- Biotechnology
- Cognitive Impairment Biomarkers Market
Cognitive Impairment Biomarkers Market Size, Share, and Growth Forecast, 2026 - 2033
Cognitive Impairment Biomarkers Market by Biomarker Type (Genomics Biomarker, Proteomics Biomarker, Metabolomics Biomarkers, Others), Application (Alzheimer's Diseases, Parkinson's Diseases, Others), and Regional Analysis for 2026 - 2033
Cognitive Impairment Biomarkers Market Size and Trends Analysis
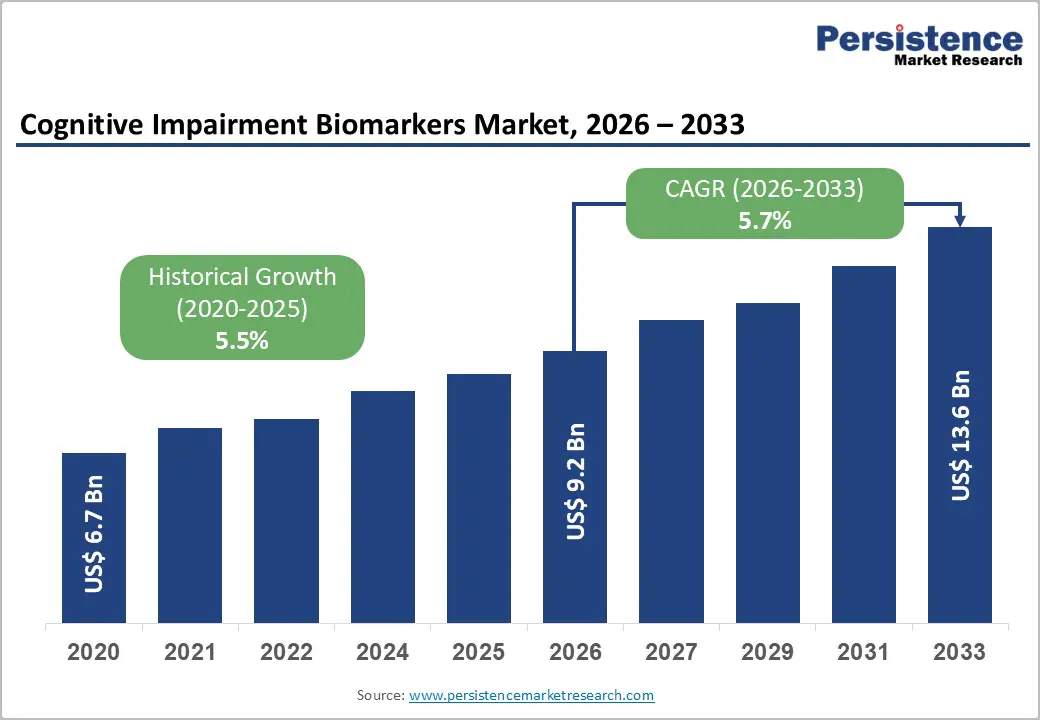
The global cognitive impairment biomarkers market size is likely to be valued at US$9.2 billion in 2026 and is expected to reach US$13.6 billion by 2033, growing at a CAGR of 5.7% during the forecast period from 2026 to 2033, driven by the growing clinical and economic burden of neurodegenerative and neuropsychiatric disorders worldwide.
According to the World Health Organization (2025), more than three billion people worldwide live with neurological conditions, which collectively account for over 11 million deaths annually. Dementia alone affects approximately 57 million individuals, with nearly 10 million new cases each year, and Alzheimer's disease represents the majority of cases. Increasing collaboration among diagnostic companies, academic institutions, and biopharmaceutical firms is accelerating translational research and commercialization of advanced biomarker panels.
