
- Medical Devices
- Lyophilization Equipment Market
Lyophilization Equipment Market Size, Share, Growth, and Regional Forecast, 2026 to 2033
Lyophilization Equipment Market by Product (Tray-Style Lyophilizers, Manifold Lyophilizers, Rotary Lyophilizers, Bench-Top Lyophilizers, Industrial Lyophilizers, and Pilot-Scale Lyophilizers), Application (Pharmaceutical Manufacturing, Biotechnology, Food & Beverage Processing, Medical & Diagnostics, and Others) End-user (Pharmaceutical & Biopharmaceutical Companies, Contract Manufacturing Organizations (CMOs), Research & Academic Institutes, Food Processing Companies, and Others), and Regional Analysis from 2026 to 2033
Lyophilization Equipment Market Share and Trends Analysis
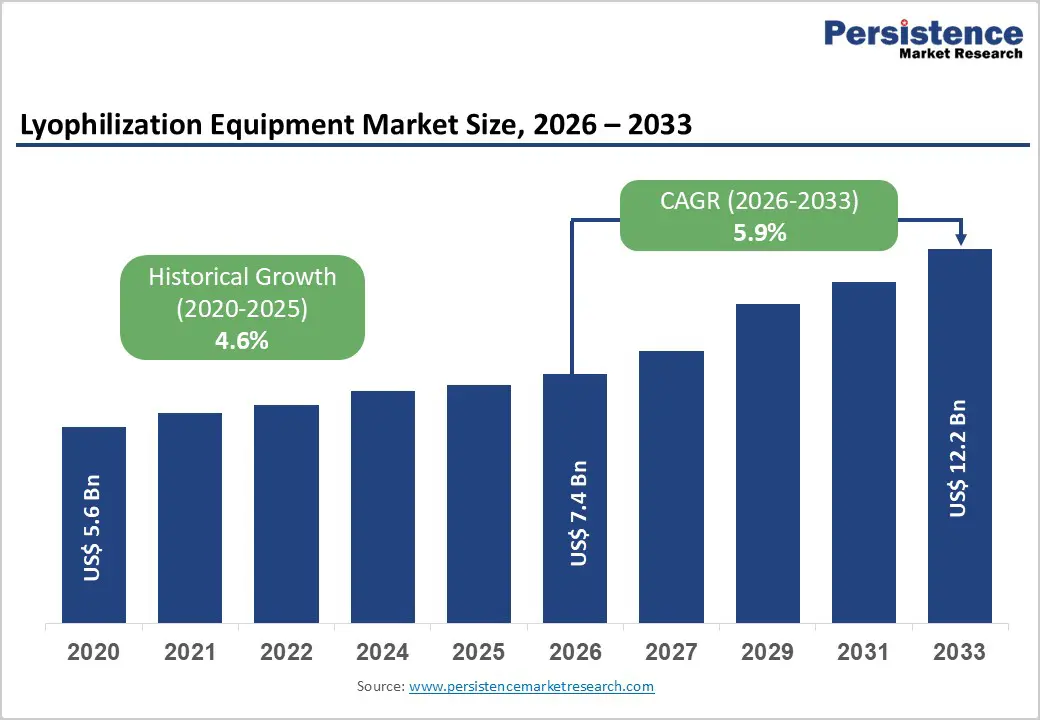
The global lyophilization equipment market size is estimated to grow from US$ 7.4 billion in 2026 to US$ 12.2 billion by 2033. The market is projected to record a CAGR of 5.9% during the forecast period from 2026 to 2033.
The global adoption of lyophilization equipment continues to expand as pharmaceutical and biotechnology manufacturers increasingly rely on advanced drying technologies to preserve temperature-sensitive formulations and improve product shelf stability. Rising production of biologics, vaccines, injectable therapeutics, and specialty drugs has intensified the need for controlled dehydration processes that maintain structural integrity and therapeutic efficacy during storage and transportation.
Deployment of freeze-drying systems across commercial manufacturing plants, research laboratories, contract manufacturing facilities, and food processing operations is supporting steady market progression. Growing emphasis on formulation stability, global drug distribution without strict cold-chain dependence, and scale-up of sterile manufacturing capacity are strengthening equipment demand worldwide.
