- Pharmaceuticals
- Anti-Bleeding Gel Market
Anti-Bleeding Gel Market Size, Share, and Growth Forecast, 2026 – 2033
Anti-Bleeding Gel Market by Product Type (Gel-based Anti-Bleeding Products, Liquid-based Anti-Bleeding Products, Spray-based Anti-Bleeding Products, Others), Indication (Biopsies of Soft Tissue, Trauma, Enucleations, Others), Distribution Channel, and Regional Analysis for 2026 – 2033
Anti-Bleeding Gel Market Size and Trends Analysis
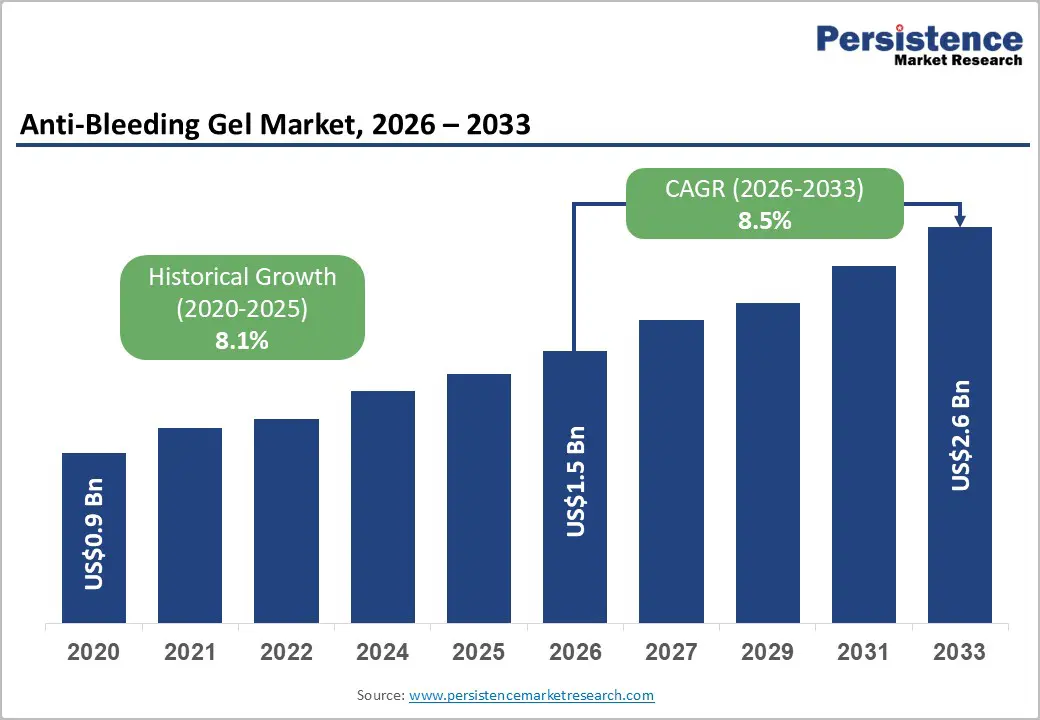
The global anti-bleeding gel market size is likely to be valued at US$1.5 billion in 2026 and is expected to reach US$2.6 billion by 2033, growing at a CAGR of 8.5% during the forecast period from 2026 to 2033, driven by rising incidences of surgical procedures, trauma cases, and emergency medical interventions across hospitals, clinics, and trauma centers worldwide. The increasing need for effective bleeding management solutions during surgical and post-operative care is supporting market expansion, as healthcare providers focus on improving patient safety and reducing procedure-related complications.
According to the World Health Organization’s global patient safety report (2024), surgical complications occur in up to 25% of patients undergoing surgery, highlighting the high-risk nature of surgical procedures and emphasizing the importance of reliable hemostatic solutions to control bleeding and improve clinical outcomes. Demographic shifts, such as the growing aging population and higher prevalence of chronic diseases, are expanding the use of anti-bleeding gels in routine surgical and post-operative care. Rising awareness among dental and orthopedic practitioners, coupled with expanding trauma care infrastructure and emergency medical services, is accelerating market adoption.
