- Medical Devices
- Digital Pills Market
Digital Pills Market Size, Share, and Growth Forecast, 2026 - 2033
Digital Pills Market by Product Type (Tablet, Capsule, Microchip Delivery Device), Indication (Mental Disorders, Others), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Others), Diseases (Cardiovascular Diseases, Others), and Regional Analysis for 2026 - 2033
Digital Pills Market Size and Trends Analysis
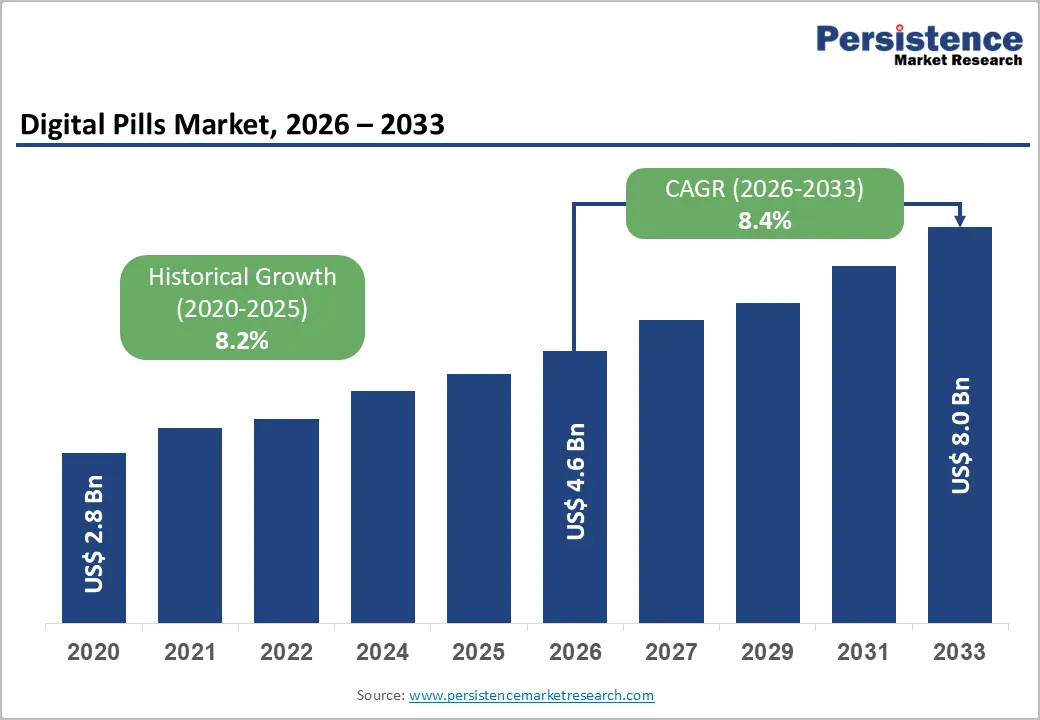
The global digital pills market size is likely to be valued at US$4.6 billion in 2026 and is expected to reach US$8.0 billion by 2033, growing at a CAGR of 8.4% during the forecast period from 2026 to 2033, driven by increasing demand for advanced medication adherence solutions, non-invasive diagnostic technologies, and real-time patient monitoring systems.
Digital pills combine ingestible sensors, microchips, or imaging components with pharmaceutical formulations to capture and transmit physiological or ingestion data to external devices such as wearable patches, smartphones, or cloud-based healthcare platforms. These technologies are increasingly being integrated into chronic disease management and clinical research workflows to improve treatment outcomes and reduce non-adherence-related complications, which remain a major challenge in healthcare systems.
Growth in this market is also supported by the rising prevalence of long-term conditions such as cardiovascular disorders, diabetes, cancer, gastrointestinal diseases, and neurological and mental health conditions, all of which require continuous monitoring and strict medication adherence. Healthcare providers are increasingly adopting smart pill technologies to enable remote patient monitoring, reduce hospital readmissions, and support personalized treatment strategies.
