Thermo Fisher Scientific Inc., Illumina, QIAGEN, PerkinElmer, and Abcam plc. These players leverage extensive global distribution networks, strong brand recognition, and continuous innovation across sequencing chemistries, library preparation workflows, bioinformatics platforms, and assay sensitivity to address a broad range of research and clinical applications.
Rising demand for oncology diagnostics, biomarker discovery, and precision medicine, along with increasing adoption of next-generation sequencing in translational research, is driving product innovation and portfolio expansion. Manufacturers are increasingly focusing on high-throughput, cost-efficient sequencing solutions, improved data accuracy, and integrated bioinformatics pipelines. Strategic priorities include strengthening collaborations with academic and clinical research centers, expanding presence in emerging genomics markets, and investing in R&D to support advanced miRNA profiling solutions and sustain long-term market growth.
Key Industry Developments:
- In April 2025, Novartis announced the acquisition of Regulus Therapeutics, gaining access to farabursen, an investigational microRNA inhibitor being developed for the treatment of autosomal dominant polycystic kidney disease (ADPKD), the most common inherited cause of renal failure. This development highlights the growing clinical and commercial relevance of microRNA-based therapeutics, reinforcing the importance of advanced miRNA sequencing and assay technologies for target identification, biomarker validation, and therapy monitoring.
- In February 2025, Roche introduced its proprietary sequencing by expansion (SBX) technology, a next-generation sequencing approach that delivers ultra-rapid, high-throughput, and scalable performance through novel chemistry and an advanced sensor module. This innovation supports the miRNA sequencing and assay market by enabling faster, more efficient small RNA profiling, improving throughput and flexibility for applications such as biomarker discovery, oncology research, and precision medicine workflows.
Companies Covered in miRNA Sequencing and Assay Market
- Thermo Fisher Scientific Inc.
- Illumina
- QIAGEN
- PerkinElmer
- Abcam plc
- New England Biolabs
- Takara Bio Inc.
- Lexogen GmbH
- Norgen Biotek Corp
- Maravai LifeSciences
- HTG Molecular Diagnostics
- Meridian Bioscience, Inc.
- System Biosciences, LLC
Frequently Asked Questions
The global miRNA sequencing and assay market is projected to be valued at US$ 384.6 Mn in 2026.
The market is driven by growing demand for precision medicine and personalized diagnostics, continuous advancements in next-generation sequencing technologies and bioinformatics, rising prevalence of chronic diseases (especially cancer) that require detailed biomarker profiling, and increased research and investment in genomics and biomarker discovery.
The global miRNA sequencing and assay market is poised to witness a CAGR of 5.8% between 2026 and 2033.
Expanding applications in non-invasive diagnostics and precision medicine (including liquid biopsies and companion diagnostics) present significant growth opportunities.








