- Pharmaceuticals
- Duchenne Muscular Dystrophy Treatment Market
Duchenne Muscular Dystrophy Treatment Market Size, Share, Growth, and Regional Forecast, 2026 to 2033
Duchenne Muscular Dystrophy Treatment Market by Treatment Type (Molecular Therapies, Steroid Therapy, Others), Route of Administration (Oral, Intravenous), End User (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and Regional Analysis from 2026 to 2033
Duchenne Muscular Dystrophy Treatment Market Share and Trends Analysis
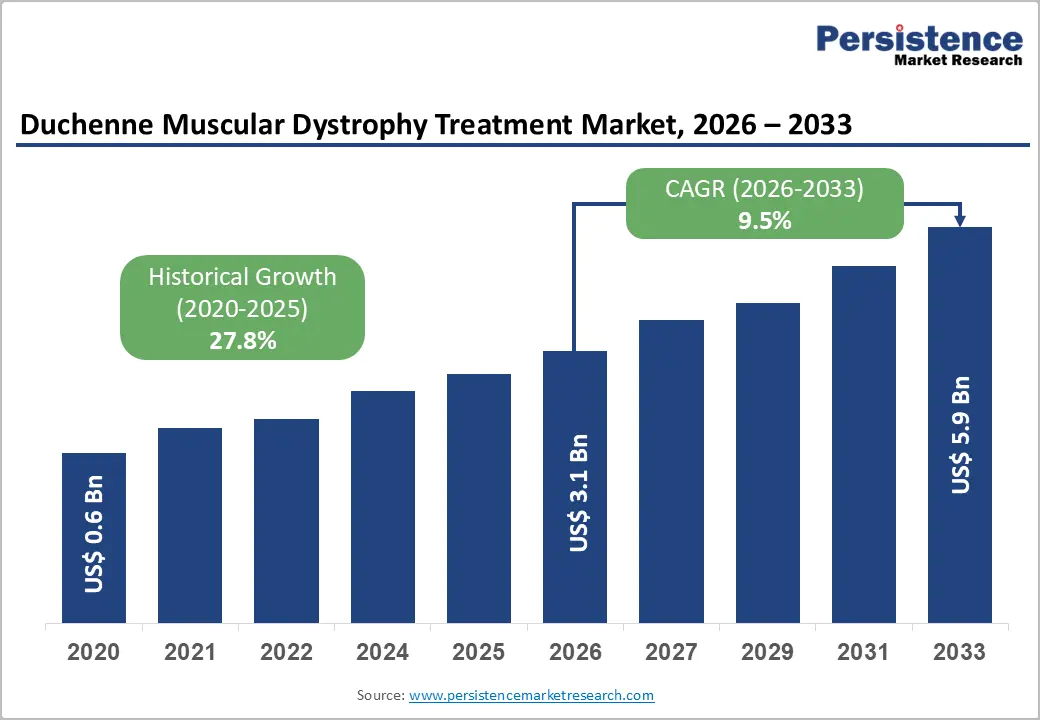
The global duchenne muscular dystrophy treatment market is estimated to grow from US$ 3.1 billion in 2026 to US$ 5.9 billion by 2033, recording a CAGR of 9.5% during the forecast period from 2026 to 2033.
It is witnessing steady growth, driven by increasing disease awareness, advancements in gene and molecular therapies, and rising investment in rare disease research. North America leads due to strong regulatory support and innovation, while Asia-Pacific is the fastest-growing region, supported by improving healthcare infrastructure and diagnosis rates.