Key Industry Highlights:
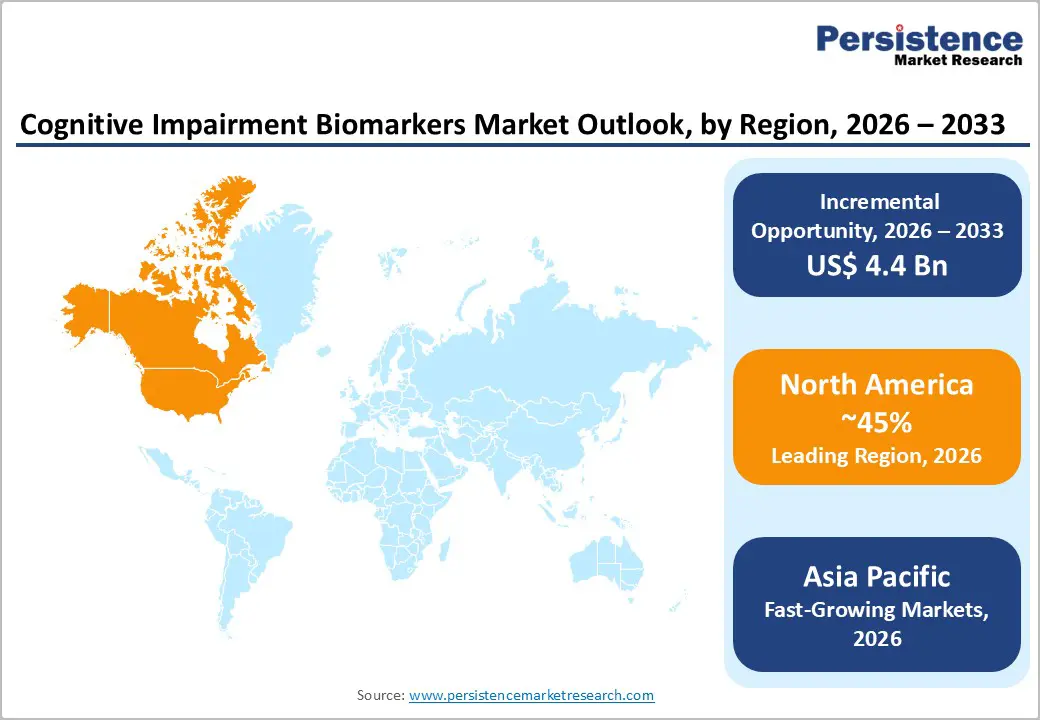
- Leading Region: North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by strong regulatory support, advanced diagnostic infrastructure, robust research funding, and early adoption of innovative biomarker technologies.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region in the cognitive impairment biomarkers in 2026, supported by accelerating healthcare investments, demographic aging, and growing regional innovation capabilities.
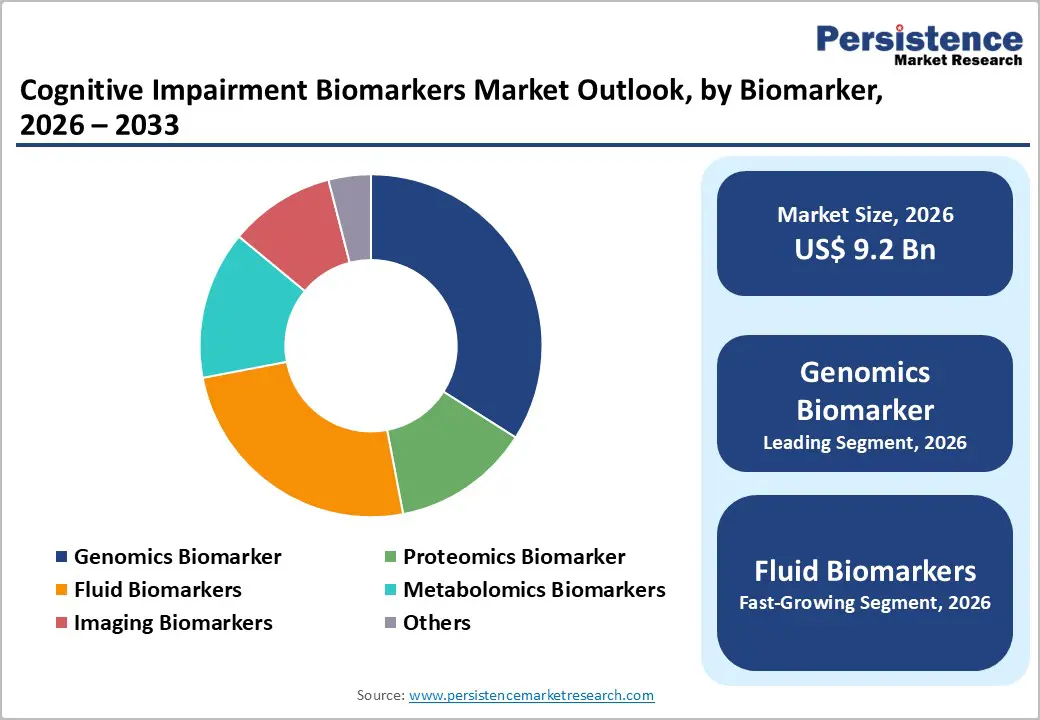
- Leading Biomarker Type: Genomics is projected to represent the leading biomarker type in 2026, accounting for 28% of the revenue share, driven by expanding genetic risk profiling and increasing adoption of precision medicine in neurodegenerative disease management.
