
- Biotechnology
- Large and Small-Scale Bioprocessing Market
Large and Small-Scale Bioprocessing Market Size, Share, and Growth Forecast, 2026 - 2033
Large and Small-Scale Bioprocessing Market by Application (Monoclonal Antibodies (mAbs) & Biosimilars, Vaccines, Cell & Gene Therapy (CGT), Recombinant Proteins, Others), Product Type (Bioreactors & Fermenters, Filtration Systems, Chromatography Systems, Cell Culture Media, Single-Use Systems, Others), Workflow Stage (Upstream Processing, Downstream Processing, Fill-Finish), and Regional Analysis for 2026 - 2033
Large and Small-Scale Bioprocessing Market Share and Trends Analysis
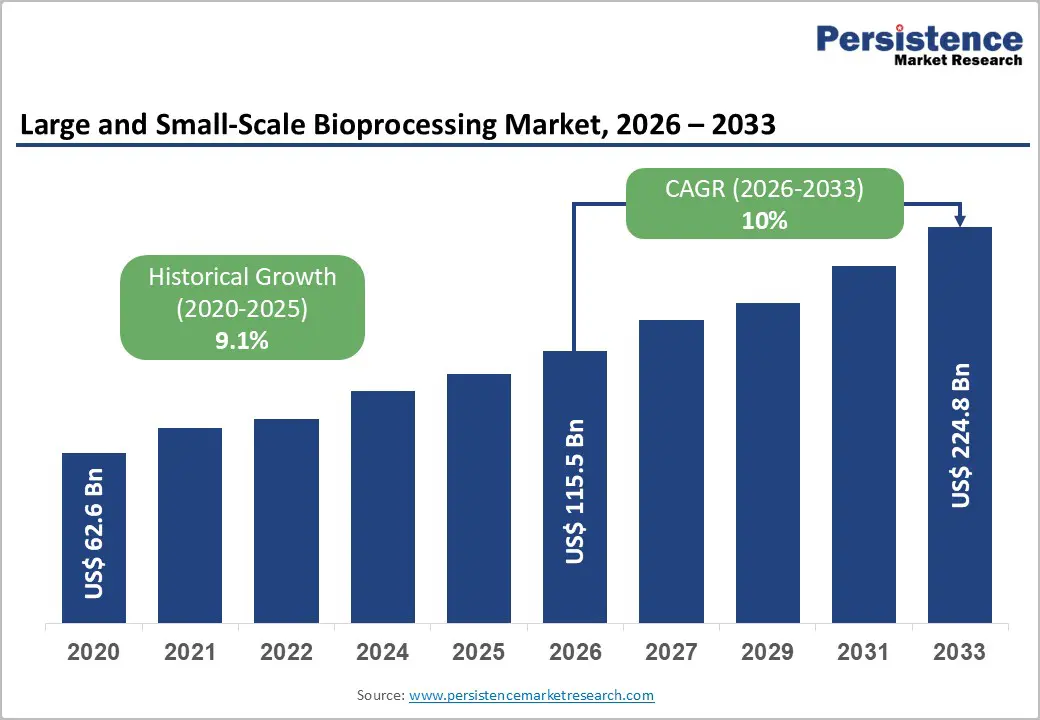
The global large and small-scale bioprocessing market size is likely to be valued at US$ 115.5 billion in 2026, and is projected to reach US$ 224.8 billion by 2033, growing at a CAGR of 10% during the forecast period 2026 - 2033.
This expansion is driven by rising monoclonal antibody (mAb) production volumes, increasing adoption of messenger RNA (mRNA) platforms, and a growing pipeline of cell and gene therapies (CGTs). At the same time, biopharmaceutical companies are increasingly outsourcing manufacturing to contract development and manufacturing organizations (CDMOs) to optimize capital allocation and accelerate commercialization timelines. The adoption of single-use technologies is further supporting this shift by reducing upfront infrastructure costs and enabling faster facility commissioning, particularly for multi-product and clinical-scale manufacturing environments.
