- Healthcare Services
- AI-based Clinical Trials Solution Providers Market
AI-based Clinical Trials Solution Providers Market Size, Share, and Growth Forecast, 2026 - 2033
AI-based Clinical Trials Solution Providers Market by Therapeutic Areas (Cardiovascular Disorders, Others), Trial Phase (Phase I, Phase II, Phase III), Application (Clinical Trial Design, Patient Enrichment and Enrollment, Others), and Regional Analysis for 2026 - 2033
AI-based Clinical Trials Solution Providers Market Size and Trends Analysis
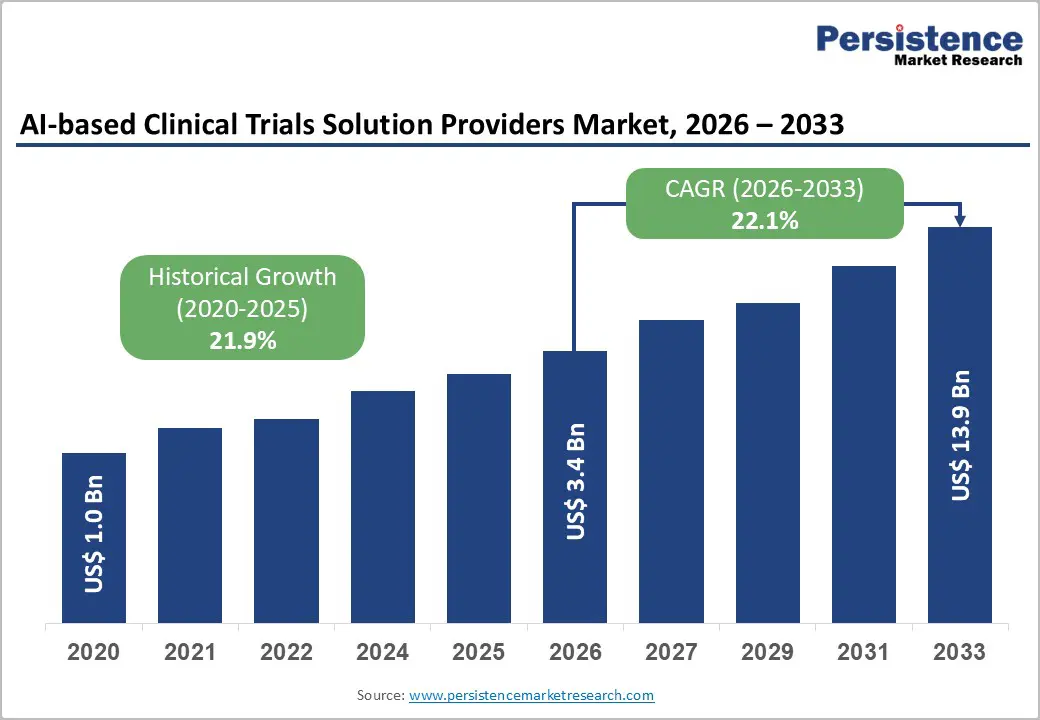
The global AI-based clinical trials solution providers market size is likely to be valued at US$3.4 billion in 2026 and is expected to reach US$13.9 billion by 2033, growing at a CAGR of 22.1% during the forecast period from 2026 to 2033, driven by the increasing need to modernize and streamline traditional clinical development processes. Conventional clinical trials are often characterized by high failure rates, lengthy timelines, and operational inefficiencies, creating significant pressure on pharmaceutical and biotechnology companies to adopt more advanced, data-driven approaches.
Artificial intelligence is emerging as a critical enabler in this transformation by enhancing multiple stages of the clinical trial lifecycle, including protocol design, patient recruitment, site selection, data management, and real-time monitoring. Rather than replacing regulatory frameworks, AI solutions complement existing processes by improving accuracy, reducing manual workload, and enabling faster, evidence-based decision-making across trial phases. The growing complexity and volume of clinical trials, particularly with the rise of precision medicine, rare disease research, and targeted therapies.