Key Industry Highlights
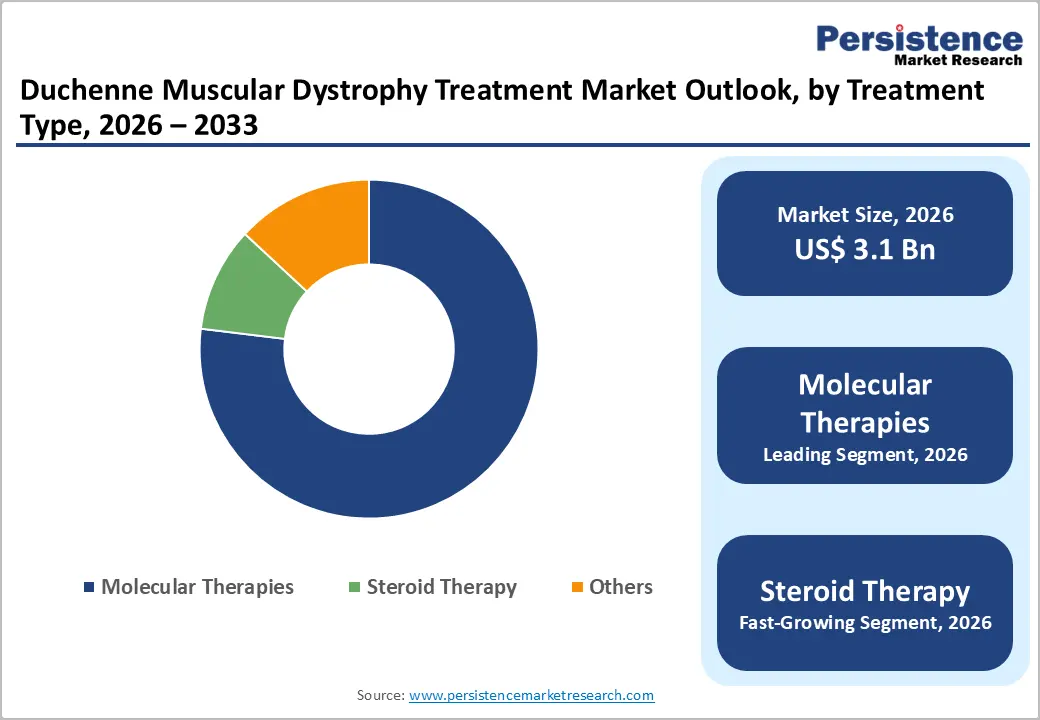
- Dominant Treatment Type: Molecular therapies, particularly exon skipping and gene therapy, held 76.9% share in 2025, driven by targeted treatment efficacy and increasing regulatory approvals.
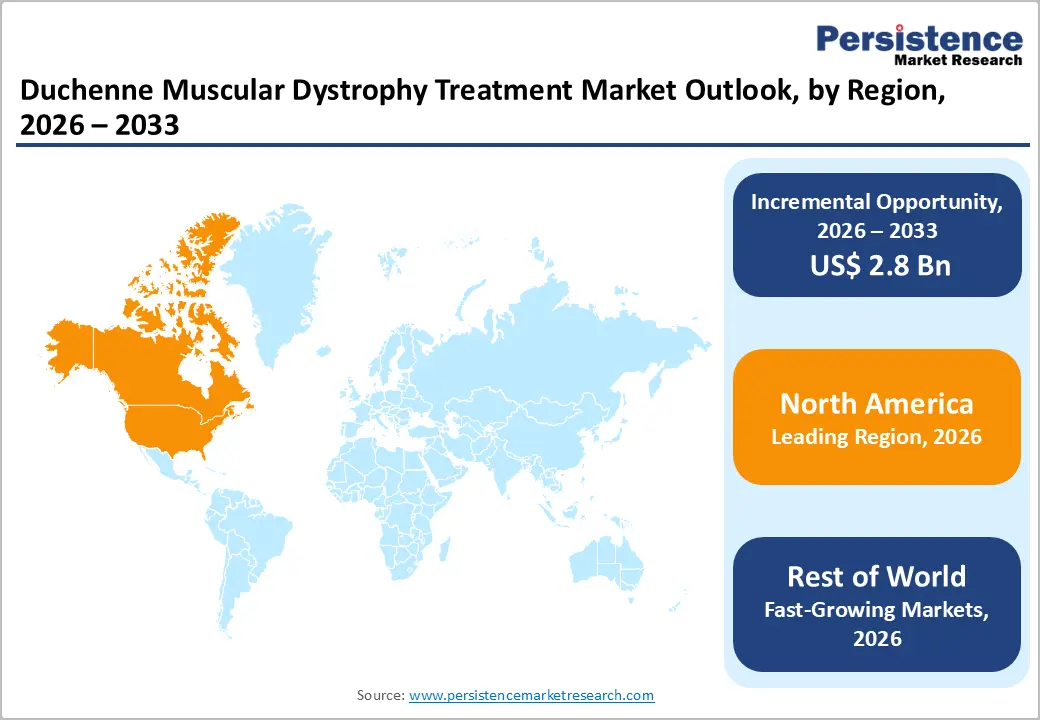
- Regional Leadership: North America led the market in 2025 with 83.0% share, supported by strong R&D investments, advanced healthcare infrastructure, favorable reimbursement, and early adoption of innovative therapies.
- Growth Indicators: Growth is driven by rising DMD prevalence, increasing awareness, advancements in gene therapy, supportive regulatory frameworks, and growing investment in rare disease research.
- Opportunity: Opportunities lie in expanding gene therapies, emerging markets, pipeline innovations, improved diagnostic rates, and strategic collaborations for advanced treatment development.
| Key Insights | Details |
|---|---|
| Duchenne Muscular Dystrophy Treatment Market Size (2026E) | US$ 3.1 Bn |
| Market Value Forecast (2033F) | US$ 5.9 Bn |
| Projected Growth (CAGR 2026 to 2033) | 9.5% |
| Historical Market Growth (CAGR 2020 to 2025) | 27.8% |
Market Dynamics
Driver: Growing Adoption of Corticosteroids and Supportive Care Regimens as Standard Background Therapy
Supportive care strategies play a complementary role by enhancing treatment outcomes and addressing disease complications. Multidisciplinary approaches, including physiotherapy, nutritional management, orthotic support, and regular cardiac and pulmonary monitoring, are essential for maintaining patient function and quality of life. Clinical guidelines consistently emphasize the importance of combining corticosteroid therapy with functional assessments such as the 6-minute walk test to monitor disease progression and guide treatment decisions.