- Leading Application: Alzheimer’s disease is expected to be the leading application, accounting for over 34% of revenue share in 2026, driven by high dementia prevalence and strong clinical adoption of validated amyloid and tau biomarkers.
| Key Insights | Details |
|---|---|
| Cognitive Impairment Biomarkers Market Size (2026E) | US$9.2 Bn |
| Market Value Forecast (2033F) | US$13.6 Bn |
| Projected Growth (CAGR 2026 to 2033) | 5.7% |
| Historical Market Growth (CAGR 2020 to 2025) | 5.5% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Growth Analysis - Rising Prevalence of Neurodegenerative Diseases Due to Demographic Shifts
Increasing life expectancy has led to a larger population aged 65 and above, the demographic most vulnerable to Alzheimer’s disease, Parkinson’s disease, and related cognitive disorders. As age is the strongest risk factor for neurodegeneration, healthcare systems are witnessing a steady rise in diagnosed and undiagnosed cases. This demographic shift intensifies demand for early diagnostic tools capable of detecting pathological changes before irreversible neuronal damage occurs, strengthening the adoption of biomarker-based screening and monitoring approaches worldwide.
Growing disease prevalence also increases the economic burden on healthcare systems, families, and caregivers, encouraging governments and private stakeholders to prioritize early intervention strategies. Biomarkers enable identification of at-risk individuals during preclinical stages, supporting preventive care and therapeutic planning. Pharmaceutical companies are increasingly relying on biomarkers for patient stratification in clinical trials, improving treatment response rates. As populations continue aging across North America, Europe, and the Asia Pacific, sustained epidemiological pressure will reinforce long-term demand for validated and scalable cognitive impairment biomarker solutions.
Technological Advancements in Multi-Omics and Imaging Platforms
Rapid progress in genomics, proteomics, and metabolomics technologies has significantly enhanced the sensitivity and specificity of biomarkers for cognitive impairment. High-throughput sequencing, advanced mass spectrometry, and multiplex immunoassays now enable simultaneous detection of multiple biological signatures associated with neurodegeneration. These platforms generate comprehensive molecular insights, improving early diagnosis and disease differentiation. Imaging innovations, including advanced MRI and PET modalities, complement molecular biomarkers by providing structural and functional visualization, strengthening clinical confidence in diagnosis and therapeutic monitoring.
Technological convergence is also reducing reliance on invasive procedures by enabling blood-based biomarker assays with accuracy comparable to cerebrospinal fluid testing and imaging methods. Automation and digital integration improve reproducibility and scalability across laboratories. Artificial intelligence algorithms increasingly assist in interpreting complex multi-omics datasets, enhancing predictive accuracy. These advancements not only accelerate clinical adoption but also support pharmaceutical research and precision medicine initiatives, positioning multimodal biomarker platforms as foundational components of modern neurodiagnostic frameworks.
Barrier Analysis - Lack of Standardization across Biomarker Platforms and Populations
One of the key restraints in the cognitive impairment biomarkers market is the absence of standardized validation protocols across testing platforms and diverse patient populations. Variability in assay methodologies, sample handling procedures, and reference thresholds can lead to inconsistent results between laboratories. Such discrepancies limit cross-study comparability and hinder broader regulatory approval. Differences in genetic backgrounds, environmental exposures, and comorbidities across populations complicate universal biomarker interpretation, slowing widespread clinical integration.
Multi-center clinical trials often encounter challenges in harmonizing data collection and analytical methods. Without standardized guidelines, healthcare providers may hesitate to adopt novel assays due to concerns regarding reliability and reproducibility. This lack of uniformity increases validation costs and extends commercialization timelines for diagnostic developers. Addressing standardization gaps through international regulatory collaboration and consensus-driven frameworks is essential to unlock the full potential of cognitive impairment biomarkers across diverse healthcare settings.