Key Industry Highlights:
- Leading Region: North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by strong healthcare infrastructure, advanced digital health adoption, and favorable regulatory and reimbursement frameworks.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region, supported by rapid healthcare digitization, a large patient base, and expanding adoption of smart monitoring technologies.
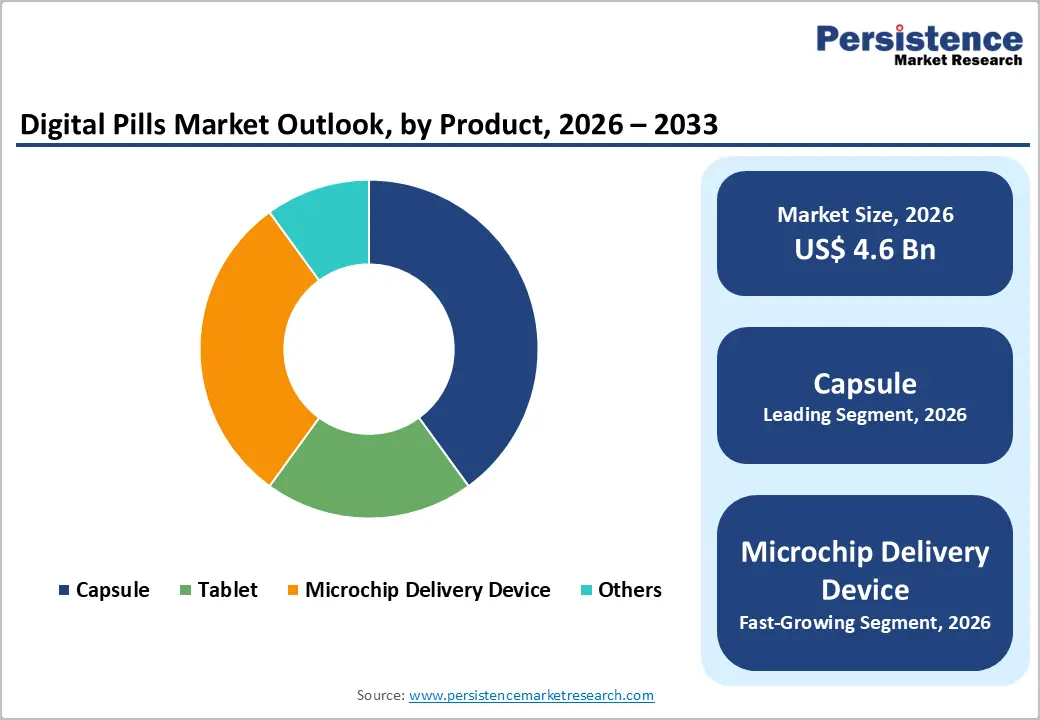
- Leading Product Type: Capsules are projected to represent the leading product type in 2026, accounting for 60% of the revenue share, driven by strong sensor integration capability and high reliability in drug delivery monitoring.
- Leading Indication: Mental disorders are anticipated to be the leading indication, accounting for over 40% of the revenue share in 2026, supported by high non-adherence rates and early regulatory approvals in psychiatric applications.
| Key Insights | Details |
|---|---|
| Digital Pills Market Size (2026E) | US$4.6 Bn |
| Market Value Forecast (2033F) | US$8.0 Bn |
| Projected Growth (CAGR 2026 to 2033) | 8.4% |
| Historical Market Growth (CAGR 2020 to 2025) | 8.2% |
DRO Analysis
Driver - Technological Advancements in Ingestible Sensors and Data Integration
Continuous innovation in microelectronics, biocompatible materials, and wireless communication has allowed sensors to be embedded into capsules and tablets without affecting drug performance. These devices transmit ingestion data to external wearables, smartphones, and cloud platforms, improving visibility into patient behavior. Integration with artificial intelligence and healthcare IT systems enhances data interpretation and clinical decision-making. The growing convergence of digital health technologies is improving the accuracy, scalability, and usability of ingestible systems across hospitals, research, and remote care settings.