Key Industry Highlights:
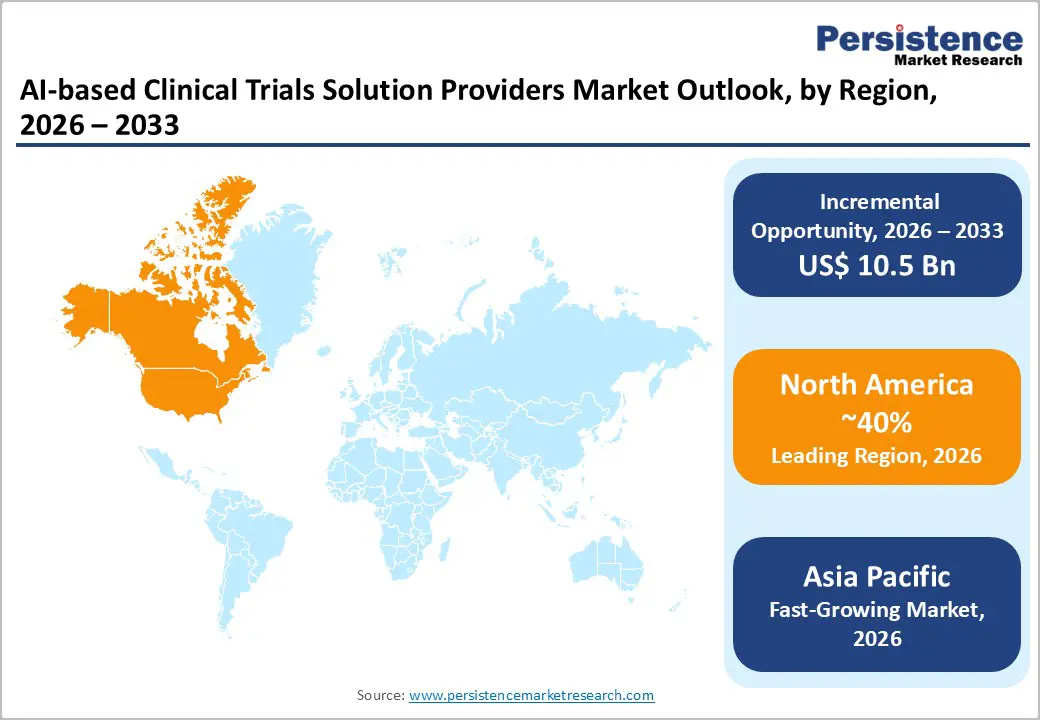
- Leading Region: North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by strong innovation ecosystems, advanced digital infrastructure, supportive regulatory frameworks, and high adoption of AI-driven clinical research solutions.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region, supported by rapid adoption, expanding clinical trial infrastructure, and increasing integration of AI-driven solutions across emerging economies.