Key Industry Highlights:
- Leading Region: North America holds the largest share at 46.7%, supported by strong biopharmaceutical manufacturing capacity, early implementation of automated freeze-drying systems, mature regulatory frameworks, and the presence of major equipment suppliers.
- Fastest-Growing Region: Asia Pacific is witnessing the fastest expansion due to rapid growth in pharmaceutical production, increasing investments in biologics manufacturing, improving healthcare infrastructure, and rising demand for shelf-stable drug formulations.
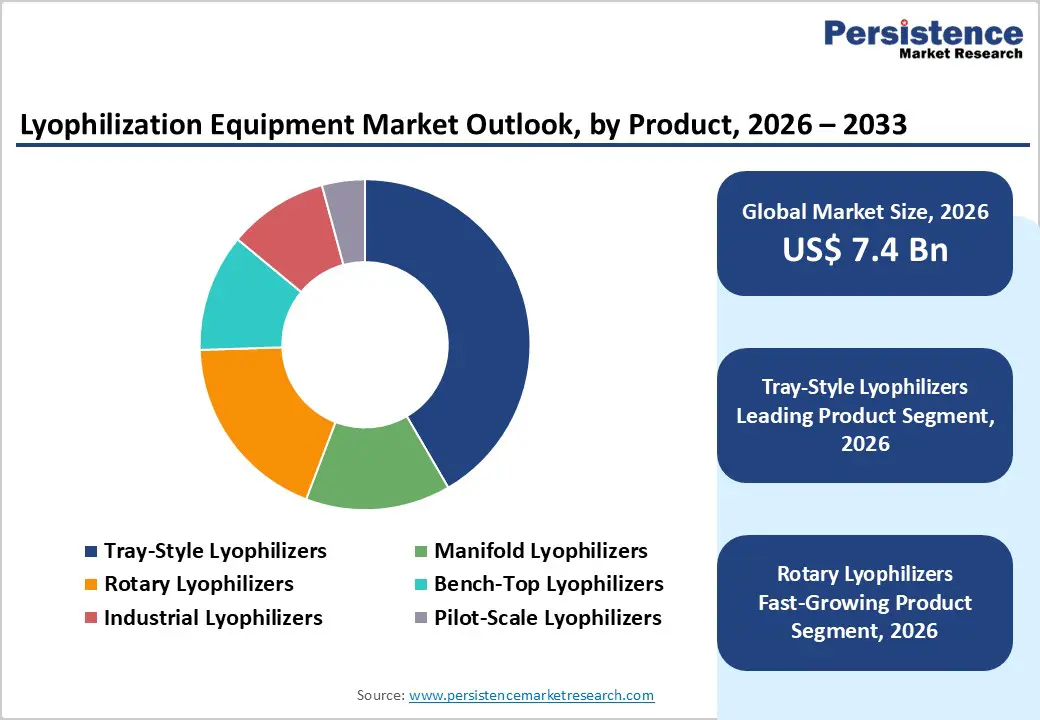
- Leading Product Segment: Tray-style lyophilizers dominate the market owing to their suitability for large-batch sterile production, consistent heat transfer performance, and widespread use in commercial pharmaceutical manufacturing.
- Fastest-Growing Product Segment: Rotary lyophilizers are gaining momentum as industries seek flexible processing solutions for food, nutraceutical, and specialty applications requiring efficient continuous drying.
- Leading Application Segment: Pharmaceutical manufacturing remains the primary application area, driven by expanding biologics pipelines and increasing global demand for stable injectable therapies.
- Fastest-Growing Application Segment: Food & beverage processing is growing rapidly as consumer demand rises for premium freeze-dried products with extended shelf life and preserved nutritional quality.
| Key Insights | Details |
|---|---|
| Lyophilization Equipment Market Size (2026E) | US$ 7.4 Bn |
| Market Value Forecast (2033F) | US$ 12.2 Bn |
| Projected Growth (CAGR 2026 to 2033) | 5.9% |
| Historical Market Growth (CAGR 2020 to 2025) | 4.6% |
Market Dynamics
Driver - Increasing Biologic Drug Development and Stringent Product Stability Requirements
The growing complexity of modern therapeutics is significantly accelerating demand for advanced preservation technologies capable of maintaining molecular integrity throughout storage and distribution. Biopharmaceutical products such as monoclonal antibodies, vaccines, recombinant proteins, and cell-derived therapies are highly sensitive to temperature fluctuations and moisture exposure, making long-term stability a critical manufacturing priority. Freeze-drying has emerged as a preferred stabilization approach because it extends shelf life while preserving biological activity, enabling safe global transportation and flexible storage conditions.