Data Privacy and Ethical Concerns in Multi-Omics Datasets
The increasing use of genomics and multi-omics datasets in cognitive impairment diagnostics raises significant data privacy and ethical considerations. Biomarker research involves highly sensitive genetic and clinical information, requiring strict compliance with data protection regulations. Concerns regarding unauthorized access, data misuse, and long-term storage security can discourage patient participation in large-scale studies. Ethical debates surrounding predictive testing for neurodegenerative diseases, particularly when no definitive cure exists, complicate widespread screening initiatives.
Cross-border data sharing for collaborative research also faces regulatory complexities due to varying privacy laws. Institutions must invest heavily in secure infrastructure and governance frameworks to maintain compliance, increasing operational costs. Ethical concerns related to genetic discrimination and informed consent procedures slow the adoption of advanced biomarker programs. Balancing innovation with responsible data management practices remains critical to sustaining trust and ensuring ethical growth within the cognitive impairment biomarkers market.
Opportunity Analysis - Technological Convergence with Digital Health and AI Analytics
The convergence of cognitive impairment biomarkers with digital health platforms and artificial intelligence is reshaping neurological diagnostics from episodic testing to continuous, data-driven disease management. This multi-modal integration improves risk stratification, predicts disease progression timelines, and enhances differential diagnosis between closely related neurodegenerative conditions. AI-enabled algorithms also refine biomarker threshold calibration, reducing false positives and improving clinical reliability across diverse populations. Beyond laboratory diagnostics, digital biomarkers derived from wearable sensors, smartphone-based cognitive assessments, speech analysis, and behavioral tracking add real-world functional data to biological signatures.
Combining molecular and digital biomarkers enables earlier detection of cognitive decline, sometimes before noticeable clinical symptoms emerge. This approach supports proactive intervention strategies and personalized therapeutic adjustments. In clinical research, decentralized trials powered by remote monitoring tools expand patient recruitment and improve longitudinal data collection, particularly for elderly or mobility-limited populations. From a commercial perspective, technology convergence fosters scalable, subscription-based diagnostic ecosystems rather than single-test revenue models. Cloud-based analytics platforms allow laboratories and healthcare systems to access centralized AI interpretation engines, improving interoperability and reducing infrastructure costs.
Addressing Unmet Needs in Underserved Applications and Personalized Therapy
Disorders such as Parkinson’s disease, multiple sclerosis, and schizophrenia lack fully validated objective diagnostic tools, creating demand for novel molecular markers. Biomarker-guided stratification can enable earlier intervention and more precise therapeutic targeting, addressing longstanding gaps in neurological care and improving patient outcomes across diverse disease categories. The expansion of alpha-synuclein, neurofilament light chain, inflammatory cytokine, and metabolomic signatures is strengthening diagnostic confidence in these conditions. Increasing research grants and cross-institutional collaborations are accelerating validation studies, improving clinical acceptance beyond traditional Alzheimer’s-focused applications.
Personalized therapy development amplifies this opportunity, as pharmaceutical companies increasingly require companion diagnostics to optimize treatment selection. Biomarkers can identify responders to disease-modifying therapies and monitor therapeutic efficacy in real time. This approach enhances clinical trial efficiency by improving patient selection and reducing attrition rates, ultimately lowering development costs. Emerging markets with expanding healthcare infrastructure also represent untapped potential for accessible, cost-effective biomarker solutions, particularly as governments prioritize early neurological screening and precision medicine frameworks.
Category-wise Analysis
Biomarker Type Insights
Genomics biomarkers are expected to lead the cognitive impairment biomarkers market, accounting for approximately 28% of revenue in 2026, driven by strong clinical adoption of genetic risk profiling in neurodegenerative disease assessment. Their leadership is driven by the integration of inherited risk markers into early screening protocols, particularly in Alzheimer’s disease, where genetic susceptibility testing enhances preventive care planning and research enrollment. For example, APOE genotyping is widely used to assess genetic predisposition and support patient stratification in both clinical practice and therapeutic trials. Genomics platforms benefit from high analytical sensitivity, scalability, and compatibility with blood or saliva samples, enabling broader accessibility across research laboratories and specialty clinics.