This well-established treatment framework supports the integration of advanced therapies, including exon-skipping and gene therapies, alongside existing regimens. It also facilitates earlier diagnosis and treatment initiation, expanding the eligible patient pool.
Moreover, increasing adoption in emerging markets, along with improved adherence and combination-therapy strategies, is expected to drive sustained growth in the global DMD therapeutics market.
Restraint: Clinical Setbacks in Gene Therapy Development Limiting Market Confidence and Commercialization Potential
Clinical setbacks in gene therapy development are emerging as a significant restraint in the Duchenne Muscular Dystrophy (DMD) treatment market, highlighting the high-risk nature of advanced therapeutic approaches. Despite strong early-stage promise, several late-stage failures have raised concerns regarding efficacy, safety, and long-term outcomes. These challenges are particularly critical in DMD, where therapies aim to deliver functional dystrophin but face complexities in consistent expression and clinical benefit.
A key example is Pfizer’s investigational gene therapy CIFFREO, which failed to meet its primary and secondary endpoints in a Phase 3 trial, showing no meaningful improvement in motor function compared to placebo. Additionally, a patient's death due to cardiac arrest in an earlier-stage trial led to dosing pauses and heightened safety concerns. Following these outcomes, Pfizer discontinued its DMD gene therapy program, representing a major setback for the field.
Similarly, other companies have faced pipeline challenges, with firms like PepGen discontinuing development of their experimental therapy after failing to achieve sufficient dystrophin production in mid-stage trials. These repeated clinical failures underscore the uncertainty surrounding gene therapy efficacy and scalability, ultimately impacting investor confidence, delaying commercialization timelines, and limiting broader adoption of gene-based treatments in the DMD market.
Opportunity: Expansion of Gene Therapy into Early-Stage Patients Creating Long-Term Value
The development of next-generation therapies, particularly gene-editing technologies such as CRISPR, represents a transformative opportunity for Duchenne Muscular Dystrophy (DMD) treatment. Unlike traditional therapies that manage symptoms or partially restore protein function, gene editing aims to directly correct mutations in the dystrophin gene, addressing the disease's root cause. This approach holds the potential for long-term or even curative outcomes.
Ongoing research by biotechnology companies and academic institutions is focused on improving the safety, precision, and delivery mechanisms of these therapies. Innovations in viral vectors and non-viral delivery systems are helping overcome key challenges such as immune response and off-target effects. For instance, Sarepta Therapeutics is actively investing in next-generation platforms, including gene-editing and enhanced delivery constructs, to improve the durability of dystrophin expression and minimize immune-related complications in DMD patients.
As clinical and preclinical programs advance, these therapies are expected to demonstrate greater durability than first-generation gene therapies. From a market perspective, next-generation gene editing therapies could significantly reshape the treatment landscape by offering one-time treatments with sustained benefits. This not only enhances patient quality of life but also creates high-value opportunities for manufacturers through premium pricing models, stronger clinical differentiation, and long-term value generation in the evolving DMD therapeutics market.
Category-wise Analysis
By Treatment Type Insights
Molecular therapies dominate the DMD treatment market because they directly target the genetic root cause of the disease, the absence of dystrophin, unlike traditional symptomatic treatments. Regulatory approvals strongly support this shift: the U.S. FDA approved multiple exon-skipping drugs (e.g., eteplirsen, golodirsen) and the first gene therapy in 2023, marking a major therapeutic advancement. Exon-skipping approaches alone can collectively address up to ~60% of DMD patients depending on mutation type, significantly expanding their applicability. Clinically, these therapies restore partial dystrophin (up to ~5% expression in studies), which is enough to slow disease progression and improve motor outcomes. Additionally, DMD affects about 1 in 3,300 boys globally, creating strong demand for targeted, disease-modifying treatments rather than supportive care. This combination of regulatory momentum, broader patient coverage, and superior mechanism of action explains why molecular therapies dominate the segment.
