- Pharmaceuticals
- Heparin-Induced Thrombocytopenia Treatment Market
Heparin-Induced Thrombocytopenia Treatment Market Size, Share, and Growth Forecast, 2025 - 2032
Heparin-Induced Thrombocytopenia Treatment Market by Drug Type (Lepirudin, Danaparoid, Argatroban), Test Type (Enzyme Immunoassay (EIA), Serotonin Release Assay (SRA), Enzyme-Linked Immunosorbent Assay (ELISA), Heparin-Induced Platelet Activation (HIPA) Test), End-use, and Regional Analysis for 2025 - 2032
Heparin-Induced Thrombocytopenia Treatment Market Size and Trend Analysis
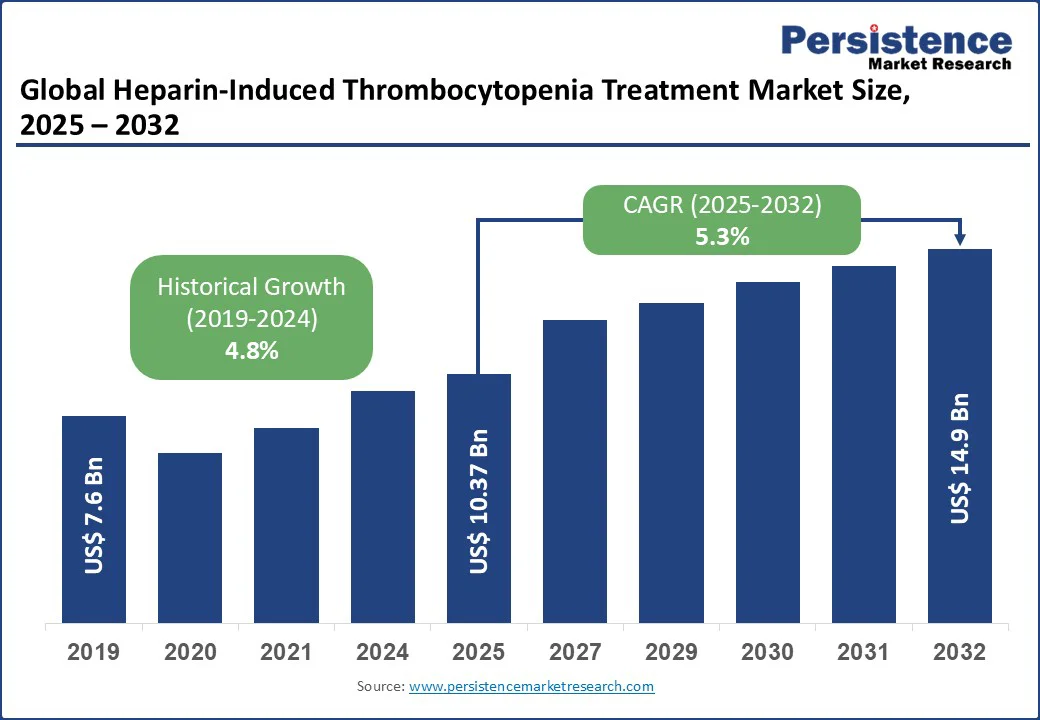
The global heparin-induced thrombocytopenia (HIT) treatment market size is likely to value at US$10.37 Bn in 2025 and reach US$14.9 Bn by 2032, growing at a CAGR of 5.3% during the forecast period from 2025 to 2032.
The heparin-induced thrombocytopenia treatment market is witnessing steady growth, driven by increasing demand from key healthcare sectors such as hospitals and specialized clinics, where rapid diagnosis and effective management of HIT are critical.
Heparin-Induced thrombocytopenia treatment, involving non-heparin anticoagulants such as Argatroban and advanced diagnostic assays is essential for managing this immune-mediated complication of heparin therapy. The rise in global surgical procedures, coupled with advancements in diagnostic technologies, supports market expansion.
