- Biotechnology
- Lipid Nanoparticle Manufacturing Market
Lipid Nanoparticle Manufacturing Market Size, Share, Growth, and Regional Forecast, 2026 to 2033
Lipid Nanoparticle Manufacturing Market by Type of LNP (Solid Lipid Nanoparticles (SLNs), Nanostructured Lipid Carriers (NLCs)), Therapeutic Area (Infectious Diseases, Rare Diseases, Oncological Disorders, Neurodegenerative Disorders, Others), End-user (Pharmaceutical and Biotechnology Companies, Academic & Research Institutes, Others), and Regional Analysis from 2026 to 2033
Lipid Nanoparticle Manufacturing Market Share and Trends Analysis
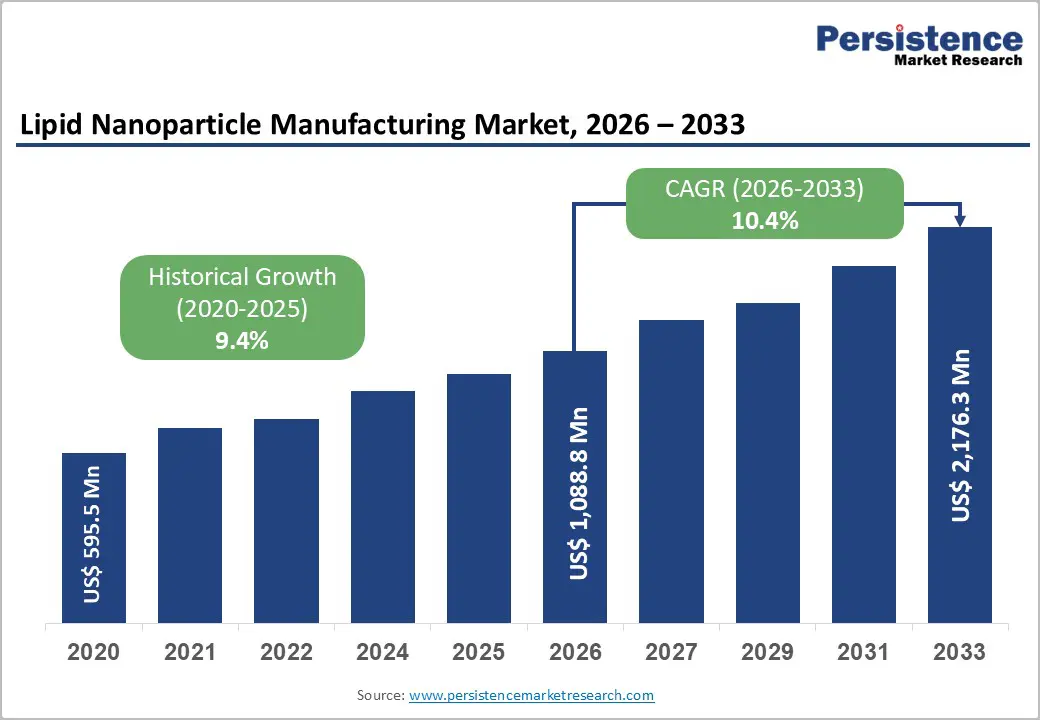
The global lipid nanoparticle manufacturing market size is likely to grow from US$ 1,088.8 million in 2026 to US$ 2,176.3 million by 2033, projected to record a CAGR of 10.4% during the forecast period from 2026 to 2033.
The global lipid nanoparticle manufacturing market is expanding steadily, driven by digital healthcare adoption, telehealth, and analytics. North America dominates with advanced infrastructure and strict regulations, ensuring high-quality production. Asia Pacific is the fastest-growing region, supported by expanding healthcare facilities, government digital initiatives, rising patient awareness, and increased investments in software, services, and interoperable manufacturing solutions.
Key Industry Highlights:
- Dominant Segment: Lipid nanoparticles for mRNA and gene therapies hold the largest share in 2025 with 81.3%, driven by widespread use in vaccines, targeted drug delivery, and advanced therapeutics. Their biocompatibility, stability, and ability to deliver nucleic acids enable precision medicine, improved efficacy, and integration with digital manufacturing and quality monitoring workflows.
- Dominant Region: North America leads with a 45.1% share in 2025, supported by advanced biotech infrastructure, strong regulatory frameworks, and high adoption of RNA therapeutics. Asia Pacific is the fastest-growing region, fueled by expanding healthcare and biotech facilities, government digital initiatives, rising R&D investments, and increased adoption of scalable lipid nanoparticle manufacturing solutions.