Fluid biomarkers are likely to represent the fastest-growing segment in 2026, supported by advancements in minimally invasive blood-based diagnostics that improve accessibility and patient compliance. Growth is fueled by increasing clinical validation and regulatory support for assays capable of detecting disease-specific protein signatures in peripheral blood. For example, plasma-based amyloid and phosphorylated tau testing has demonstrated diagnostic performance comparable to advanced neuroimaging in identifying Alzheimer’s pathology. These innovations reduce dependence on invasive cerebrospinal fluid procedures and costly imaging infrastructure, expanding adoption across community healthcare settings.
Application Insights
Alzheimer’s disease is projected to lead the market, capturing around 34% of the revenue share in 2026, supported by its large patient population and extensive biomarker validation. The dominance of this segment is supported by well-characterized pathological pathways involving amyloid beta and tau proteins, which have been incorporated into standardized diagnostic frameworks. For example, amyloid PET imaging and cerebrospinal fluid tau assessment are both widely used in clinical trials and specialty neurology centers to confirm disease pathology. These validated tools have strengthened confidence among clinicians and regulators, embedding biomarker-guided diagnosis into routine practice.
Parkinson’s disease is likely to be the fastest-growing application in 2026, driven by the emergence of novel molecular targets and increasing therapeutic development activity. Growth is supported by expanding validation of biomarkers capable of detecting disease-specific neurodegenerative processes at earlier stages. For example, alpha-synuclein assays in Parkinson’s disease are advancing toward broader clinical adoption for diagnostic confirmation and progression monitoring. Neurofilament light chain biomarkers are gaining attention in multiple sclerosis for tracking neuronal damage and therapeutic response. These advancements address longstanding gaps in objective disease measurement, particularly in conditions where diagnosis has historically relied heavily on clinical observation.
Regional Insights
North America Cognitive Impairment Biomarkers Market Trends
North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by robust healthcare infrastructure, proactive regulatory frameworks, and a strong innovation ecosystem. The U.S. has been at the forefront of integrating biomarker-based diagnostics into clinical and research settings, supported by substantial funding from federal agencies and private organizations for neurodegenerative disease research. For example, Quanterix has gained prominence with its ultra-sensitive Simoa platform, enabling precise quantification of low-abundance proteins such as phosphorylated tau and neurofilament light chain in blood samples.
North America’s cognitive impairment biomarkers market trends reflect evolving reimbursement policies and increasing integration of biomarkers within therapeutic development. U.S. regulatory agencies have created expedited pathways for diagnostic tools that address unmet clinical needs, facilitating earlier market entry for novel assays with demonstrated clinical utility. This regulatory support, combined with strong health insurer reimbursement frameworks for validated tests, has improved clinician confidence in ordering biomarker diagnostics in both outpatient and inpatient settings.
Europe Cognitive Impairment Biomarkers Market Trends
Europe is likely to be a significant market for cognitive impairment biomarkers in 2026, due to well-established healthcare systems, significant public and private research investments, and collaborative frameworks that advance diagnostic development and clinical application. European countries are particularly influential due to robust funding mechanisms and integrated neurodegenerative disease initiatives that emphasize early detection and personalized care pathways. For example, Quibim, a Spanish biotechnology company that combines advanced imaging biomarkers with artificial intelligence to enhance quantitative brain analysis, contributes to improved diagnostic workflows for cognitive decline.
Europe’s cognitive impairment biomarkers market trends reflect expansion in regulatory support, strategic clinical partnerships, and targeted investment in translational research. Regulatory agencies, including the European Medicines Agency (EMA), are advancing pathways to qualify biomarkers as surrogate endpoints, which accelerates their use in clinical trials and therapy development. Public-private collaboration platforms and research initiatives are instrumental in standardizing biomarker data, harmonizing clinical methodologies, and improving cross-border study integration.
Asia Pacific Cognitive Impairment Biomarkers Market Trends
The Asia Pacific region is likely to be the fastest-growing region in 2026, driven by rising neurological disease prevalence, rapid demographic aging, and expanding healthcare investments. Countries such as China, Japan, India, and Southeast Asian nations are increasingly adopting advanced biomarker diagnostics for early detection and disease monitoring, reflecting greater clinical demand and research focus on neurological conditions. The region’s diverse population with significant incidence rates of Alzheimer’s and Parkinson’s disease has pushed clinical research institutions and diagnostic labs to integrate multi-modal biomarker platforms combining molecular testing and imaging to improve diagnostic precision.