By Route of Administration Insights
Intravenous (IV) administration dominates the route of administration segment primarily because most advanced DMD therapies, including exon-skipping drugs and gene therapies, require systemic delivery to reach widespread muscle tissues. FDA-approved exon-skipping drugs are administered through repeated IV infusions, ensuring adequate distribution across skeletal and cardiac muscles. Similarly, gene therapy such as Elevidys is delivered as a one-time intravenous infusion, enabling body-wide delivery of the therapeutic gene via viral vectors. From a pharmacokinetic perspective, IV administration ensures immediate bioavailability and controlled dosing, which is critical in a progressive disease affecting multiple organ systems. Furthermore, DMD involves both skeletal and cardiac muscle degeneration, necessitating systemic exposure that oral or localized routes cannot achieve effectively. The reliance of nearly all approved and pipeline advanced therapies on IV delivery reinforces its dominance in clinical practice and market share.
Regional Insights
North America Duchenne Muscular Dystrophy Treatment Market Trends
North America dominates the DMD treatment market primarily due to higher disease identification, strong healthcare systems, and early access to advanced therapies. Epidemiologically, the Americas show the highest muscular dystrophy prevalence at ~5.1 per 100,000 people, higher than Europe and other regions, indicating a larger treated population base. In the U.S., surveillance programs such as CDC’s MD STARnet report DMD prevalence of ~1 in 5,000 boys, supported by structured registries and early diagnosis systems. Additionally, North America has robust clinical trial infrastructure and regulatory support, accelerating approvals of gene and exon-skipping therapies. High healthcare expenditure and insurance coverage further improve treatment accessibility, reinforcing the region’s leadership in the adoption of advanced, high-cost therapies.
Europe Duchenne Muscular Dystrophy Treatment Market Trends
Europe is an important region in the DMD treatment market due to its strong rare-disease policy framework and widespread patient registries. The European Medicines Agency (EMA) estimates DMD prevalence at ~0.44-0.85 per 10,000 males, reflecting a well-documented patient population supported by national registries. Europe accounts for the majority of epidemiological studies globally, indicating a strong research focus and robust data infrastructure. Government-backed programs and cross-border collaborations under EU rare disease initiatives have standardized diagnosis and care pathways. Furthermore, universal healthcare systems in many countries improve access to genetic testing and long-term treatment. This combination of structured policy support, high diagnostic coverage, and coordinated research makes Europe a critical region for clinical development and therapy adoption.
Rest of World (ROW) Duchenne Muscular Dystrophy Treatment Market Trends
The rest of the World in the DMD treatment market is likely to witness a positive growth due to improving diagnosis rates, expanding healthcare infrastructure, and historically underdiagnosed patient populations. Global prevalence estimates indicate ~7.1 cases per 100,000 males, but regions in Asia, Africa, and Latin America have significantly lower reported rates due to limited screening and awareness, suggesting a large unmet population. As healthcare systems strengthen, early diagnosis is increasing, particularly in emerging economies. Urbanization and government investment in genetic testing are improving case identification. Additionally, multinational clinical trials are increasingly including these regions, expanding access to advanced therapies. This low baseline penetration, combined with improving healthcare access, creates strong growth potential compared to already saturated North American and European markets.
Competitive Landscape:
The Duchenne muscular dystrophy treatment market is highly competitive, driven by companies focusing on gene therapies, exon-skipping drugs, and innovative biologics. Players prioritize clinical advancements, regulatory approvals, strategic collaborations, and expanding global access. Strong pipelines, ongoing trials, and investment in rare disease research are key strategies to address unmet patient needs.
Key Industry Developments:
- In February 2026, Sarepta Therapeutics, Inc. launched ELEVIDYS in Japan via Chugai Pharmaceutical after securing NHI reimbursement listing.
- In January 2026, Santhera Pharmaceuticals signed a licensing deal with Nxera Pharma for AGAMREE in key Asia-Pacific markets.
- In December 2025, Sarepta Therapeutics, Inc. supported the inclusion of Duchenne muscular dystrophy in the U.S. RUSP.
Companies Covered in Duchenne Muscular Dystrophy Treatment Market
- Sarepta Therapeutics
- PTC Therapeutics
- Santhera Pharmaceuticals
- Catalyst Pharmaceuticals
- ITF THERAPEUTICS (Italfarmaco S.p.A.)
- NS Pharma, Inc. (Nippon Shinyaku Co., Ltd.)
Frequently Asked Questions
The global Duchenne muscular dystrophy treatment market is projected to be valued at US$ 3.1 Bn in 2026.
Rising DMD prevalence, gene therapy advancements, increasing awareness, supportive regulations, and growing rare disease investments.
The global Duchenne muscular dystrophy treatment market is poised to witness a CAGR of 9.5% between 2026 and 2033.
Expanding gene therapies, emerging markets, improved diagnostics, pipeline innovations, partnerships, and increasing access to advanced treatments.
Sarepta Therapeutics, PTC Therapeutics, Santhera Pharmaceuticals, Catalyst Pharmaceuticals, ITF THERAPEUTICS (Italfarmaco S.p.A.), NS Pharma, Inc. (Nippon Shinyaku Co., Ltd.)