Rapid expansion of biologics pipelines, combined with increasing approvals of injectable and specialty drugs, is pushing pharmaceutical manufacturers to expand lyophilization capacity across commercial and clinical production facilities. Regulatory agencies are also emphasizing product quality, sterility assurance, and consistent dosage performance, encouraging adoption of validated and automated drying systems. The rise of global vaccination programs, pandemic preparedness initiatives, and temperature-sensitive therapeutics has further reinforced the importance of reliable drying infrastructure. Additionally, pharmaceutical companies are investing in scalable equipment that supports seamless transition from laboratory development to commercial manufacturing. As drug formulations become more complex and stability expectations tighten, lyophilization equipment continues to play a central role in ensuring product efficacy, supply chain resilience, and regulatory compliance across modern pharmaceutical production environments.
Restraints - High Capital Investment, Operational Complexity, and Process Optimization Challenges
The implementation of advanced freeze-drying systems presents notable financial and operational barriers for manufacturers. Industrial lyophilizers require significant upfront capital expenditure, particularly when integrated with automated loading systems, clean-in-place technologies, and GMP-compliant sterile environments. Installation demands specialized facility design, including controlled cleanrooms, vacuum infrastructure, and energy-intensive refrigeration systems, increasing total ownership cost. For small and mid-scale manufacturers, these investments can delay modernization or capacity expansion decisions.
Operational complexity further limits adoption. Lyophilization is a highly sensitive process involving freezing, primary drying, and secondary drying phases that must be precisely controlled to prevent product collapse or loss of activity. Process development often requires extensive experimentation and technical expertise, prolonging validation timelines. Skilled personnel capable of managing thermal profiles, chamber pressure, and formulation behavior are not universally available, particularly in emerging manufacturing hubs. Energy consumption and long cycle times also affect production efficiency, increasing operational expenditure. Additionally, strict regulatory validation requirements demand extensive documentation and performance qualification, creating additional compliance burdens. Variability in batch performance and scale-up challenges between pilot and commercial production remain persistent concerns, collectively moderating adoption despite growing demand for stabilized pharmaceutical and food products.
Opportunity - Expansion of Contract Manufacturing, Continuous Processing, and Advanced Automation Integration
Evolving pharmaceutical outsourcing models are opening substantial growth avenues for equipment suppliers focused on flexible and high-efficiency drying solutions. Contract development and manufacturing organizations are rapidly expanding sterile manufacturing capabilities to support small biotech firms lacking internal infrastructure. This shift is increasing demand for modular, multi-product lyophilizers capable of handling diverse formulations and smaller batch sizes. Automation technologies, including robotic loading, real-time monitoring sensors, and data-driven process control, are creating opportunities to improve throughput while minimizing contamination risks and operator dependency.
The industry is also witnessing growing interest in continuous and semi-continuous freeze-drying concepts designed to reduce cycle duration and energy consumption. Digitalization initiatives integrating process analytical technology and predictive analytics enable improved batch consistency and faster scale-up, enhancing manufacturing productivity. Beyond pharmaceuticals, rising consumption of premium freeze-dried foods, nutraceuticals, and diagnostic reagents is expanding application scope across multiple industries. Emerging markets are investing heavily in domestic drug production and cold-chain-independent formulations, further stimulating equipment demand. Sustainability considerations are encouraging the development of energy-efficient refrigeration systems and optimized drying cycles. As manufacturers prioritize flexibility, automation, and operational efficiency, next-generation lyophilization platforms are positioned to benefit from long-term structural shifts in global manufacturing strategies.