Key Industry Highlights:
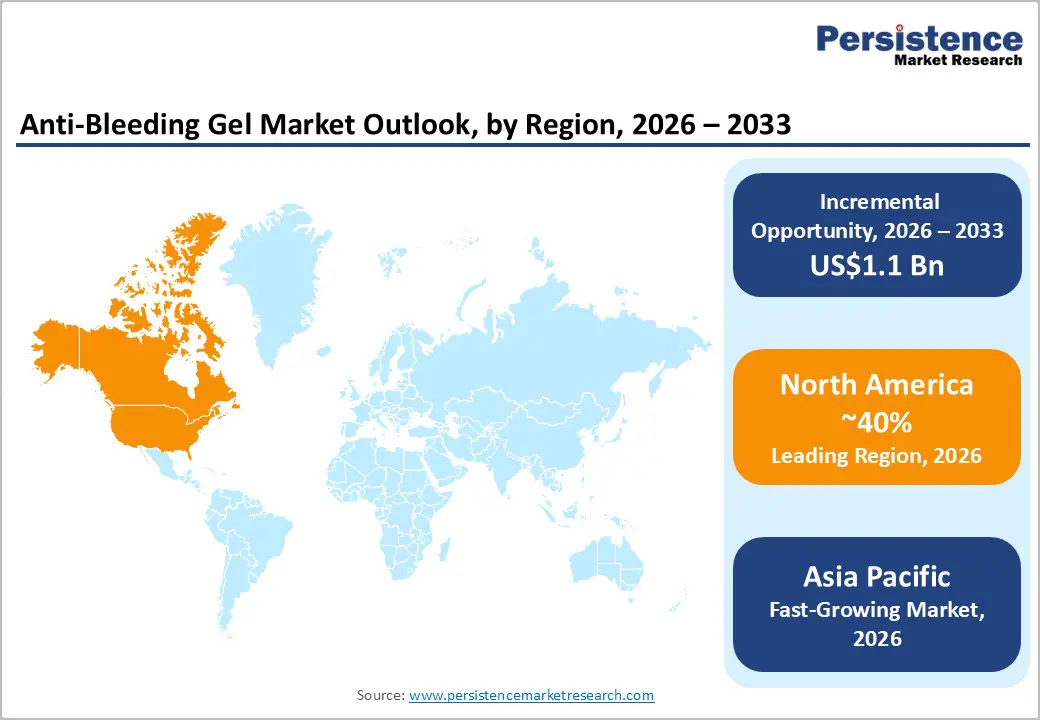
- Leading Region: North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by advanced healthcare infrastructure, high surgical volumes, strong presence of leading medical device companies, and increasing adoption of innovative hemostatic technologies.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region in the anti-bleeding gel in 2026, supported by expanding healthcare infrastructure, rising surgical procedures, increasing trauma cases, and growing adoption of advanced hemostatic products across emerging economies in the region.
- Leading Product Type: Gel-based anti-bleeding products are projected to represent the leading product type in 2026, accounting for 45% of the revenue share, driven by strong adhesion and effectiveness in surgical bleeding control.
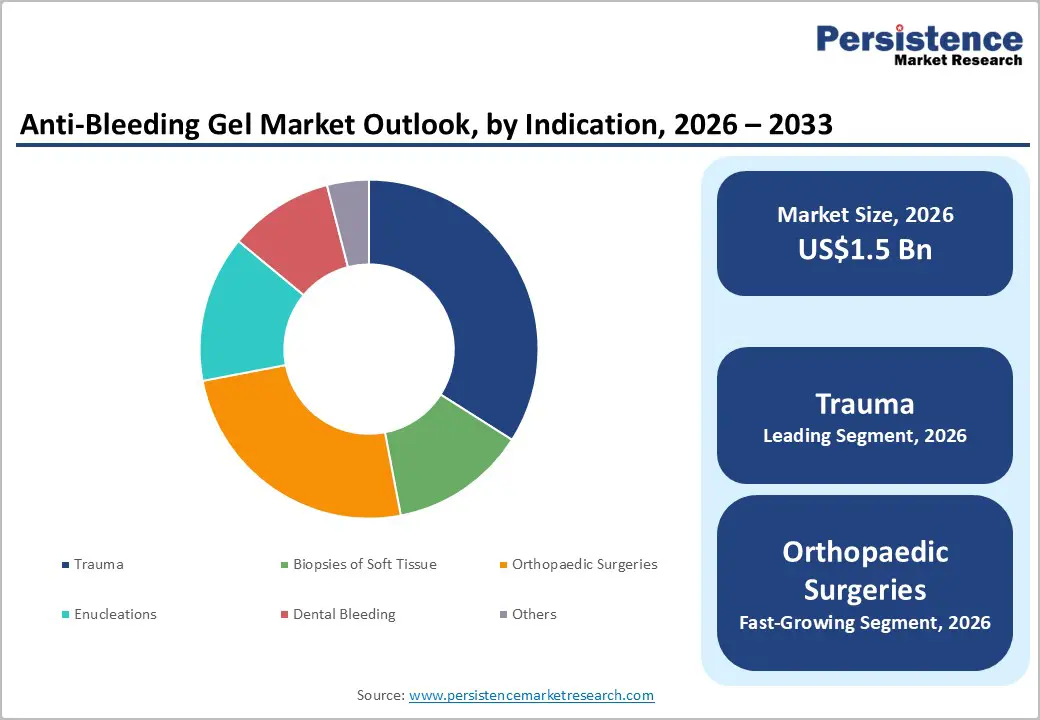
- Leading Indication: Trauma is anticipated to be the leading indication, accounting for over 35% of the revenue share in 2026, supported by high injury and accident cases requiring rapid bleeding control.