Key Industry Highlights:
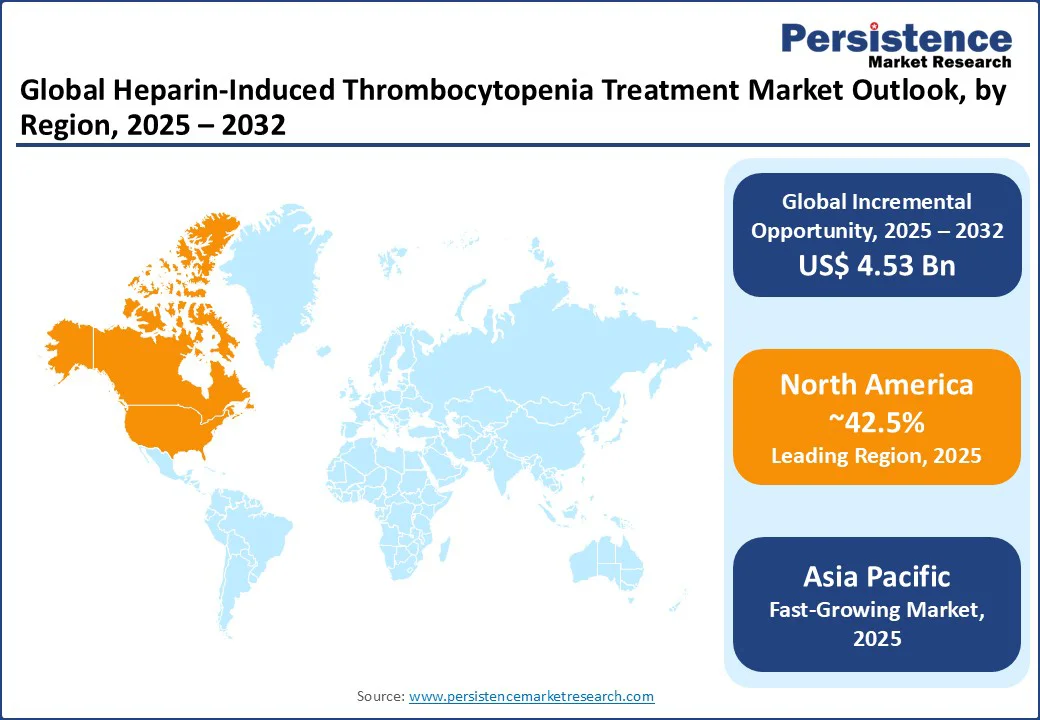
- Leading Region: North America holds 42.5% market share in 2025, driven by robust hospital infrastructure and advanced diagnostic capabilities in the U.S. and Canada, supported by high clinical awareness.
- Fastest-growing Region: Asia Pacific is the fastest-growing region, propelled by rapid healthcare infrastructure growth, increasing surgical volumes, and high heparin usage in countries such as China and India.
- Investment Plans: The National Health Commission (NHC) of China issued guidelines (May 2024) to enhance critical care medical services, aiming to provide 15 intensive care (ICU) beds per 100,000 people-in addition to 10 beds convertible to ICU-by the end of 2025, boosting demand for HIT treatments in surgical applications.
- Dominant Drug Type: Argatroban, accounting for nearly 43.8% of the market share, due to its efficacy and safety in managing acute HIT cases.
- Leading End-use: Hospitals, contributing over 40.2% of market revenue, driven by global increases in heparin-related procedures and critical care needs.
|
Global Market Attribute |
Key Insights |
|
Heparin-Induced Thrombocytopenia Treatment Market Size (2025E) |
US$ 10.37Bn |
|
Market Value Forecast (2032F) |
US$ 14.9Bn |
|
Projected Growth (CAGR 2025 to 2032) |
5.3% |
|
Historical Market Growth (CAGR 2019 to 2024) |
4.8% |
Market Dynamics
Driver: Rising Surgical Volumes and Heparin Usage Fuel Market Expansion
The global Heparin-Induced Thrombocytopenia (HIT) treatment market is gaining strong momentum, primarily driven by the increasing number of surgical procedures and the extensive use of heparin as an anticoagulant.