Advancements are improving connectivity between ingestible sensors and digital healthcare ecosystems, enabling seamless data flow between patients and providers. Enhanced cloud computing and analytics platforms allow real-time tracking of medication adherence and physiological responses. These systems reduce dependency on manual reporting and improve treatment precision. Pharmaceutical companies and medtech firms are increasingly integrating smart sensor technologies into drug development and clinical trials.
Rising Prevalence of Chronic Diseases and Medication Non-Adherence Challenges
Conditions such as cardiovascular disorders, diabetes, cancer, and neurological illnesses require long-term medication adherence, where missed doses can lead to severe health complications. Digital pills help address this challenge by providing accurate, real-time confirmation of drug ingestion, reducing reliance on patient self-reporting. This improves clinical outcomes and enables physicians to monitor adherence remotely. Aging populations and rising multimorbidity cases increase the need for continuous monitoring solutions, making ingestible sensor technologies highly valuable in modern healthcare systems focused on efficiency and outcome-based care delivery.
Healthcare systems are increasingly focusing on reducing hospital readmissions and treatment failures caused by non-adherence. Digital pills provide objective data that helps clinicians adjust therapies more effectively and ensure consistent medication use. This is especially important in psychiatric care, oncology, and chronic disease management, where adherence directly impacts survival rates and recovery outcomes. Growing awareness among healthcare providers about the economic burden of non-adherence is also supporting adoption.
Restraint - Patient and Provider Acceptance and Privacy Concerns
Many patients feel uncomfortable with internal monitoring technologies, associating them with loss of privacy and constant surveillance. This psychological resistance reduces willingness to adopt such solutions despite their clinical benefits. Healthcare providers also express concerns regarding ethical implications, data ownership, and the extent of monitoring required in routine care. Varying levels of digital literacy among patients slow adoption. These acceptance challenges limit widespread integration of digital pills into mainstream healthcare systems, especially in traditional or conservative medical environments.
The continuous collection and transmission of sensitive health data raises questions about data security, consent, and potential misuse. Strict regulatory frameworks, such as data protection laws, add compliance complexity for healthcare providers and technology developers. Hospitals and clinics must ensure secure storage and transmission of ingestion data, increasing operational challenges. Lack of clear standards for ethical data use also creates uncertainty among stakeholders.
Technical Reliability and Integration Challenges
Variations in pH levels, digestive movement, and patient physiology can affect sensor activation and data accuracy. Maintaining stable signal transmission from inside the body to external devices is also difficult. Inconsistent performance can lead to incomplete or inaccurate adherence data, limiting clinical reliability. Ensuring long-term biocompatibility and safe ingestion without affecting drug efficacy requires extensive testing. These technical limitations increase development time and cost, slowing widespread commercialization and adoption of digital pill technologies in healthcare systems.
Integration challenges hinder the scalability of digital pill systems across healthcare infrastructures. Compatibility with hospital information systems, electronic health records, and mobile health applications remains inconsistent across regions and providers. Complex data synchronization between ingestible sensors, wearable devices, and cloud platforms adds operational difficulty. High regulatory requirements for combination products delay market entry. Ensuring cybersecurity and protecting sensitive patient data during transmission increases system complexity.
Opportunity - Technological Convergence with AI and Personalized Medicine
AI algorithms can analyze real-time ingestion data to detect adherence patterns, predict health risks, and optimize treatment plans. This enables healthcare providers to move from reactive to proactive care models. Combining ingestible sensor data with genomics and patient history allows for highly individualized treatment strategies. Digital pills enhance precision medicine by providing continuous feedback on drug response. As healthcare systems increasingly adopt data-driven decision-making, AI-enabled ingestible technologies are becoming essential tools for improving therapeutic outcomes and reducing healthcare inefficiencies.
The convergence of digital pills with personalized medicine is also transforming clinical trial design and drug development processes. Real-time adherence tracking ensures more accurate trial results and reduces data gaps caused by patient non-compliance. Pharmaceutical companies can optimize dosing strategies based on live patient feedback, improving drug efficacy and safety profiles. Integration with cloud-based analytics platforms enables large-scale data processing and predictive modeling. Growing investment in digital health ecosystems is accelerating innovation in this space.