| Key Insights | Details |
|---|---|
| Anti-Bleeding Gel Market Size (2026E) | US$1.5 Bn |
| Market Value Forecast (2033F) | US$2.6 Bn |
| Projected Growth (CAGR 2026 to 2033) | 8.5% |
| Historical Market Growth (CAGR 2020 to 2025) | 8.1% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Growth Analysis- Increasing Surgical Volumes and Trauma Incidences
The growing number of surgical procedures worldwide is a major factor driving demand for anti-bleeding gels. Hospitals and surgical centers increasingly rely on effective hemostatic solutions to control bleeding during operations such as orthopedic, cardiovascular, and general surgeries. Anti-bleeding gels provide rapid clot formation, improve visibility at the surgical site, and reduce the risk of excessive blood loss. As healthcare systems expand access to surgical treatments and emergency care services, the need for reliable bleeding management products continues to rise. These gels are widely adopted because they are easy to apply, work quickly, and help surgeons manage bleeding efficiently across multiple types of medical procedures.
Trauma cases resulting from road accidents, workplace injuries, sports injuries, and natural disasters strengthen demand for anti-bleeding gels. Emergency departments and trauma centers require fast-acting bleeding control solutions to stabilize patients before definitive treatment. Anti-bleeding gels are increasingly included in trauma kits, ambulances, and emergency medical systems because they can rapidly control external and internal bleeding. Rising urbanization and growing vehicle usage contribute to higher injury rates, which increases the need for effective hemostatic products. In addition, military and disaster response teams utilize anti-bleeding gels to manage critical injuries in field conditions, expanding the product’s importance in emergency medicine.
Advancements in Minimally Invasive Procedures
Surgical techniques such as laparoscopy, endoscopy, and robotic surgeries require precise bleeding control to maintain visibility and minimize complications. Anti-bleeding gels are highly suitable for these procedures because they can be applied accurately to targeted areas without damaging surrounding tissues. Their ability to quickly form clots and seal minor bleeding points improves surgical efficiency and reduces procedure time. As hospitals increasingly shift toward minimally invasive techniques to improve patient recovery and reduce hospital stays, the demand for advanced hemostatic gels continues to grow steadily.
Technological advancements in medical materials have also improved the performance of anti-bleeding gels used in minimally invasive surgeries. Modern gels are developed using biocompatible polymers, collagen, and bioactive compounds that promote faster clotting and tissue healing. These formulations are designed to be easily delivered through surgical instruments used in minimally invasive procedures. Improved adhesion and controlled degradation properties allow the gels to function effectively without interfering with tissue recovery. As healthcare providers prioritize patient safety, shorter recovery times, and lower surgical risks, anti-bleeding gels have become an important supportive product in advanced surgical techniques and modern operating environments.
Barrier Analysis - Risk of Adverse Reactions and Biocompatibility Concerns
Despite their clinical benefits, anti-bleeding gels present certain safety concerns that limit wider adoption. Some formulations cause allergic reactions, inflammation, or tissue irritation in sensitive patients. Biocompatibility issues can arise when materials used in the gels interact negatively with surrounding tissues. Such complications affect healing outcomes and create hesitation among healthcare providers when selecting hemostatic products. Regulatory authorities also require extensive testing to ensure safety, which can delay product approvals. Hospitals and clinicians must carefully evaluate product composition and compatibility before use, particularly in complex surgeries where tissue sensitivity and infection risks are higher.