Heparin is a standard drug in surgeries such as cardiac bypass, dialysis, and venous thromboembolism (VTE) management, all of which contribute to a growing pool of patients at risk for HIT-a serious immune-mediated reaction to heparin. In the U.S. alone, more than 600,000 percutaneous coronary interventions (PCIs) and nearly 400,000 coronary artery bypass grafts (CABGs) are performed annually.
In Asia Pacific, countries such as China and India are fueling demand through major public health initiatives. China’s National Health Commission plans to expand intensive care services significantly by 2025 while India’s National Health Mission (NHM) continues to scale access to critical care, boosting heparin administration in surgical and ICU settings. Additionally, Pfizer reported a rise in Argatroban sales in 2024, underscoring increased HIT diagnosis and management.
Restraint: High Costs of Advanced Diagnostics and Competition from Alternative Anticoagulants
The heparin-induced thrombocytopenia treatment market faces challenges due to the high costs of advanced diagnostic assays and competition from alternative anticoagulants. Diagnostic tools such as enzyme-linked Immunosorbent assays (ELISA) and serotonin release assays (SRA) require specialized equipment and skilled personnel, increasing operational costs. In 2023, diagnostic assay costs fluctuated, impacting affordability for smaller healthcare providers. This cost burden limits adoption, particularly in resource-constrained regions.
Additionally, direct oral anticoagulants (DOACs), such as rivaroxaban, are gaining traction due to their ease of use and reduced monitoring needs. Limited awareness in some regions and concerns over assay availability in harsh healthcare environments further hinder adoption, particularly in cost-sensitive markets, restraining overall market growth.
Opportunity: Rising Demand in Precision Medicine and Novel Anticoagulant Development
The increasing focus on precision medicine and novel anticoagulant development presents significant opportunities for the Heparin-Induced Thrombocytopenia Treatment market. Advanced therapies, such as bivalirudin and next-generation direct thrombin inhibitors, are critical for personalized HIT management in critical care settings.
In the hospital sector, HIT treatments are used in tailored protocols for high-risk patients. Companies such as Veralox Therapeutics are innovating with drugs such as VLX-1005 for HIT applications, aligning with personalized medicine trends.
Government incentives, such as the EU’s Horizon Europe program, further encourage investments in novel therapies, creating opportunities for manufacturers to develop advanced, patient-specific Heparin-Induced Thrombocytopenia Treatments to meet evolving healthcare needs through 2032.
Category-wise Analysis
By Drug Type
- Argatroban holds the largest market share, approximately 43.8% in 2025, due to its high efficacy and safety profile in managing acute HIT. Widely used in hospitals and specialized clinics, argatroban is favored for its rapid onset and reversibility in critical care settings. Companies such as Pfizer and Fresenius Kabi lead with extensive portfolios, catering to demand in the surgical and dialysis sectors across North America and the Asia Pacific.
- Danaparoid is the fastest-growing drug type segment, driven by its increasing adoption in regions with limited access to argatroban. Its versatility in managing HIT in various patient demographics, such as pediatric and elderly populations, makes it ideal for specialized clinics. Brands such as Aspen Pharmacare are expanding offerings in Europe and the Asia Pacific, supported by growing demand for cost-effective, non-heparin anticoagulants.