Expansion into Emerging-Market Chronic-Care and Infectious-Disease Programs
Emerging markets present strong growth opportunities for digital pills due to rising chronic disease prevalence and improving healthcare infrastructure. Countries in Asia, Africa, and Latin America are witnessing increased adoption of digital health technologies driven by government initiatives and expanding mobile connectivity. Digital pills can support large-scale public health programs by improving medication adherence in diseases such as tuberculosis, HIV, and hepatitis. These regions often face challenges related to treatment compliance, making ingestible monitoring solutions highly valuable.
Partnerships between medtech companies and local healthcare providers are accelerating market penetration in emerging economies. These collaborations help adapt digital pill technologies to regional healthcare needs and affordability constraints. Growing investment in telemedicine and remote patient monitoring systems is supporting adoption. Governments are also promoting digital health transformation to improve access and reduce disease burden. As healthcare infrastructure continues to expand, digital pills offer a practical solution for improving treatment adherence and patient outcomes.
Category-wise Analysis
Product Type Insights
Capsules are expected to lead the digital pills market, accounting for approximately 60% of revenue in 2026, driven by their superior design flexibility that allows seamless integration of ingestible sensors, antennas, and drug payloads without compromising drug stability or bioavailability. Capsules are widely preferred in clinical settings because they ensure reliable sensor activation and are easier to manufacture at scale compared to other formats. For example, CapsoVision’s CapsoCam Plus, a capsule endoscopy system, demonstrates how capsule-based digital pills can capture high-resolution gastrointestinal images as it travels through the digestive tract and transmit diagnostic insights for non-invasive disease detection, reinforcing their leadership in the segment.
Tablets are likely to represent the fastest-growing segment, supported by increasing demand for scalable and cost-efficient manufacturing solutions. They are gaining momentum due to their compatibility with high-volume pharmaceutical production lines and advancements in flexible micro-circuit integration that allow sensors to be embedded without affecting compression processes. For example, ongoing development in smart antihypertensive tablets, where digital tracking technologies are being integrated to monitor long-term medication adherence in cardiovascular patients, enabling better disease management and reducing treatment failures.
Indication Insights
Mental disorders are projected to lead the market, capturing around 40% of the revenue share in 2026, supported by early regulatory adoption and high unmet needs in psychiatric medication adherence. Conditions such as schizophrenia and bipolar disorder require strict and continuous drug compliance, where missed doses can lead to severe relapse or hospitalization. For example, Abilify MyCite, the first FDA-approved digital pill system for schizophrenia, which demonstrated the ability to digitally confirm medication ingestion and support behavioral health monitoring through connected digital platforms.
Infectious diseases are likely to be the fastest-growing indication, driven by increasing efforts to improve treatment adherence in tuberculosis, HIV, and hepatitis C management. These conditions require strict long-term medication compliance, and non-adherence can contribute to drug resistance and treatment failure. For example, the use of digital adherence technologies in tuberculosis treatment programs, where ingestion tracking systems are being deployed to ensure patients complete full antibiotic courses, improves treatment success rates and reduces disease transmission.
Regional Insights
North America Digital Pills Market Trends
North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by advanced healthcare infrastructure, strong digital health adoption, and supportive regulatory frameworks such as FDA guidance for combination products. The region benefits from widespread use of electronic health records, remote patient monitoring systems, and high investment in AI-enabled healthcare solutions. For example, Medtronic, which is advancing ingestible and capsule-based monitoring technologies to enhance gastrointestinal diagnostics and remote patient management across the U.S. healthcare system.
Pharmaceutical companies are increasingly collaborating with technology firms to integrate smart ingestion tracking into clinical trials and post-market surveillance. Expansion of telehealth platforms and hospital-at-home programs is strengthening market penetration. Continuous innovation in miniaturized sensors and wireless data transmission is improving the reliability and scalability of solutions. Growing patient awareness and preference for non-invasive monitoring tools are also supporting adoption.
Europe Digital Pills Market Trends
Europe is likely to be a significant market for digital pills, due to strong public healthcare systems, a high burden of gastrointestinal and chronic diseases, and increasing adoption of non-invasive diagnostic technologies. Healthcare providers focus on improving treatment adherence and reducing hospital burden through digital health solutions. For example, CapsoVision, which offers capsule endoscopy systems widely used in Europe for advanced gastrointestinal visualization and minimally invasive diagnostics.