Regulatory bodies such as the United States Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are streamlining approval pathways for biologics, which is accelerating demand for scalable and compliant manufacturing systems. The industry is progressively shifting toward modular and digitally integrated bioprocessing systems, where automation and data-driven optimization are enhancing process control and reducing variability. This transition is positioning manufacturers to efficiently produce high-value, low-volume therapies such as chimeric antigen receptor T-cell (CAR-T) treatments while maintaining cost discipline and operational flexibility.
Key Industry Highlights
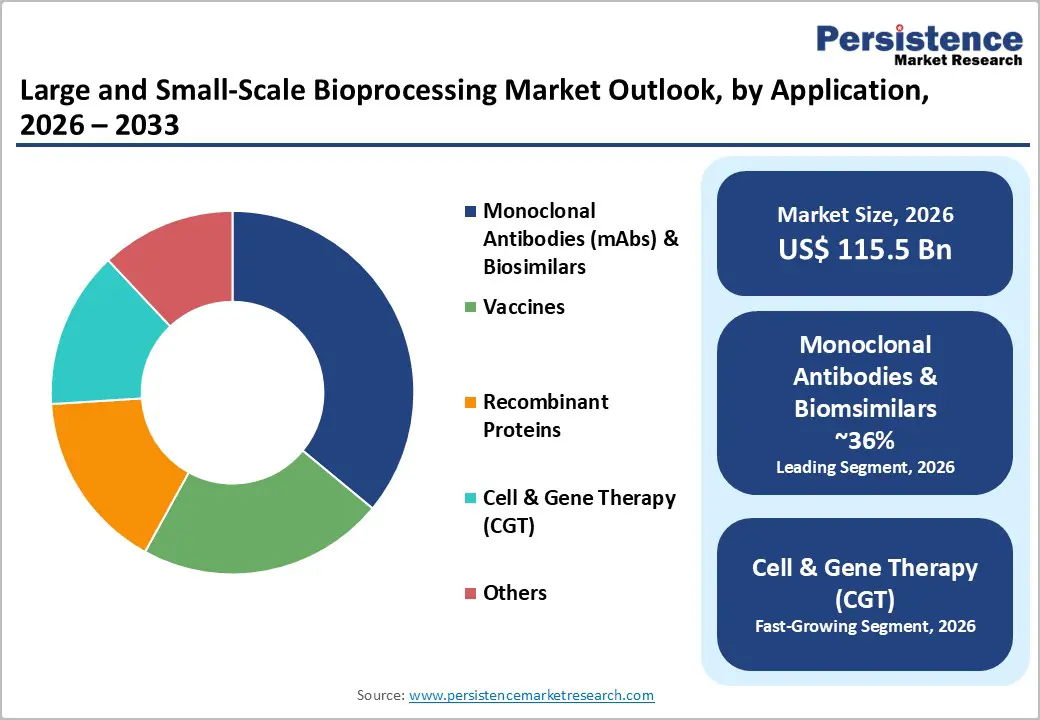
- Dominant Application: Monoclonal antibodies and biosimilars are projected to account for nearly 36% of total market revenue in 2026, driven by strong oncology demand and increasing biosimilar penetration.
- Fastest-growing Application: Cell and gene therapy (CGT) is expected to expand at the highest CAGR of approximately 17% during 2026 - 2033, supported by clinical pipeline expansion and regulatory acceleration.
- Product Leadership: Single-use systems are anticipated to hold around 32% revenue share in 2026, reflecting their role in enabling flexible manufacturing.
- Regional Dominance: North America is projected to hold approximately 40% of the global market value in 2026, supported by strong biopharmaceutical research and advanced manufacturing infrastructure.
- Fastest-growing Market: Asia Pacific is expected to be the fastest-growing market through 2033, with a CAGR of around 13.5%, driven by cost advantages and expanding biologics production capacity.