Concerns about long-term effects of certain hemostatic materials also act as a restraint in the market. Some gels remain in the body longer than expected or interfere with natural healing processes. In rare cases, residual material can lead to infection, delayed wound healing, or inflammatory responses. These potential risks encourage healthcare providers to use anti-bleeding gels selectively and prefer products with proven safety profiles. Manufacturers must invest in extensive clinical trials and advanced formulation technologies to minimize adverse reactions. Addressing biocompatibility challenges through improved materials and regulatory compliance is essential for maintaining trust and expanding product acceptance in clinical settings.
Storage, Shelf-Life, and Logistical Constraints
Storage and shelf-life limitations present another challenge for the anti-bleeding gel market. Many formulations require controlled storage conditions to maintain their stability and effectiveness. Exposure to excessive heat, moisture, or light degrade the active components and reduce product performance. Healthcare facilities must therefore follow strict storage guidelines, which can increase operational complexity. In regions with limited infrastructure or unstable supply chains, maintaining proper storage conditions becomes difficult. These logistical challenges limit product availability in remote healthcare facilities and emergency response environments where rapid access to bleeding control solutions is critically important.
Transportation and distribution issues can also affect the availability of anti-bleeding gels in markets. Some products require temperature-controlled packaging or specialized handling during shipping to preserve their chemical stability. This increases supply chain costs and may create delays in product delivery. Hospitals and pharmacies must carefully manage inventory to avoid product expiration or wastage. In developing regions where medical supply chains are less developed, these logistical constraints can slow adoption. Manufacturers are increasingly working to develop more stable formulations with longer shelf lives to improve accessibility and reduce distribution challenges in diverse healthcare environments.
Opportunity Analysis - Technological Convergence with Smart Devices
The integration of medical products with smart healthcare technologies can improve monitoring and treatment outcomes. Researchers are exploring advanced hemostatic gels that work alongside digital health tools to monitor wound healing and bleeding control. Smart delivery systems and connected medical devices help healthcare professionals apply hemostatic products more precisely during procedures. Such technological convergence enhances treatment efficiency and supports better patient outcomes. As healthcare systems increasingly adopt digital solutions, integrating advanced materials with smart monitoring technologies presents promising growth opportunities for anti-bleeding gel manufacturers.
Advanced biomaterials and nanotechnology are also opening new possibilities for next-generation anti-bleeding gels. Innovative formulations can respond to biological signals, activate clotting mechanisms faster, and support tissue regeneration. Researchers are developing gels that release therapeutic agents or antimicrobial compounds to prevent infection while controlling bleeding. These smart materials can potentially interact with sensors and medical devices used during surgical procedures. As investment in medical technology research grows, these advancements create significant opportunities for innovative products in the anti-bleeding gel market.
Veterinary and Dual-Use Applications
The veterinary healthcare sector offers a promising opportunity for the expansion of anti-bleeding gel products. Veterinary surgeries and animal trauma care require effective bleeding control solutions similar to those used in human medicine. Anti-bleeding gels can be applied in procedures involving pets, livestock, and wildlife to manage bleeding quickly and safely. As veterinary healthcare services continue to improve, demand for specialized hemostatic products in animal treatment is increasing. Veterinary clinics and hospitals are adopting advanced medical supplies to improve surgical outcomes and animal recovery, creating new commercial opportunities for manufacturers of anti-bleeding gels.
Dual-use applications expand the potential market for anti-bleeding gels beyond traditional healthcare environments. These products can be used in emergency response kits, military medical units, disaster relief operations, and outdoor safety equipment. Their portability and rapid action make them valuable tools for controlling bleeding in critical situations before professional medical care is available. Increasing awareness about first-aid preparedness and trauma response has encouraged organizations and individuals to include hemostatic products in emergency kits. As focus on emergency preparedness grows, anti-bleeding gels are widely used across multiple professional and consumer applications for rapid bleeding control in emergency situations.
Category-wise Analysis
Product Type Insights
Gel-based anti-bleeding products are expected to lead the anti-bleeding gel market, accounting for approximately 45% of revenue in 2026, driven by their strong adhesion and effective bleeding control across a wide range of surgical and trauma applications. These gels easily conform to irregular wound surfaces, making them highly suitable for complex surgical environments where precise hemostasis is required. Hospitals and surgical centers prefer gel formulations as they create a protective barrier over the wound, support rapid clot formation, and reduce the risk of infection. For example, Cresilon developed Traumagel®, a plant-based hemostatic gel designed to rapidly stop severe bleeding in trauma and emergency care settings, highlighting the growing clinical adoption of advanced gel-based bleeding control technologies.