Asia Pacific market is marked by increasing participation from regional diagnostic companies expanding their footprint and product offerings. For example, Thermo Fisher Scientific’s presence in the region has supported the availability of high-throughput immunoassays and proteomic platforms that enable sensitive detection of neurological biomarkers, facilitating broader clinical adoption. Investments in research and development, coupled with regional regulatory harmonization efforts, are improving the landscape for introducing new assays and diagnostic tools focused on neurodegenerative diseases.
Competitive Landscape
The global cognitive impairment biomarkers market exhibits a moderately fragmented structure, driven by a mix of large multinational diagnostics firms, specialized biotechnology innovators, and emerging precision medicine startups. Broad adoption of biomarker technologies across clinical, research, and pharmaceutical sectors has created space for a wide range of companies offering genomics, proteomics, metabolomics, imaging, and fluid-based solutions. Innovation is fueled by expanding neurological research funding, regulatory support for validated diagnostic assays, and increasing integration of digital health analytics.
With key leaders including Thermo Fisher Scientific, Quanterix, Qiagen, Olink Biosciences, Bio-Rad Laboratories Inc, and Abbott Laboratories actively driving product innovation and market expansion, the competitive landscape reflects both depth and diversity. These players compete through continuous R&D investments, expanded product portfolios, and collaborations with pharmaceutical companies to co-develop companion diagnostics for neurodegenerative therapies. Strategic mergers and acquisitions are common as firms seek to strengthen technological capabilities and enter new regional markets.
Key Industry Developments:
- In February 2026, researchers at Washington University in St. Louis, led by Suzanne E. Schindler, MD, PhD, and Kellen K. Petersen, PhD, demonstrated that a simple blood test measuring the protein p-tau217 can predict when Alzheimer’s symptoms are likely to begin roughly three to four years before memory loss appears. This approach uses levels of p-tau217 to estimate the age at which cognitive symptoms will start, offering a more affordable and accessible alternative to PET imaging or cerebrospinal fluid analysis and potentially expediting preventive drug trials and early intervention strategies.
- In February 2026, Quanterix announced it submitted a 510(k) premarket notification to the U.S. Food and Drug Administration (FDA) for a multi-analyte algorithmic blood test designed to help identify whether symptomatic individuals are likely to have amyloid brain plaques, a hallmark of Alzheimer’s disease. The test uses Quanterix’s ultra-sensitive Simoa® technology to measure a panel of five key biomarkers, including p-Tau 217, Aβ40, Aβ42, GFAP, and NfL, and combines them through an algorithm to provide a comprehensive assessment of disease biology.
- In May 2025, the U.S. Food and Drug Administration cleared the first blood test to help diagnose Alzheimer’s disease, marking a major milestone in cognitive impairment biomarkers. The Lumipulse G pTau217/β-Amyloid 1-42 Plasma Ratio test, developed by Fujirebio Diagnostics, measures the ratio of two proteins in plasma to indicate the presence of amyloid plaques in the brain, a hallmark of Alzheimer’s disease, offering a less invasive and more accessible alternative to traditional tests such as PET scans or cerebrospinal fluid analysis.
Companies Covered in Cognitive Impairment Biomarkers Market
- Quanterix
- Qiagen
- ADx NeuroSciences NV
- Merck KGaA
- Enrolmmun
- Thermo Fisher Scientific
- ACOBIOM
- Banyan Biomarkers Inc
- Olink Biosciences
- Bio-Rad Laboratories Inc
- Roche Diagnostics
- Siemens Healthineers
- Abbott Laboratories
- GE Healthcare
- Fujifilm Holdings Corporation
Frequently Asked Questions
The global cognitive impairment biomarkers market is projected to reach US$9.2 billion in 2026.
The cognitive impairment biomarkers market is driven by the rising prevalence of neurodegenerative diseases, increasing demand for early and accurate diagnosis, and advancements in minimally invasive multi-omics and blood-based diagnostic technologies.
The cognitive impairment biomarkers market is expected to grow at a CAGR of 5.7% from 2026 to 2033.
Key market opportunities lie in the expansion of blood-based and AI-integrated biomarker platforms, the development of companion diagnostics for personalized therapies, and growing adoption in underserved neurological applications beyond Alzheimer’s disease.
Quanterix, Qiagen, ADx NeuroSciences NV, Merck KGaA, Enrolmmun, Thermo Fisher Scientific, and Banyan Biomarkers Inc. are the leading players.