- September 2025: Thermo Fisher Scientific partnered with South Korea’s Dr. Park CDMO to supply advanced bioprocessing solutions, enhancing large-scale viral vector manufacturing capacity and accelerating CGT production.
| Key Insights | Details |
|---|---|
| Large and Small-Scale Bioprocessing Market Size (2026E) | US$ 115.5 Bn |
| Market Value Forecast (2033F) | US$ 224.8 Bn |
| Projected Growth (CAGR 2026 to 2033) | 10% |
| Historical Market Growth (CAGR 2020 to 2025) | 9.1% |
Market Factors - Growth, Barriers, and Opportunity
Market Drivers
Proliferating Biologics Pipelines and the Biosimilar Production Wave
The global biologics pipeline is driving sustained demand for bioprocessing infrastructure as regulatory approvals increasingly favor complex biologics. The U.S. FDA’s Center for Drug Evaluation and Research (CDER) approved 73 novel drugs in 2023, with biologics accounting for nearly 40%, indicating a structural transition in drug development. At the same time, patent expiries for high-revenue biologics such as adalimumab and trastuzumab are enabling large-scale biosimilar production across regions, including Europe, India, and South Korea. Similarly, the EMA has approved more than 90 biosimilars, each requiring dedicated upstream and downstream capacity. This trend is strengthening demand for core consumables such as chromatography resins, cell culture media, and large-scale single-use bioreactors, while also creating predictable, long-term procurement cycles for suppliers.
The CGT segment has introduced a distinct growth dynamic driven by high manufacturing complexity and limited capacity. The Alliance for Regenerative Medicine (ARM) reported over 3,600 active CGT clinical trials globally in 2024, while the number of approved therapies remains relatively low, highlighting a widening supply gap. Autologous therapies, including CAR-T treatments, require individualized manufacturing, which increases cost and operational intensity. This imbalance is creating a capacity shortfall estimated at US$ 3-5 billion annually in contract development and manufacturing organization (CDMO) services, presenting targeted opportunities for equipment providers and technology developers to address scalability constraints.
Digital Transformation and Process Intensification Reshaping Unit Economics
The integration of process analytical technology (PAT), artificial intelligence (AI), and digital twin modelling is improving biomanufacturing efficiency and shaping investment decisions. Regulatory frameworks are evolving, with the International Council for Harmonisation (ICH) guideline Q14 enabling real-time release testing (RTRT) and reducing reliance on end-of-batch quality checks. Manufacturers are adopting tools such as Raman spectroscopy, near-infrared (NIR), and mass spectrometry to enable continuous monitoring, which is reducing cycle times by nearly 30% and lowering material losses. The bioprocess automation software market is being driven by increasing adoption of manufacturing execution systems (MES) and digital platforms across clinical and commercial facilities.
Process intensification strategies are improving output while reducing facility size and capital requirements. Techniques such as N-1 perfusion and concentrated fed-batch operations are enabling higher productivity within smaller footprints. Continuous manufacturing is being adopted across a significant share of new production lines in North America, with artificial intelligence integration expected to scale further. Companies such as Samsung Biologics and Eli Lilly and Company are deploying digital twins and advanced process platforms to reduce the cost of goods (COGs) by up to 50%. These improvements are shifting capital allocation toward modular and single-use systems, expanding the consumables market and lowering entry barriers for emerging biotechnology firms.
Market Restraints
Chronic Skilled Workforce Shortage and GMP Training Deficits
The bioprocessing sector is facing a structural shortage of skilled labour, limiting production capacity and increasing operating costs. Cytiva’s Biomanufacturing State of the Industry survey indicates that nearly 80% of facilities reported hiring challenges in 2024, while its resilience index declined between 2021 and 2023, reflecting reduced ability to meet demand. The gap is arising from a mismatch between academic training and good manufacturing practice (GMP) requirements, where capabilities such as automation, digital integration, and continuous processing are in demand but underrepresented in formal education. The National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) estimates that new employees require up to 9 months of additional training, resulting in opportunity costs of about US$100,000 per worker. This constraint is becoming more pronounced in CGT, where biosafety protocols and patient-specific workflows are increasing workforce complexity.