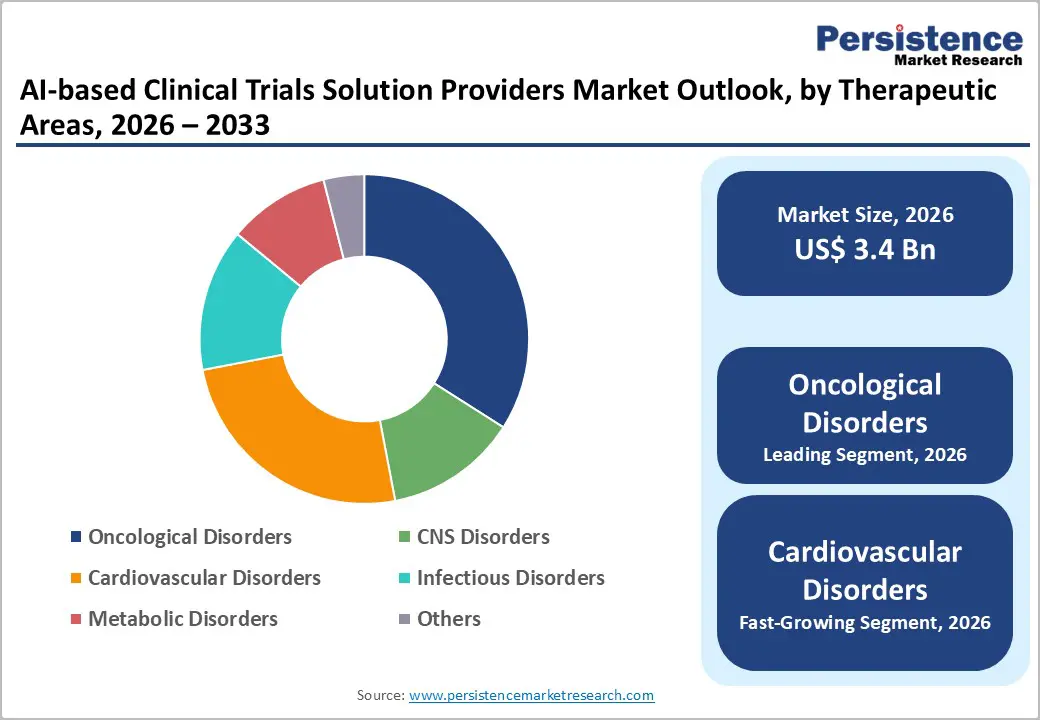
- Leading Therapeutic Areas: Oncological disorders are projected to lead, accounting for 40% of the revenue share, driven by high trial volumes and AI suitability in precision medicine.
- Leading Trial Phase: Phase III is anticipated to be the leading trial phase, accounting for over 50% of the revenue share in 2026, supported by higher complexity, cost, and need for AI-driven optimization in late-stage trials.
| Key Insights | Details |
|---|---|
|
AI-based Clinical Trials Solution Providers Market Size (2026E) |
US$3.4 Bn |
|
Market Value Forecast (2033F) |
US$13.9 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
22.1% |
|
Historical Market Growth (CAGR 2020 to 2025) |
21.9% |
Market Factors - Driver, Restraint, and Opportunity Analysis
Driver Analysis - Technological Advancements in AI/ML and Integration with Real-World Evidence (RWE)
Advancements in artificial intelligence and machine learning are significantly enhancing the efficiency and accuracy of clinical trials by enabling predictive modeling, automated data processing, and intelligent protocol design. AI algorithms can analyze vast datasets, including electronic health records, genomics, and imaging data, to identify suitable patient populations and predict trial outcomes. The integration of real-world evidence strengthens these capabilities by providing continuous, real-time insights beyond controlled trial environments, improving decision-making, and reducing dependency on traditional data collection methods.
The convergence of AI with real-world data sources is transforming how clinical trials are conducted by enabling adaptive trial designs and decentralized approaches. Sponsors can leverage these technologies to monitor patient behavior, treatment responses, and safety signals more effectively. This leads to faster recruitment, improved patient retention, and enhanced data reliability. As regulatory bodies increasingly acknowledge the value of RWE in supporting clinical decisions, the adoption of AI-driven platforms continues to accelerate, fostering innovation and improving overall trial success rates.
Rising Prevalence of Chronic Diseases and Increasing Clinical Trial Complexity
The growing burden of chronic diseases such as cancer, cardiovascular conditions, and neurological disorders is driving the need for more complex and large-scale clinical trials. These trials often involve diverse patient populations, multiple endpoints, and longer durations, making traditional methods inefficient and resource-intensive. AI-based solutions help manage this complexity by enabling precise patient stratification, optimizing trial protocols, and improving data analysis across multiple variables, thereby enhancing the overall effectiveness of clinical research.