- Market Drivers: Growth is propelled by rising demand for mRNA vaccines and gene therapies, increasing investment in biopharmaceuticals, advances in nanoparticle formulation technologies, and the need for targeted, efficient drug delivery systems.
- Market Opportunity: Key opportunities include next-generation mRNA and siRNA therapeutics, oncology and rare disease applications, scalable manufacturing platforms, AI-enabled formulation optimization, integration with cell and gene therapy pipelines, and rapid market expansion in emerging biotech regions.
| Key Insights | Details |
|---|---|
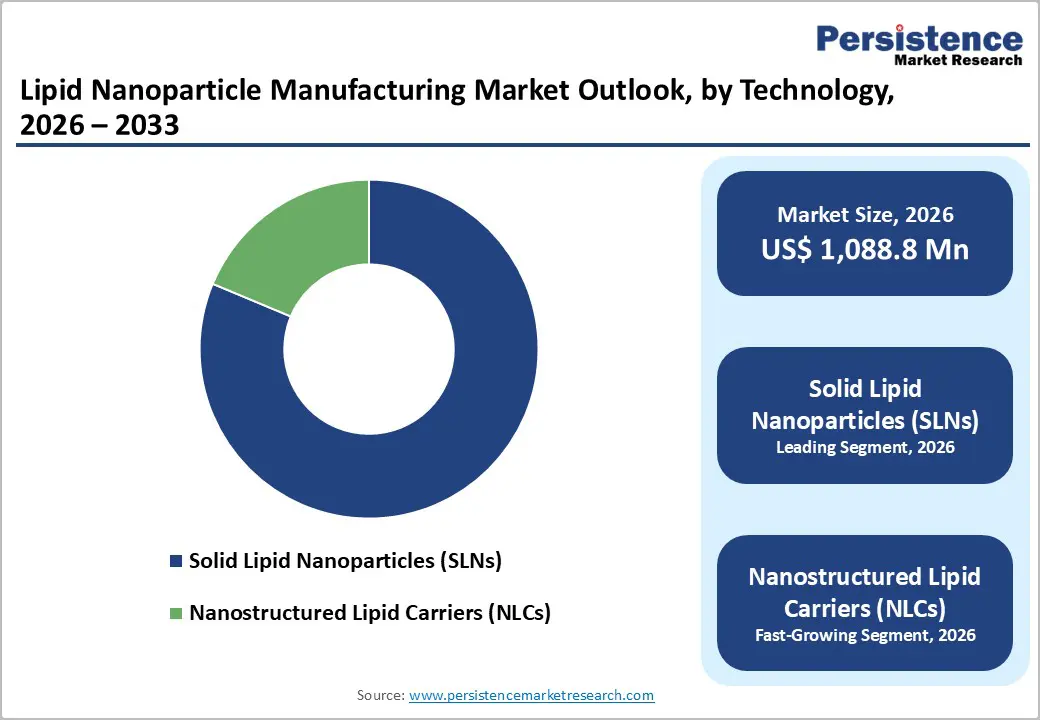
| Global Lipid Nanoparticle Manufacturing Market Size (2026E) | US$ 1,088.8 Mn |
| Market Value Forecast (2033F) | US$ 2,176.3 Mn |
| Projected Growth (CAGR 2026 to 2033) | 10.4% |
| Historical Market Growth (CAGR 2020 to 2025) | 9.4% |
Market Dynamics
Driver: Increasing Capabilities of LNPs in Viral Vaccine Delivery
The increasing capabilities of lipid nanoparticles (LNPs) in viral vaccine delivery represent a significant driver for their adoption and growth in the pharmaceutical industry. LNPs have demonstrated remarkable potential in enhancing the efficacy of viral vaccines, particularly highlighted during the COVID-19 pandemic with the successful development of mRNA vaccines.
LNPs serve as efficient carriers for delivering viral antigens or genetic material (such as mRNA) into cells, where they can induce robust immune responses. This capability is crucial in developing vaccines against rapidly mutating viruses or emerging infectious diseases, where traditional vaccine approaches may be less effective or time-consuming.