Regional disparities are intensifying this constraint and shaping competitive dynamics. Asia Pacific is rapidly expanding manufacturing capacity, yet experienced talent in areas such as cell culture development and downstream purification remains limited. South Korea is supporting large-scale CDMO operations but is still depending on international expertise for advanced roles. India is scaling its CDMO sector, though infrastructure reliability and curriculum alignment with biologics manufacturing pose challenges. Companies are responding by investing in internal training ecosystems and specialized academies, which are improving workforce readiness and throughput reliability. This shift is positioning talent development as a strategic lever, with firms possessing integrated training capabilities gaining operational advantages beyond equipment and technology investments.
Polymer Supply Chain Vulnerability and Single-Use Component Integrity Risk
The rapid adoption of single-use bioprocessing systems is resulting in excessive dependency on a limited group of polymer suppliers. Critical components, such as bioreactor bags and tubing, are manufactured from materials including ultra-low-density polyethylene (ULDPE), ethylene vinyl acetate (EVA), and multilayer nylon films. The supply of irradiation-grade bioprocess films is controlled by a small number of compounders, which has exposed structural fragility during periods of disruption. For example, during the COVID pandemic, lead times for single-use assemblies exceeded 52 weeks, while prices increased by up to 80%. Regulatory requirements for leachables and extractables (L&E), governed by United States Pharmacopeia (USP) standards such as Chapters 665 and 1665, are further constraining supplier flexibility, as any material change triggers lengthy revalidation cycles that can extend beyond a year.
The financial impact of supply disruptions is significant and often asymmetric. A single failure in a bioreactor system can result in batch losses ranging from U$ 1 million to US$ 5 million, along with compliance and reporting implications. Even minor defects, such as leaks in mixing systems, are leading to immediate material losses and operational delays. Manufacturers are responding by diversifying sourcing and investing in regional production capacity, including new polymer facilities and expanded membrane manufacturing. However, supply chain resilience requires multi-year capital investment and cannot be achieved in the short term. Companies are therefore adopting risk mitigation strategies such as dual sourcing, inventory buffering, and hybrid facility designs that combine single-use and stainless-steel systems to maintain operational continuity.
Market Opportunities
Precision Fermentation and Industrial Bioprocessing for the Bio-Based Economy
Precision fermentation is emerging as a distinct growth driver within the broader bioprocessing market, expanding beyond pharmaceutical manufacturing into the bio-based economy. The expansion of this process has generated substantial demand for core bioprocessing infrastructure, including bioreactors, culture media, downstream purification systems, and process monitoring tools. Key applications include dairy protein alternatives, animal-free heme proteins, human milk oligosaccharides (HMOs), and recombinant collagen, all of which rely on engineered microbial systems. Policy frameworks such as the European Commission (EC)’s Farm to Fork strategy and the United States Bioeconomy Executive Order are supporting this transition by directing funding toward sustainable production platforms, thereby accelerating industrial-scale fermentation investments.
From a supplier perspective, precision fermentation is creating a new addressable market with technical requirements distinct from those of traditional biologics manufacturing. Facilities are operating at significantly larger volumes, often between 50,000 and 200,000 liters, using microbial hosts such as Escherichia coli (E. coli) and yeast, which require specialized aeration, agitation, and downstream processing configurations. This shift is increasing demand for industrial-grade fermentation equipment and high-throughput purification systems, giving companies with microbial processing expertise an advantage. Equipment providers focused on mammalian cell culture are facing a strategic inflection point, as expanding into industrial fermentation technologies will be critical to capturing growth in this segment. Early investment in tailored product portfolios and partnerships is positioning suppliers to benefit from scaling demand as precision fermentation reaches broader commercial adoption over the next decade.