As therapeutic approaches become more personalized, particularly in areas such as oncology and rare diseases, clinical trials require advanced tools to handle biomarker-driven studies and targeted therapies. AI facilitates the identification of patient subgroups and predicts treatment responses, reducing trial failures and improving outcomes. This increasing complexity, combined with the need for faster drug development, is pushing pharmaceutical companies and research organizations to adopt AI-driven clinical trial solutions to remain competitive and efficient.
Restraint Analysis - Data Privacy, Security, and Algorithmic Bias Concerns
The use of AI in clinical trials involves handling large volumes of sensitive patient data, raising significant concerns around data privacy and security. Ensuring compliance with data protection regulations while maintaining data accessibility for AI models is a complex challenge. Any breach or misuse of data can undermine trust among patients and stakeholders, limiting the adoption of AI-based solutions. The reliance on digital platforms increases vulnerability to cybersecurity threats, requiring robust infrastructure and governance frameworks.
Algorithmic bias is another critical concern, as AI models trained on incomplete or unrepresentative datasets may produce skewed results. This can impact patient selection, trial outcomes, and overall reliability of findings. Addressing these issues requires continuous monitoring, validation, and improvement of AI systems to ensure fairness and accuracy. Without proper mitigation strategies, these challenges can hinder the scalability and acceptance of AI technologies in clinical trial environments.
Interoperability and Change Management within Organizations
Integrating AI solutions into existing clinical trial systems is challenging due to interoperability issues, as many organizations rely on legacy infrastructure that may not be compatible with advanced technologies. Data silos, inconsistent formats, and a lack of standardized systems can limit the seamless flow of information, reducing the effectiveness of AI-driven insights. Overcoming these technical barriers requires significant investment in infrastructure modernization and system integration.
In technical challenges, organizations face resistance to change when adopting AI-based solutions. Clinical teams may be hesitant to rely on automated systems due to a lack of familiarity or concerns about accuracy and control. Effective change management strategies, including training, stakeholder engagement, and clear demonstration of value, are essential to drive adoption. Without addressing these organizational and cultural barriers, the full potential of AI in clinical trials may not be realized.
Opportunity Analysis - Technological Convergence with Decentralized and Hybrid Trial Models
The integration of AI with decentralized and hybrid clinical trial models presents a significant opportunity to transform the clinical research landscape. These models leverage digital tools such as remote monitoring, wearable devices, and telemedicine to conduct trials beyond traditional clinical sites. AI enhances these approaches by enabling real-time data analysis, patient monitoring, and predictive insights, improving both efficiency and patient experience while reducing operational costs.
As decentralized trials gain traction, AI plays a crucial role in ensuring data quality, patient engagement, and protocol adherence across distributed environments. This convergence allows sponsors to reach broader and more diverse patient populations, accelerating recruitment and improving trial inclusivity. The ability to conduct flexible and scalable trials using AI-driven platforms positions organizations to adapt to evolving healthcare needs and regulatory expectations, creating long-term growth opportunities in the market.
Bias Mitigation and Diversity Tools
The development of AI-driven tools focused on bias mitigation and diversity enhancement is creating new opportunities in clinical trials. These tools are designed to identify and address disparities in patient recruitment and data representation, ensuring that trial populations are more inclusive and reflective of real-world demographics. By improving diversity, clinical trials can generate more accurate and generalizable results, which are critical for regulatory approval and patient safety.
AI technologies can analyze demographic and clinical data to detect potential biases and recommend corrective actions during trial design and execution. This enables sponsors to proactively improve representation across different populations, including underrepresented groups. As regulatory agencies increasingly emphasize diversity in clinical research, the adoption of such tools is growing. This improves trial quality and also builds greater trust and credibility in the healthcare ecosystem.