Liquid-based anti-bleeding products are likely to represent the fastest-growing segment, supported by their ability to provide precise and controlled application during minimally invasive procedures. These formulations are especially valuable in modern surgical techniques such as laparoscopy and endoscopy, where targeted bleeding control is required in confined surgical spaces. Liquid formulations can easily spread across small bleeding areas and seal tissues without applying excessive pressure, making them effective for delicate procedures. For example, Baxter International provides liquid hemostatic solutions used in surgical settings to manage bleeding efficiently during complex procedures.
Indication Insights
Trauma is projected to lead the market, capturing around 35% of the revenue share in 2026, supported by the increasing number of accident-related injuries and emergency medical cases requiring immediate bleeding control. Trauma cases often involve severe external or internal bleeding that requires rapid stabilization before medical treatment. Anti-bleeding gels play a crucial role in emergency medicine by enabling quick clot formation and reducing blood loss in critical situations. For example, Z-Medica produces hemostatic gel products used in trauma kits by emergency responders and military medical teams, highlighting the importance of anti-bleeding gels in managing acute bleeding situations.
Orthopaedic surgeries are likely to be the fastest-growing segment, driven by rising number of procedures related to bone injuries, joint replacements, and musculoskeletal disorders. These surgeries often involve significant bleeding from bone surfaces and surrounding tissues, making effective hemostatic management essential for maintaining surgical visibility and ensuring patient safety. Anti-bleeding gels are particularly effective in orthopedic procedures because they can adapt to uneven bone structures and provide localized bleeding control without interfering with surgical instruments. For example, Johnson & Johnson’s Ethicon division provides surgical hemostatic products that are commonly used during orthopedic procedures to support effective bleeding control and improve surgical outcomes.
Regional Insights
North America Anti-Bleeding Gel Market Trends
North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by strong adoption of advanced surgical technologies and well-developed healthcare infrastructure. Hospitals and trauma centers across the region increasingly use hemostatic gels to control bleeding during general, orthopedic, cardiovascular, and dental surgeries. The high prevalence of surgical procedures and trauma cases continues to support consistent demand for effective bleeding management products. The growing use of minimally invasive and robotic-assisted surgeries has increased the need for precise hemostatic materials that can be easily applied in narrow surgical fields. Healthcare providers prefer gel-based hemostatic products because they improve surgical visibility and reduce procedure complications.
Innovation and product development remain key trends shaping the regional market. Biotechnology and medical device companies in the U.S. and Canada are actively developing new hemostatic gel technologies designed for rapid bleeding control in trauma and surgical care. For example, Cresilon introduced its plant-based hemostatic product TRAUMAGEL, which received U.S. FDA clearance for controlling moderate to severe external bleeding and is designed for use by emergency medical teams, trauma centers, and first responders. The product is supplied in a pre-filled syringe and can rapidly control bleeding when applied to wounds, improving emergency response efficiency.
Europe Anti-Bleeding Gel Market Trends
Europe is likely to be a significant market for anti-bleeding gel, due to strong healthcare infrastructure, rising surgical procedures, and increasing demand for advanced hemostatic technologies across hospitals and surgical centers. Countries such as Germany, the U.K., and France are major contributors due to their well-established healthcare systems and growing adoption of modern surgical techniques. Anti-bleeding gels are widely used in cardiovascular, orthopedic, and minimally invasive surgeries because they help surgeons control bleeding quickly and maintain clear visibility at the surgical site. In Europe, the increasing focus on patient safety and infection prevention is also encouraging the use of biocompatible hemostatic gels in surgical procedures.
Innovation and collaboration between medical technology companies and healthcare institutions remain important trends in the European anti-bleeding gel market. Several companies are investing in research and product development to introduce advanced hemostatic solutions that improve surgical efficiency and patient outcomes. For example, B. Braun Melsungen AG, a Germany-based medical technology company, has expanded its hemostatic product portfolio and collaborates with healthcare providers to develop innovative bleeding control solutions for surgical procedures. Such initiatives highlight how European companies are focusing on high-quality biomaterials and improved clinical performance to support surgeons in complex operations.