Digitalization and AI-Driven Process Optimization
Bioprocessing is integrating AI, machine learning (ML), and digital twin technologies to enhance yield, reduce batch variability, and improve process reliability. These tools enable real-time decision-making by combining process data with predictive models, thereby strengthening control over complex biological systems. Analysis from the Organisation for Economic Co-operation and Development (OECD) indicates that digital biomanufacturing adoption is expected to grow at a CAGR of over 15%, outpacing traditional equipment segments. Manufacturers are deploying advanced analytics across upstream and downstream operations to identify deviations early and optimize production parameters. This transition is improving asset utilization and reducing unplanned downtime, which is directly influencing cost structures and throughput efficiency.
The growing reliance on digital platforms is also leading to the creation of new value pools for software providers and automation vendors across the bioprocessing lifecycle. Predictive analytics and integrated monitoring systems are enabling manufacturers to anticipate equipment failures, minimize material losses, and maintain consistent product quality. These capabilities also support compliance with evolving regulatory expectations by enhancing data integrity and traceability. The cumulative impact of digital integration is expected to unlock efficiency gains of approximately US$ 10-15 billion across the value chain by 2030. Companies that are investing in interoperable systems and scalable digital architectures are positioning themselves to capture these gains while strengthening operational resilience and accelerating time to market.
Category-wise Analysis
Application Insights
Monoclonal antibodies (mAbs) and biosimilars are anticipated to hold about 38% of the large and small-scale bioprocessing market revenue share in 2026. This position is supported by the maturity of Chinese hamster ovary (CHO) cell culture systems and a strong base of approved biologics generating over US$ 220 billion in annual sales. Biosimilar production is strengthening demand because each manufacturer requires dedicated and fully validated manufacturing lines, increasing infrastructure duplication. Companies such as Samsung Biologics, Celltrion, and Biocon Biologics are expanding large-scale capacity to meet this demand. The rise of antibody-drug conjugates (ADCs), which are growing at over 20% annually, is adding complexity to downstream processing by requiring specialized conjugation capabilities, thereby increasing investment in purification and handling systems.
Cell and gene therapy is poised to emerge as the fastest-growing application, with an estimated CAGR of about 17% from 2026 to 2033. The U.S. FDA is projecting up to 20 approvals annually from 2025, indicating a strong pipeline and sustained demand for manufacturing capacity. Autologous therapies such as CAR-T treatments require patient-specific production in controlled environments, which increases cost and operational complexity. At the same time, allogeneic platforms are moving toward standardized large-scale production, which will expand bioprocessing volumes in the coming years. Viral vector manufacturing, particularly for adeno-associated virus (AAV) and lentiviral vector (LVV) systems, remains constrained, with limited global capacity for late-stage production. This gap is creating targeted investment opportunities in specialized facilities and scalable production technologies.
Product Type Insights
Bioreactors and fermenters are set to capture roughly 32% of the market revenue share in 2026, driven by their central role in cell culture and fermentation across all scales, as well as their capital-intensive nature. The segment includes stainless steel stirred-tank systems, single-use bioreactors, rocking platforms, and hollow-fibre configurations. Advanced platforms from suppliers such as Thermo Fisher Scientific and Sartorius are improving scalability and process control, which is supporting adoption in both clinical and commercial settings. Stainless steel systems remain relevant for high-volume production above 10,000 liters, particularly for monoclonal antibodies and industrial fermentation, while single-use formats are expanding into larger capacity ranges, enabling more flexible facility design and faster technology transfer.