Category-wise Analysis
Therapeutic Areas Insights
Oncological disorders are expected to lead the AI-based clinical trials solution providers market, accounting for approximately 40% of revenue in 2026, driven by the high volume of oncology trials and the increasing reliance on biomarker-driven and precision medicine approaches. The complexity of cancer studies, which often involve genomic profiling, imaging analytics, and targeted therapies, makes them highly suitable for AI integration. AI technologies enhance patient stratification, optimize protocol design, and improve endpoint prediction, significantly increasing trial efficiency. For example, AI is widely used in oncology trials to identify eligible patients through analysis of imaging and electronic health records, enabling faster recruitment and improved trial success rates.
Cardiovascular disorders are likely to represent the fastest-growing segment, supported by the increasing burden of heart diseases and the need for more efficient clinical trial methodologies. AI is increasingly utilized to analyze large-scale patient datasets, identify risk patterns, and support adaptive trial designs in cardiovascular studies. The integration of wearable devices and remote monitoring tools strengthens AI capabilities in capturing real-time patient data. This enables a more accurate assessment of treatment efficacy and safety. For example, AI-powered platforms are being used in cardiovascular trials to monitor patient vitals remotely and predict adverse events, supporting faster decision-making and improved patient outcomes.
Trial Phase Insights
Phase III is projected to lead the market, capturing around 50% of the revenue share in 2026, supported by large patient populations, higher costs, and critical regulatory decision points where AI provides substantial value. AI solutions are used to optimize site selection, predict patient enrollment timelines, and enhance data monitoring, reducing delays and improving trial efficiency. The complexity and scale of Phase III trials make them ideal for AI-driven risk-based monitoring and predictive analytics. For example, AI tools are commonly deployed in Phase III trials to forecast enrollment rates and identify underperforming sites, enabling timely corrective actions.
Phase I is likely to be the fastest-growing trial phase, driven by the increasing adoption of AI in early-stage drug development. These trials focus on safety, dosage, and initial efficacy, where AI helps design adaptive protocols and identify suitable patient cohorts more efficiently. The shift toward biomarker-driven and precision therapies accelerates the use of AI in early phases. AI enables better prediction of toxicity and response, reducing early-stage attrition rates. For example, AI platforms are used in Phase I trials to simulate drug behavior and optimize dosing strategies, allowing sponsors to make faster and more informed development decisions.
Regional Insights
North America AI-based Clinical Trials Solution Providers Market Trends
North America is anticipated to be the leading region, accounting for a market share of 40% in 2026, driven by strong technological integration, regulatory support, and high clinical trial activity. The region is witnessing rapid adoption of AI across trial functions such as patient recruitment, site feasibility, and predictive analytics, supported by the availability of large-scale healthcare datasets and advanced digital infrastructure. Regulatory bodies such as the U.S. FDA are increasingly incorporating AI into review processes and encouraging innovation through guidance on AI/ML-based tools, which is accelerating adoption across sponsors and CROs.
A key trend shaping the region is the rise of specialized AI solution providers and strategic collaborations aimed at optimizing clinical development workflows. Companies such as Saama are leveraging advanced analytics platforms to enhance data management, automate clinical operations, and improve decision-making across trial phases. For example, Saama’s AI-driven clinical analytics solutions are widely used by pharmaceutical companies in North America to streamline data processing and enable faster insights from complex trial datasets.
Europe AI-based Clinical Trials Solution Providers Market Trends
Europe is likely to be a significant market for AI-based clinical trials solution providers in 2026, due to a strategically important region in the AI-based clinical trials solution providers market, supported by strong regulatory harmonization, advanced healthcare infrastructure, and increasing public-private investments in digital health. The region benefits from coordinated initiatives led by the European Medicines Agency and programs such as Horizon Europe, which promote AI adoption in clinical research through funding and collaborative innovation frameworks.
The increasing collaboration between AI technology providers, academic institutions, and pharmaceutical companies is accelerating innovation in clinical trials. Companies, for example, Exscientia PLC, are playing a pivotal role by leveraging AI to optimize clinical development processes, including patient selection and predictive modeling. These collaborations are enabling the development of integrated AI platforms that support end-to-end trial optimization, from design to execution. Initiatives focused on standardization and interoperability are improving the scalability of AI solutions across different healthcare systems.