Asia Pacific Anti-Bleeding Gel Market Trends
The Asia Pacific region is likely to be the fastest-growing region, driven by expanding healthcare infrastructure, increasing surgical procedures, and rising awareness of advanced wound-care technologies across the region. Countries such as China and India are major contributors to regional demand as hospitals increasingly adopt modern hemostatic solutions to manage bleeding during surgeries and trauma treatments. Rapid urbanization and a growing population are also contributing to higher incidences of injuries and chronic diseases that require surgical intervention. The growing number of ambulatory surgical centers and specialized clinics supports the use of anti-bleeding gels for efficient bleeding management.
Another important trend in the region is the increasing presence of medical technology companies expanding their operations and distribution networks in Asia Pacific. Many companies are collaborating with regional hospitals and healthcare providers to introduce advanced hemostatic products suitable for local clinical requirements. For example, Medtronic has strengthened its presence in several Asia Pacific countries by supplying surgical hemostatic solutions used in abdominal and minimally invasive procedures across hospitals in the region. These products help surgeons manage bleeding efficiently while supporting faster surgical workflows.
Competitive Landscape
The global anti-bleeding gel market exhibits a moderately fragmented structure, driven by continuous product innovation, expanding surgical applications, and increasing demand for advanced hemostatic solutions in trauma and surgical care. The market includes a mix of large multinational medical device manufacturers and specialized biotechnology companies focused on bleeding control technologies. Rising surgical volumes, the need for rapid hemostasis during minimally invasive procedures, and advancements in biomaterial formulations have intensified competition among manufacturers.
With key leaders including Johnson & Johnson (through Ethicon), Baxter International, Medtronic, Pfizer, Becton, Dickinson and Company, and Integra LifeSciences. These players compete through continuous innovation in hemostatic gel formulations, clinical validation, and expansion of product portfolios designed for surgical, trauma, and emergency care applications. Leading companies leverage strong distribution networks, regulatory expertise, and established relationships with hospitals and surgical centers to strengthen their market position.
Key Industry Developments:
- In January 2026, researchers led by Professor Steve Park and Professor Sangyong Jon from the Korea Advanced Institute of Science and Technology (KAIST) developed a spray-on powder hemostatic agent that forms a hydrogel barrier within one second to rapidly stop severe bleeding in trauma and emergency situations.
- In October 2025, Cresilon announced the U.S. nationwide launch of TRAUMAGEL, a plant-based hemostatic gel designed to rapidly control moderate to severe external bleeding. The product previously received clearance from the U.S. Food and Drug Administration in August 2024 and is intended for use by emergency responders, trauma centers, and healthcare providers to quickly stop bleeding when applied directly to wounds.
- In May 2025, Baxter International Inc. announced the launch of Hemopatch Sealing Hemostat, a room-temperature absorbable collagen pad designed for rapid hemostasis and tissue sealing during surgical procedures, suitable for both open surgery and minimally invasive surgical procedures. The product eliminates the need for refrigeration, improving accessibility and convenience for surgeons in operating rooms.
Companies Covered in Anti-Bleeding Gel Market
- Pfizer Inc.
- Johnson & Johnson
- Baxter International Inc.
- B. Braun Melsungen AG
- Sanofi S.A.
- Becton, Dickinson and Company
- Medtronic plc
- Ethicon, Inc.
- CryoLife, Inc.
- CSL Behring
- HemCon Medical Technologies, Inc.
- Z-Medica, LLC
- Marine Polymer Technologies, Inc.
- Advanced Medical Solutions Group plc
- Arch Therapeutics, Inc.
- Cohera Medical, Inc.
- Mallinckrodt Pharmaceuticals
- Integra LifeSciences Corporation
- Stryker Corporation
- Biolife LLC
Frequently Asked Questions
The global anti-bleeding gel market is projected to reach US$1.5 billion in 2026.
The anti-bleeding gel market is driven by the rising number of surgical procedures, increasing trauma and accident cases, and growing demand for rapid hemostatic solutions in emergency and surgical care.
The anti-bleeding gel market is expected to grow at a CAGR of 8.5% from 2026 to 2033.
Key opportunities in the anti-bleeding gel market arise from advancements in hemostatic biomaterials and the expanding use of rapid bleeding control products in trauma care, military medicine, and emergency response applications.
Pfizer Inc., Johnson & Johnson, Baxter International Inc., B. Braun Melsungen AG, and Sanofi S.A. are the leading players.