Single-use systems and assemblies are likely to register the fastest growth through 2033. This segment includes disposable bags, tubing, connectors, mixers, and sensors that reduce contamination risk and simplify operations. These systems are reducing capital expenditure by up to US$ 100 million per facility and shortening deployment timelines, making them attractive to CDMOs and multi-product facilities. Adoption is increasing at a commercial scale as larger vessel formats are becoming viable, removing previous capacity constraints. Asia Pacific is driving much of this growth through large-scale biologics investments and new facility construction. Suppliers that are expanding single-use portfolios and regional manufacturing capabilities are positioning themselves to capture sustained demand as flexible production models gain wider acceptance.
Regional Insights
North America Large and Small-Scale Bioprocessing Market Trends
North America is anticipated to dominate in 2026, accounting for about 40% of the large and small-scale bioprocessing market share. The United States is driving regional performance through its concentration of commercial biopharmaceutical manufacturing sites and a strong biotechnology innovation ecosystem. Major production hubs are supporting both large-scale biologics and early-stage development, which is sustaining demand across upstream and downstream systems. A steady flow of clinical-stage assets is reinforcing the need for flexible manufacturing infrastructure, particularly for small-batch and high-value therapies.
The regulatory environment is accelerating technology adoption and capacity expansion across the region. The U.S. FDA is enabling advanced manufacturing through programs such as the Emerging Technology Program (ETP), which supports the adoption of continuous processing and reduces approval timelines. This framework is improving cost efficiency and encouraging investment in next-generation platforms. The anticipated rise in CGT approvals is driving capital allocation toward viral vector and mRNA lipid nanoparticle (LNP) manufacturing. CDMOs are also expanding capacity to address this demand, while Canada is strengthening its position through initiatives supported by the National Research Council Industrial Research Assistance Program (NRC-IRAP), particularly in mRNA and advanced biologics production.
Europe Large and Small-Scale Bioprocessing Market Trends
In Europe, the market for large and small-scale bioprocessing is spearheaded by Germany, Switzerland, and the United Kingdom due to the concentration of pharmaceutical manufacturers and bioprocessing equipment suppliers. Companies such as Roche, Novartis, and AstraZeneca are anchoring demand, while suppliers such as Sartorius and Eppendorf are strengthening the regional technology base. This integrated ecosystem supports both innovation and large-scale manufacturing, enabling steady expansion across biologics and advanced therapies.
Policy alignment with sustainability and healthcare resilience is shaping investment priorities across Europe. The European Union (EU) Green Deal and the Pharmaceutical Strategy for Europe are encouraging local production of critical medicines, thereby accelerating capacity expansion across the European Economic Area (EEA). Initiatives in countries such as Germany are allocating significant funding to biotechnology and industrial biomanufacturing, supporting applications such as bio-based chemicals and enzymes. The United Kingdom is strengthening its position in CGT through institutions such as the Cell and Gene Therapy Catapult, while regulatory engagement with the Medicines and Healthcare products Regulatory Agency (MHRA) is improving market access. These coordinated efforts are positioning Europe as a stable and policy-driven market with strong capabilities in both pharmaceutical and industrial bioprocessing.
Asia Pacific Large and Small-Scale Bioprocessing Market Trends
The Asia Pacific market is set to record the highest growth at a projected CAGR of approximately 13.5% through 2033. Key markets such as South Korea, China, India, Japan, and Singapore are driving expansion through targeted policy support and capacity investments. Each country is developing a distinct positioning, ranging from large-scale contract development and manufacturing organization operations to cost-efficient biosimilar production and advanced therapy capabilities. This regional diversity is strengthening supply chain resilience and attracting global biopharmaceutical investment.
South Korea is scaling large CDMO infrastructure through facilities such as Samsung Biologics, supported by national bioeconomy initiatives and infrastructure funding. China is accelerating domestic biologics production under the National Medical Products Administration (NMPA), which is reducing approval timelines and enabling faster commercialization. India is expanding its presence as a cost-competitive manufacturing hub, supported by the Production Linked Incentive (PLI) scheme and investments in GMP-compliant facilities. Singapore is focusing on high-value segments such as CGT, supported by institutions such as the Agency for Science, Technology and Research (A*STAR). These coordinated strategies are positioning Asia Pacific as a critical growth engine with increasing influence over global bioprocessing capacity and supply dynamics.