Europe is also experiencing growing collaboration between research institutions, medtech companies, and hospitals to develop advanced ingestible technologies. Strict data privacy regulations such as GDPR influence system design, ensuring secure handling of patient ingestion data. Expansion of AI-driven healthcare analytics and telemedicine services is supporting the integration of digital pills into broader care pathways. Countries with advanced healthcare infrastructure are leading adoption, while Eastern Europe is gradually emerging with modernization initiatives.
Asia Pacific Digital Pills Market Trends
The Asia Pacific region is likely to be the fastest-growing region, driven by large patient populations, rising chronic disease burden, and rapid healthcare digitalization. Countries such as China and India are investing heavily in smart healthcare infrastructure and telemedicine platforms. The increasing prevalence of diabetes, cardiovascular diseases, and infectious diseases is driving demand for medication adherence solutions. Growing smartphone penetration and internet connectivity are enabling remote monitoring and data-driven healthcare delivery across the region.
Hospitals are increasingly using ingestible sensors for clinical monitoring and diagnostic applications, particularly in gastrointestinal and chronic disease management. For example, Olympus Corporation, which is actively involved in capsule endoscopy technologies used widely across Japan and other Asia Pacific markets for non-invasive gastrointestinal diagnostics. Expanding healthcare investments, rising medical tourism, and supportive regulatory reforms are accelerating growth.
Competitive Landscape
The global digital pills market exhibits a moderately fragmented structure, driven by the presence of both established pharmaceutical and medical device companies alongside emerging digital health innovators. The competitive environment is shaped by continuous advancements in ingestible sensor technology, capsule endoscopy, and AI-enabled monitoring systems.
With key leaders including Medtronic, Olympus Corporation, Proteus Digital Health, Otsuka Holdings Co. Ltd., and CapsoVision, the market demonstrates a mix of innovation-driven startups and large multinational players contributing to technological development and commercialization. These players compete through strategic collaborations, product innovation, and expansion into new therapeutic areas such as mental health, gastrointestinal disorders, and chronic disease management.
Key Industry Developments:
- In March 2025, etectRx expanded the deployment of its ID-Cap System across VA hospitals, transplant clinics, and behavioral health programs, strengthening its presence in medication adherence monitoring. The system uses ingestible sensors to track real-time drug ingestion and integrates with remote patient monitoring platforms through an open API. This development highlights increasing adoption of digital pill technologies in specialized care settings, particularly for high-risk patient populations requiring strict adherence.
- In January 2025, CapsoVision, Inc. received FDA clearance for its CapsoCam Plus system for use in pediatric patients aged two and older. This approval expands the application of capsule endoscopy by enabling non-invasive gastrointestinal diagnostics in children, reducing the need for traditional endoscopic procedures. The development highlights growing adoption of ingestible diagnostic technologies and supports broader use of digital pill systems in improving patient comfort, early disease detection, and clinical efficiency across diverse patient populations.
Companies Covered in Digital Pills Market
- 2MORROW Inc.
- Otsuka Holdings Co. Ltd.
- PureTech Health
- Abbott
- Olympus Corporation
- Medtronic
- Smith+Nephew
- Novartis AG
- MOCACARE
- GENTAG, Inc.
- Zio by iRhythm Technologies, Inc.
- Perficient Inc.
Frequently Asked Questions
The global digital pills market is projected to reach US$4.6 billion in 2026.
Rising demand for medication adherence monitoring and growing adoption of non-invasive, real-time patient tracking technologies drive the digital pills market.
The digital pills market is expected to grow at a CAGR of 8.4% from 2026 to 2033.
Expansion of AI-integrated personalized medicine and increasing adoption in emerging markets for chronic and infectious disease management create key opportunities in the digital pills market.
2MORROW Inc., Otsuka Holdings Co. Ltd., PureTech, Teladoc Health, Inc., and Medtronic are the leading players.