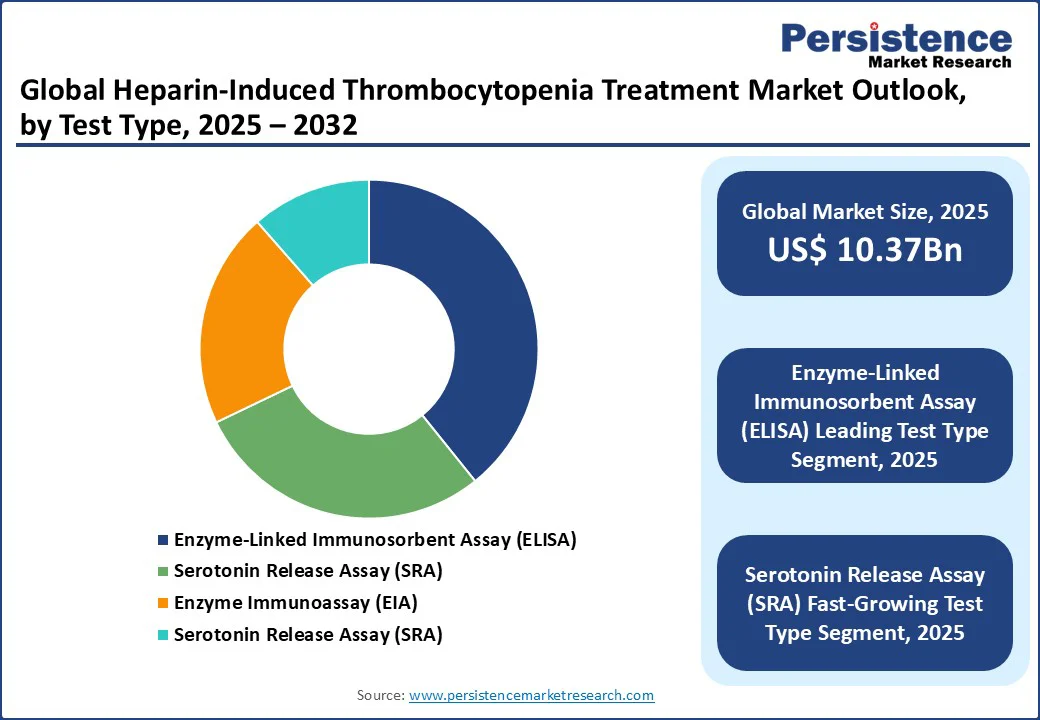
By Test Type
- Enzyme-Linked Immunosorbent Assay (ELISA) accounts for over 34.2% of market revenue in 2025, driven by its high sensitivity and widespread use in hospital diagnostics. ELISA tests are critical for detecting Heparin-Induced Thrombocytopenia Treatment antibodies in high-risk patients, with major players such as Sandoz supplying reliable assays for projects in the U.S. and China, where healthcare investments fuel demand.
- Serotonin Release Assay (SRA) is the fastest-growing test type, propelled by its superior specificity in confirming HIT diagnoses. SRAs are increasingly used in specialized clinics and research settings, with companies such as Diapharma innovating for high-performance applications. Growth in the Asia Pacific and Europe, driven by diagnostic advancements, supports this segment’s rapid expansion.
By End-use
- Hospitals account for over 40.2% of market revenue in 2025, driven by global increases in heparin-related procedures and critical care needs. Heparin-Induced Thrombocytopenia Treatments are critical for managing complications in surgical and ICU settings. Major players such as Fresenius Kabi supply advanced therapies for projects in the U.S. and China, where healthcare infrastructure investments fuel demand.
- Specialized Clinics are the fastest-growing end-use, propelled by rising investments in targeted HIT management and outpatient care. These clinics use advanced diagnostics and therapies for high-risk patients, with companies such as Veralox Therapeutics innovating for precision applications. Growth in the Asia Pacific and Europe, driven by outpatient care trends, supports this segment’s rapid expansion.
Regional Insights
North America Heparin-Induced Thrombocytopenia Treatment Market Trends
North America dominates the Heparin-Induced Thrombocytopenia Treatment market, accounting for 42.5%, driven by robust demand from hospital and diagnostic sectors in the U.S. and Canada, supported by advanced healthcare infrastructure. The U.S. healthcare industry reached $4.9 trillion, accounting for 17.6% of GDP in 2023 relies heavily on HIT treatments for surgical and critical care applications.
Canada’s hospital sector drives demand for advanced diagnostics, per the Canadian Medical Association. Major players such as Pfizer and Eagle Pharmaceuticals dominate with extensive distribution networks, catering to healthcare projects such as ICU expansions and cardiac care upgrades. Consumer preference for high-quality, rapid diagnostics further strengthens North America’s market position.