Furthermore, LNPs offer advantages such as improved stability of vaccine components, targeted delivery to immune cells, and reduced immune response variability compared to conventional adjuvants. These factors contribute to their role in accelerating vaccine development timelines and enhancing vaccine effectiveness. As research continues to advance in viral immunology and LNP technology, the capabilities of LNPs in viral vaccine delivery are expected to expand, driving innovation and shaping the future of preventative medicine against infectious diseases.
Restraints: Scale-up Challenges in LNP Manufacturing Limiting Wide-scale Adoption
LNPs, while promising for drug delivery due to their ability to encapsulate and deliver therapeutic payloads effectively, face hurdles in achieving consistent quality and scalability. One major challenge is the complexity of LNP formulation and manufacturing processes. LNPs require precise control over lipid composition, particle size distribution, and payload encapsulation efficiency to ensure therapeutic efficacy and safety.
Achieving these parameters consistently at large scales is difficult and often leads to batch-to-batch variability, which can affect product quality and regulatory approval timelines. Moreover, scaling up LNP production involves optimizing manufacturing processes to meet commercial demands without compromising product integrity or increasing costs prohibitively.
Addressing these challenges requires investments in infrastructure, technology, expertise in lipid chemistry and nanoparticle engineering while necessitating ongoing research and development to streamline LNP manufacturing, improve scalability, and reduce production costs. Successful resolution of these issues is crucial for LNPs to realize their full potential in revolutionizing drug delivery across various therapeutic areas.
Opportunity: Increasing Biotech and Pharma Investment
The global biotechnology and pharmaceutical sectors are experiencing significant investment growth, fuelling R&D activities aimed at advancing LNP technologies, particularly in the realm of mRNA vaccines and therapeutics. Biotech and pharmaceutical companies are intensifying their focus on enhancing LNP formulation, manufacturing processes, and delivery mechanisms to meet the burgeoning demand for innovative healthcare solutions.
Moreover, the increased financial backing is enabling LNP manufacturers to expand their production capacities, streamline operations, and innovate in nanoparticle design. Collaborations between industry stakeholders, research institutions, and academic centers are becoming more prevalent, fostering a collaborative ecosystem that accelerates the pace of technological innovation and regulatory approvals.
Furthermore, the growing investment landscape is facilitating strategic partnerships and mergers, enhancing market consolidation and enabling market players to leverage combined expertise and resources for faster commercialization of LNPs across diverse therapeutic areas. As a result, the biotech and pharmaceutical investment boom represents a pivotal moment for LNPs, offering ample opportunities for growth, innovation, and market leadership.
Category-wise Analysis
By LNP Type Insights
Electrochemical biosensors occupies 55.3% share of the global market in 2025, because they combine biocompatibility, scalability, stability, and controlled drug release, making them ideal for pharmaceutical applications. SLNs enhance the bioavailability of poorly soluble drugs and protect labile compounds from degradation, which supports broader clinical use compared with other carriers such as liposomes or polymeric nanoparticles. Their production methods, such as high-pressure homogenization, are well-established and cost-effective, enabling industrial-scale manufacture with reproducible quality. Research literature highlights that SLNs improve therapeutic efficacy across multiple administration routes (oral, parenteral, pulmonary) and offer easier sterilization and long-term stability due to their solid lipid core matrix, driving widespread adoption in drug delivery systems. Government and academic sources note these advantages as key to their predominance in current lipid nanoparticle formulation platforms.
By Therapeutic Area Insights
Infectious Diseases dominate the lipid nanoparticle manufacturing market because lipid nanoparticle (LNP) technologies have become foundational to modern vaccine delivery, particularly for viral illnesses. LNP-encapsulated mRNA vaccines, such as those developed for COVID-19, demonstrated rapid scalability, strong immunogenicity, and high efficacy, establishing LNPs as the preferred platform for preventing infectious diseases. LNP systems protect mRNA from degradation and facilitate efficient cellular uptake, enabling robust immune responses against targeted pathogens—this capability underpins many global vaccination strategies. Public health data show persistent global infectious disease burdens, with diseases like measles still affecting millions despite vaccine efforts, and substantial gaps in immunization coverage remain (e.g., 14.3 million children missed basic vaccines in 2024). This ongoing burden reinforces demand for effective infectious disease vaccines and thus drives LNP manufacturing growth.