Category-wise Analysis
By Product Insights
The tray-style lyophilizers segment is projected to anchor the global market in 2026, commanding a significant 41.6% revenue share. This leadership stems from the indispensable role these systems play in high-volume industrial applications, particularly in the stabilization of bulk active pharmaceutical ingredients (APIs) and heat-sensitive food products. Tray-style units offer superior shelf-area-to-footprint ratios, allowing for the uniform drying of diverse materials ranging from biological samples to nutraceuticals. Their design facilitates precise shelf temperature mapping and pressure control, ensuring that delicate structures remain intact during the sublimation process. As global manufacturing scales up to meet the demands for shelf-stable biologicals and high-quality dehydrated foods, the robustness and versatility of tray-based systems remain unparalleled. Continuous engineering improvements in automated loading and unloading systems, coupled with advanced CIP/SIP (Clean-in-Place/Sterilize-in-Place) capabilities, further solidify their position as the preferred choice for facilities requiring consistent, large-batch productivity and stringent contamination control.
By Application Insights
The pharmaceutical manufacturing segment is anticipated to emerge as the primary revenue generator in 2026, capturing a 52.4% market share. This sector’s expansion is inextricably linked to the global boom in biopharmaceutical development, including the rise of mRNA vaccines, monoclonal antibodies, and personalized cell therapies. Because these advanced therapeutics are often chemically unstable in liquid form, lyophilization serves as the critical "gold standard" for extending shelf life and ensuring potency during global distribution. The shift toward injectable drugs that require reconstitution at the point of care has necessitated massive investments in high-capacity freeze-drying infrastructure. Furthermore, the industry is increasingly adopting Continuous Manufacturing and Process Analytical Technology (PAT) to monitor critical parameters like primary drying endpoints in real-time. This integration of digital monitoring and specialized equipment ensures high batch-to-batch reproducibility, making the pharmaceutical sector the most consistent driver of high-end lyophilization technology adoption worldwide.
By End-user Insights
The pharmaceutical & biopharmaceutical companies segment is expected to maintain its market stronghold in 2026 with a 47.3% value share. These entities represent the core engine of the market, fueled by aggressive R&D pipelines and the transition from laboratory-scale pilot studies to full-scale commercialization. These organizations invest heavily in sophisticated, integrated lyophilization lines that feature robotic isolation technology to minimize human intervention and environmental risk. The dominance of this segment is also bolstered by strict regulatory frameworks (such as FDA and EMA standards) that mandate the use of high-precision equipment to guarantee drug safety. Strategic partnerships between equipment manufacturers and drug developers are increasingly common, leading to the creation of customized freeze-drying cycles tailored to specific molecular profiles. As global health initiatives continue to prioritize vaccine accessibility and chronic disease management, the procurement of advanced freeze-drying systems by these corporations remains the primary catalyst for market sustainability and technical evolution.
Regional Insights
North America Lyophilization Equipment Market Trends
North America is poised to remain the preeminent force in the global lyophilization equipment landscape for 2026, securing a 46.7% value share. The United States, acting as the primary hub for biomedical innovation, drives this dominance through a highly concentrated ecosystem of elite research universities, multinational pharmaceutical giants, and cutting-edge biotech startups. The region’s leadership is defined by a rapid transition toward "smart" lyophilization, where AI-driven modeling and cloud-based monitoring optimize energy consumption and drying cycles.
North American manufacturers benefit from a regulatory environment that encourages the adoption of next-generation safety features, such as advanced leak detection and eco-friendly refrigerants. Furthermore, the strong presence of local industry leaders like Labconco and Millrock Technology ensures a steady supply of high-precision instruments. The region also sees significant capital expenditure directed toward the modernization of aging production facilities, with a focus on replacing legacy systems with automated, high-efficiency units that support the increasing volume of specialized, shelf-stable healthcare products.
Europe Lyophilization Equipment Market Trends
Europe’s lyophilization equipment market is set for a period of steady, high-value growth in 2026, anchored by the manufacturing powerhouses of Germany, France, Switzerland, and Italy. The European market is characterized by a profound emphasis on engineering precision and environmental sustainability. With strict EU regulations regarding greenhouse gas emissions, there is a significant movement toward the adoption of liquid nitrogen (LN2) cooling systems and natural refrigerants over traditional CFC-based models. European hospitals and specialized research institutes are early adopters of pilot-scale lyophilizers for clinical trials, fostering a robust market for versatile, medium-capacity units.