Asia Pacific Heparin-Induced Thrombocytopenia Treatment Market Trends
Asia Pacific is poised to achieve a fastest-growth, fueled by rapid healthcare infrastructure growth, increasing surgical volumes, and high heparin usage in countries such as China and India. China, a major pharmaceutical hub, contributes significantly to global heparin production, per the World Health Organization, driving the availability of Heparin-Induced Thrombocytopenia Treatment. \
India’s healthcare sector, supported by initiatives such as the National Health Mission, boosts demand for hospital-grade therapies. The region’s dialysis and cardiac surgery industries also contribute, with companies such as ACG Group and Syntegon Technology expanding their presence. Rising healthcare manufacturing and government-led projects ensure the Asia Pacific’s rapid market growth through 2032.
Europe Heparin-Induced Thrombocytopenia Treatment Market Trends
Europe is the second fastest-growing region for the Heparin-Induced Thrombocytopenia Treatment market, driven by stringent regulatory standards, rising demand in hospital and specialized clinic sectors, and healthcare development in countries such as Germany and France. In 2022, the European Union (EU) allocated approximately €1.3 trillion to current healthcare expenditure, equating to about 10.4% of its Gross Domestic Product (GDP), which supports demand for HIT treatments in surgical and diagnostic applications.
Germany’s hospital sector, a key consumer of advanced assays, benefits from players such as Korber AG and Marchesini Group. The EU’s Horizon Europe program promotes precision medicine projects, increasing demand for novel anticoagulants in critical care settings. Europe’s focus on regulatory compliance and high-quality standards drives market growth, with companies innovating to meet clinical and patient demands.
Competitive Landscape
The global heparin-induced thrombocytopenia treatment market is highly competitive, dominated by extensive product portfolios and global distribution networks. The heparin-induced thrombocytopenia treatment market is characterized as a fragmented market based on competitors due to the presence of numerous domestic and international players, ranging from large, established companies to smaller, regional manufacturers.
Regional players such as ACG Group focus on localized offerings in the Asia Pacific. Companies are investing in advanced diagnostic technologies and novel anticoagulants to enhance market share, driven by demand for high-performance treatments in hospital and specialized clinic sectors.
Industry Developments:
- May 2025: Sanofi completed the acquisition of DR-0201, a targeted bispecific myeloid cell engager from Dren Bio. This acquisition aims to bolster Sanofi's capabilities in treating autoimmune diseases, which may intersect with conditions such as HIT. The acquisition includes an upfront payment and potential future milestone payments, reflecting the strategic importance of this addition to Sanofi's portfolio.
- June 2024: Veralox Therapeutics launched VLX-1005, a next-generation direct thrombin inhibitor for HIT treatment, strengthening its position in North America and Europe with an FDA fast-track designation. This enzyme plays a critical role in platelet activation and thrombosis, making VLX-1005 a promising candidate for treating Heparin-Induced Thrombocytopenia (HIT).
Companies Covered in Heparin-Induced Thrombocytopenia Treatment Market
- ACG Group, Inc.
- Korber AG
- Romaco Group
- Marchesini Group S.p.A.
- Coesia S.p.A.
- Multivac Group
- Syntegon Technology GmbH
- Bausch + Strobel
- OPTIMA industries GmbH & Co. KG.
- Industria Machine Automatiche S.p.A.,
- Others
Frequently Asked Questions
The Heparin-induced Thrombocytopenia Treatment market is projected to reach US$10.37 Bn in 2025.
Rising surgical volumes and heparin usage, along with expanding applications in precision medicine, are the key market drivers.
The Heparin-induced Thrombocytopenia Treatment market is poised to witness a CAGR of 5.3% from 2025 to 2032.
The rising demand in precision medicine and novel anticoagulant development is the key market opportunity.
ACG Group, Inc., Korber AG, Romaco Group, Marchesini Group S.p.A., and Coesia S.p.A. are key market players.