Regional Insights
North America Lipid Nanoparticle Manufacturing Market Trends
North America dominates the lipid nanoparticle manufacturing market with 45.1% share in 2025, because it combines advanced biopharmaceutical infrastructure, substantial public research funding, and strong regulatory support. The region holds significant share, underpinned by extensive research ecosystems and manufacturing capacity centered in the United States and Canada. Robust NIH and federal nanoscale science investments support foundational nanotechnology and drug delivery research, reinforcing innovation capacity. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) have authorized multiple mRNA vaccines that rely on lipid nanoparticle delivery, showcasing scalable manufacturing and quality standards. High clinical trial activity and collaboration between biotech firms and research institutions further solidify North America’s leadership in LNP development and production.
Europe Lipid Nanoparticle Manufacturing Market Trends
Europe is important in the lipid nanoparticle manufacturing market because it remains a leading centre for pharmaceutical and biotech production, research, and regulation. The EU pharmaceutical industry contributes roughly €260 billion annually and employs about 700,000 people, generating significant economic impact and high-value jobs. Its biotech sector adds nearly €40 billion to the economy, with health biotechnology accounting for over 80 per cent of that value and 41 per cent of total pharmaceutical spending directed toward biologics, which often use lipid nanoparticle delivery systems. Europe also produces a substantial share of global biotech scientific publications (about 21 per cent) and conducts one-quarter of worldwide clinical trials. These factors underscore Europe’s capacity for advanced therapeutic development and manufacturing infrastructure, supporting leadership in LNP-based therapies.
Asia Pacific Lipid Nanoparticle Manufacturing Market Trends
Asia Pacific is the fastest-growing region in the lipid nanoparticle manufacturing market because its biotech and pharmaceutical ecosystem is rapidly expanding, supported by strong government R&D investments, regulatory reforms, and large patient pools that accelerate development. Asia’s share of the global innovative drug pipeline climbed sharply from about 28 % to 43 % in recent years, outpacing the United States and Europe in clinical assets and novel therapies.China’s R&D growth rate (~8.7 percent) significantly exceeds that of the US (~1.7 percent), narrowing the gap in research spending. Asia Pacific also hosts tens of thousands of clinical trials across key therapeutic areas, reflecting scalable research capacity. These factors create attractive conditions for lipid nanoparticle manufacturing growth compared with slower innovation increases in the US.

Competitive Landscape
Leading lipid nanoparticle manufacturing market companies focus on advanced formulation technologies, scalable production, and regulatory compliance. Investments target process optimization, AI-enabled design, and high-throughput screening to improve yield, stability, and reproducibility. Collaborations with biotech firms, academic institutions, and regulatory agencies accelerate development of mRNA, siRNA, and gene therapies. Emphasis on quality control, GMP standards, and global supply chain integration drives growth, enabling rapid adoption of LNP-based therapeutics, supporting vaccine production, targeted drug delivery, and precision medicine worldwide.
Key Industry Developments:
- In March 2025, Croda’s Avanti Research expanded its ionizable lipid offerings for lipid nanoparticle (LNP) drug development through a collaboration with Certest. The partnership aimed to provide enhanced lipid formulations to improve delivery efficiency, stability, and therapeutic performance of mRNA and gene therapies.
- In October 2024, Curapath and Certest joined forces to offer next-generation Lipid Nanoparticle (LNP) formulations incorporating proprietary excipients. The collaboration aimed to enhance LNP stability, delivery efficiency, and therapeutic performance for mRNA and gene therapies.
Companies Covered in Lipid Nanoparticle Manufacturing Market
- Certest Biotec
- Evonik
- Ascendia Pharmaceuticals
- Lonza
- Exelead (Merck KGaA)
- CordenPharma
- Curia Global, Inc.
- Creative Biostructure
- CD Bioparticles
- BOC Sciences
- Acuitas Therapeutics
- ABP Biosciences, LLC
- Recipharm AB
- Others
Frequently Asked Questions
The global lipid nanoparticle manufacturing market is projected to be valued at US$ 1,088.8 Mn in 2026.
Rising mRNA and gene therapy demand, vaccine development, targeted drug delivery, biotech investments, and scalable, stable lipid nanoparticle production drive growth.
The global lipid nanoparticle manufacturing market is poised to witness a CAGR of 10.4% between 2026 and 2033.
Opportunities include mRNA and siRNA therapeutics, oncology applications, rare disease treatments, scalable manufacturing, AI-enabled formulation, and emerging biotech regions.
Certest Biotec, Evonik, Ascendia Pharmaceuticals, Lonza, Exelead (Merck KGaA), CordenPharma.