Additionally, the region’s strong heritage in machinery manufacturing represented by companies like GEA and Azbil Telstar supports a culture of continuous technical refinement. The integration of Industry 4.0 principles, such as predictive maintenance and remote diagnostic capabilities, is becoming a standard requirement for European procurement teams. This focus on long-term operational reliability and specialized application support ensures that Europe remains a critical pillar of the global freeze-drying industry.
Asia Pacific Lyophilization Equipment Market Trends
The Asia Pacific region is forecast to be the most dynamic growth theater, registering an impressive CAGR of approximately 8.0% between 2026 and 2033. This accelerated trajectory is fueled by massive infrastructure upgrades in China and India, where the government is incentivizing the localization of drug and vaccine production. As these nations transition from being primarily generic drug hubs to becoming centers for biologic innovation, the demand for high-spec lyophilization equipment has surged. Furthermore, the region’s vast food and beverage sector is increasingly adopting freeze-drying technology to produce high-value, nutrient-dense products for a burgeoning middle class.
Cost-effective manufacturing and the presence of major regional players like Tofflon and IlShin BioBase have improved the accessibility of industrial-grade systems. Efforts to strengthen regional supply chains and increase domestic self-sufficiency in medical technologies are prompting significant private and public investments. This rapid modernization, combined with an expanding clinical research landscape, positions Asia Pacific as the most influential emerging market for the next decade of lyophilization technology.

Competitive Landscape
The global lyophilization equipment market is highly competitive, with strong participation from ATS Corporation, Azbil Telstar, S.L.U., BÜCHI Labortechnik AG, Cuddon Freeze Dry, GEA Group Aktiengesellschaft, and HOF Sonderanlagenbau GmbH. These players leverage extensive global distribution networks, strong brand equity, and continuous innovation in vacuum technology, thermal control, and aseptic automation to address a wide range of pharmaceutical and food processing applications.
Rising demand for biopharmaceuticals, increasing stability requirements for vaccines, and the shift toward shelf-stable injectable drugs are driving innovation. Manufacturers are focusing on advanced PAT tools, robotic loading systems, energy-efficient condensers, and digital workflow integration while strengthening biotech collaborations and expanding in emerging markets for efficient, scalable freeze-drying solutions.
Key Industry Developments:
- In October 2025, IS-Instruments secured funding support to advance next-generation innovations in freeze-drying technologies aimed at improving process efficiency and product stability. The initiative is expected to accelerate development of more energy-efficient systems and enhance scalability for pharmaceutical and biotechnology manufacturing applications.
- In May 2024, Millrock Technology announced a strategic collaboration with LyoWave to advance next-generation freeze-drying solutions integrating innovative microwave heating technology originally developed at Purdue University. The partnership followed LyoWave’s licensing of the underlying intellectual property through the Purdue Innovates Office of Technology Commercialization, aiming to accelerate commercialization of faster and more energy-efficient lyophilization processes.
Companies Covered in Lyophilization Equipment Market
- ATS Corporation
- Azbil Telstar, S.L.U.
- BÜCHI Labortechnik AG
- Cuddon Freeze Dry
- GEA Group Aktiengesellschaft
- HOF Sonderanlagenbau GmbH
- IlShin BioBase Co., Ltd.
- Labconco Corporation
- MechaTech Systems Ltd
- Millrock Technology, Inc.
- Optima Packaging Group GmbH
- ZIRBUS technology GmbH
- IMA S.p.A.
- SP Industries, Inc.
- Tofflon Science and Technology Group Co., Ltd.
- Others
Frequently Asked Questions
The global lyophilization equipment market is projected to be valued at US$ 7.4 Bn in 2026.
Rising production of biologics, vaccines, and injectable pharmaceuticals requiring long-term stability and extended shelf life is the primary factor driving adoption of lyophilization equipment globall
The global lyophilization equipment market is poised to witness a CAGR of 5.9%between 2026 and 2033.
Growing demand for personalized medicines, biologics, and shelf-stable food products along with the expansion of CMOs and e-commerce food supply chain is creating strong opportunities for advanced, automated, and small-batch lyophilization systems.
ATS Corporation, Azbil Telstar, S.L.U., BÜCHI Labortechnik AG, Cuddon Freeze Dry, GEA Group Aktiengesellschaft, and HOF Sonderanlagenbau GmbH are some of the key players in the lyophilization equipment market.