Competitive Landscape
The global bioprocessing market structure is showing moderate consolidation, with Danaher, Thermo Fisher, Sartorius, and Merck controlling more than half of the revenue. This concentration is strongest in single-use systems, where the same companies dominate global share. Below this tier, a fragmented group of specialized players is operating across niche segments. Companies such as Repligen Corporation are focusing on perfusion and chromatography solutions, Eppendorf SE is addressing small-scale bioreactors and mixing systems, and Avantor is supplying process chemicals and media. Lonza Group and Corning Incorporated are also contributing through contract manufacturing and cell culture technologies, creating a layered competitive structure with both scale and specialization.
Competitive positioning is shifting toward integrated platforms rather than standalone products. Danaher Corporation is leveraging its Cytiva and Pall businesses to offer end-to-end solutions across the bioprocessing workflow, which is appealing to CDMOs managing complex portfolios. Sartorius AG is integrating hardware with PAT and data analytics platforms to support quality by design (QbD) manufacturing. Thermo Fisher is aligning bioprocessing systems with analytical and testing capabilities to cover the full development lifecycle. Merck is strengthening its presence in single-use and filtration systems, particularly in downstream processing. At the same time, emerging companies such as PBS Biotech, Meissner Filtration Products, Inc., and Univercells Technologies are targeting high-growth niches in CGT and viral vector manufacturing, where specialized solutions are gaining traction.
Key Industry Developments
- In March 2026, researchers at Hannover Medical School developed a benchtop bioreactor protocol that enables reproducible, scalable production of macrophages from induced pluripotent stem cells (iPSCs), yielding tens of millions of cells per harvest with minimal manual intervention. The semi-automated system improves consistency and accessibility for early-stage research, supporting applications in disease modelling, drug testing, and immunotherapy development.
- In February 2026, Glatt and PreferCo launched an AI-enabled bioprocess scale-up centre in Hyderabad to support precision fermentation and bridge the gap between laboratory research and commercial manufacturing, strengthening India’s BioE3 bioeconomy push. The facility offers up to 1,500-liter bioreactor capacity with advanced automation and process analytics, enabling faster scale-up, reduced risk, and broader applications.
- In June 2025, Univercells Technologies unveiled the scale-X nitro controller, a compact and cost-efficient system designed to scale viral vector and advanced therapy manufacturing from research to commercial production within months. The controller supports large-scale fixed-bed bioreactors and reduces production costs by up to 82%, enabling faster deployment, improved scalability, and broader access to advanced biomanufacturing.
Companies Covered in Large and Small-Scale Bioprocessing Market
- Thermo Fisher Scientific Inc.
- Sartorius AG
- Danaher Corporation
- Merck KGaA
- Eppendorf SE
- Getinge AB
- Lonza Group AG
- Agilent Technologies, Inc.
- Repligen Corporation
- Corning Incorporated
- Avantor, Inc.
- Bio-Rad Laboratories, Inc.
- FUJIFILM Diosynth Biotechnologies
- WuXi Biologics
- Boehringer Ingelheim International GmbH
Frequently Asked Questions
The global large and small-scale bioprocessing market is projected to reach US$ 115.5 billion in 2026.
Rising production volumes of monoclonal antibodies, increasing adoption of mRNA platforms, and a growing pipeline of cell and gene therapies are fueling the market.
The market is poised to witness a CAGR of 10% from 2026 to 2033.
Streamlining of approval pathways for biologics by regulatory bodies such as the U.S. FDA and the EMA, and industry-wide shift toward modular and digitally integrated bioprocessing systems are key market opportunities.
Thermo Fisher Scientific Inc., Sartorius AG, Danaher Corporation, and Merck KGaA are some of the key players in the market.