Asia Pacific AI-based Clinical Trials Solution Providers Market Trends
The Asia Pacific region is likely to be the fastest-growing region in 2026, driven by expanding healthcare digitization, increasing clinical trial activity, and strong government support for AI adoption. Countries such as China and India are investing heavily in digital health infrastructure, enabling the integration of AI across clinical development processes. The region benefits from large and diverse patient populations, which are highly valuable for training AI models and conducting scalable trials. Cost advantages and faster patient recruitment timelines make Asia Pacific an attractive destination for sponsors.
A notable trend in the region is the rise of indigenous AI healthcare companies and increased cross-border collaborations aimed at advancing clinical trial innovation. For example, Innovaccer, which has strong operational roots in India, is leveraging its AI-driven data platforms to integrate diverse healthcare datasets and support advanced analytics in clinical research environments. The company’s ability to unify real-world data and enable actionable insights reflects the broader regional shift toward data-centric and AI-enabled healthcare ecosystems.
Competitive Landscape
The global AI-based clinical trials solution providers market exhibits a moderately fragmented structure, driven by the presence of both established technology firms and a growing number of specialized AI startups focusing on niche clinical trial functions. The market is characterized by continuous innovation, with companies leveraging machine learning, natural language processing, and predictive analytics to enhance trial efficiency, reduce timelines, and improve patient outcomes. Increasing clinical trial complexity and demand for data-driven decision-making are intensifying competition, while high investment activity and strategic collaborations are shaping the landscape.
With key leaders including Unlearn.ai, Inc., Saama, Antidote Technologies, Inc., Deep6.ai, Innoplexus, Mendel Health Inc., Median Technologies, SymphonyAI, and BioAge Labs, Inc., the market reflects a mix of innovation-led startups and established analytics providers. These players compete through continuous product innovation, strategic partnerships with pharmaceutical companies and CROs, expansion of AI-enabled platforms across the clinical trial lifecycle, and geographic expansion to capture emerging market opportunities.
Key Industry Developments:
- In December 2025, Mass General Brigham announced the spinout of AIwithCare, a new company leveraging generative AI to enhance clinical trial screening and patient recruitment through its advanced platform, improving eligibility matching and accelerating trial enrollment processes.
- In July 2025, PhaseV announced the launch of its ClinOps AI platform, designed to transform clinical trial operations through precision-guided site selection, enabling sponsors to improve trial efficiency, optimize site performance, and enhance overall study outcomes using advanced analytics and machine learning.
Companies Covered in AI-based Clinical Trials Solution Providers Market
- Unlearn.ai, Inc.
- Saama
- Antidote Technologies, Inc.
- Phesi
- Deep6.ai
- Innoplexus
- Mendel Health Inc.
- Intelligencia AI
- Median Technologies
- SymphonyAI
- BioAge Labs, Inc.
- AiCure
- Consilx
- DeepLens.AI
- HaloHealth
- PHARMASEAL
- Ardigen
- Trials.ai
Frequently Asked Questions
The global AI-based clinical trials solution providers market is projected to reach US$3.4 billion in 2026.
The market is driven by the need to improve clinical trial efficiency, reduce costs and timelines, and enhance patient recruitment through AI-driven data analytics and automation.
The AI-based clinical trials solution providers market is expected to grow at a CAGR of 22.1% from 2026 to 2033.
Key opportunities lie in the expansion of decentralized and hybrid clinical trials, integration of real-world data, and development of AI-driven patient recruitment and monitoring solutions.
Unlearn.ai, Inc., Saama, Antidote Technologies, Inc., Phesi, Deep6.ai, Innoplexus, Mendel Health Inc., and Intelligencia AI are the leading players.








