- Executive Summary
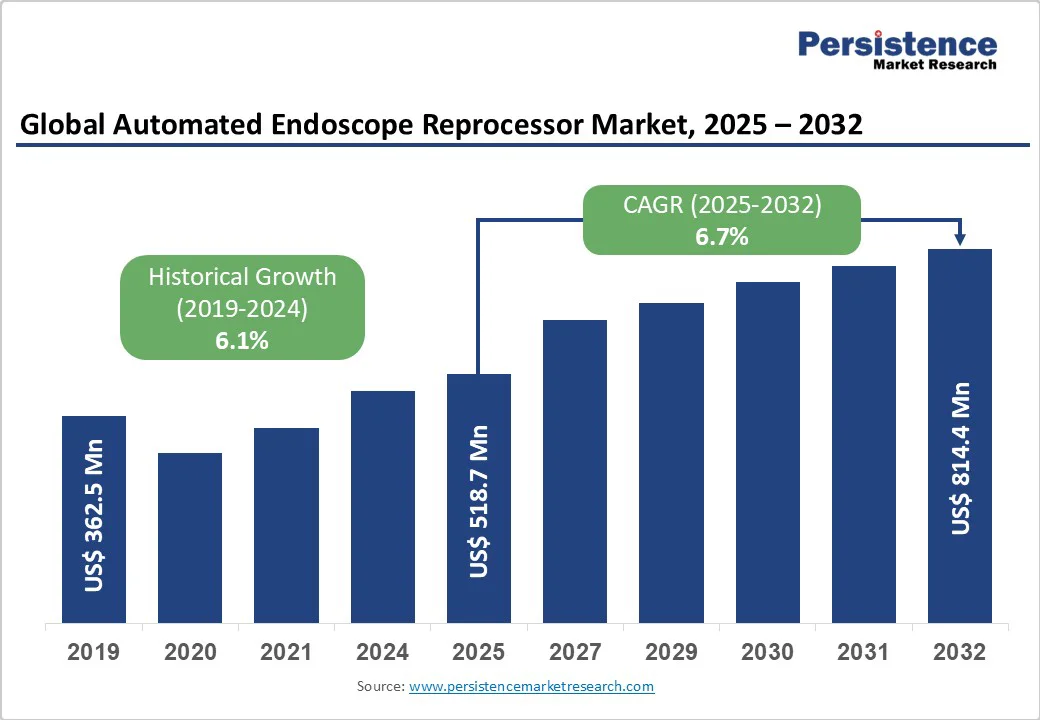
- Global Automated Endoscope Reprocessor Market Snapshot, 2025 and 2032
- Market Opportunity Assessment, 2025 - 2032, US$ Mn
- Key Market Trends
- Future Market Projections
- Premium Market Insights
- Industry Developments and Key Market Events
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Key Trends
- Macro-Economic Factors
- Global Sectorial Outlook
- Global GDP Growth Outlook
- COVID-19 Impact Analysis
- Forecast Factors - Relevance and Impact
- Value Added Insights
- Product Adoption Analysis
- Regulatory Landscape
- Value Chain Analysis
- Key Deals and Mergers
- PESTLE Analysis
- Porter’s Five Force Analysis
- Price Analysis, 2024A
- Key Highlights
- Key Factors Impacting Product Prices
- Pricing Analysis, By Product
- Regional Prices and Product Preferences
- Global Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Market Volume (Units) Projections
- Market Size (US$ Mn) and Y-o-Y Growth
- Absolute $ Opportunity
- Market Size (US$ Mn) Analysis and Forecast
- Historical Market Size (US$ Mn) Analysis, 2019-2024
- Market Size (US$ Mn) Analysis and Forecast, 2025-2032
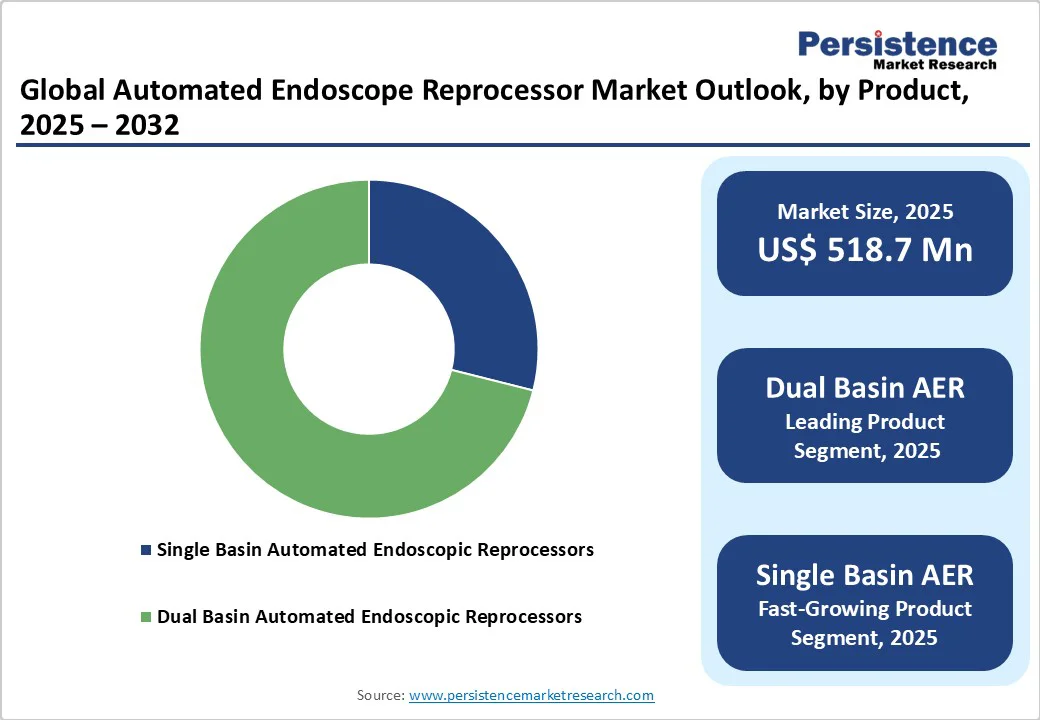
- Global Automated Endoscope Reprocessor Market Outlook: Product
- Introduction / Key Findings
- Historical Market Size (US$ Mn) and Volume (Units) Analysis, By Product, 2019-2024
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Attractiveness Analysis: Product
- Global Automated Endoscope Reprocessor Market Outlook: Modality
- Introduction / Key Findings
- Historical Market Size (US$ Mn) Analysis, By Modality, 2019-2024
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Attractiveness Analysis: Modality
- Global Automated Endoscope Reprocessor Market Outlook: End User
- Introduction / Key Findings
- Historical Market Size (US$ Mn) Analysis, By End User, 2019-2024
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis: End User
- Key Highlights
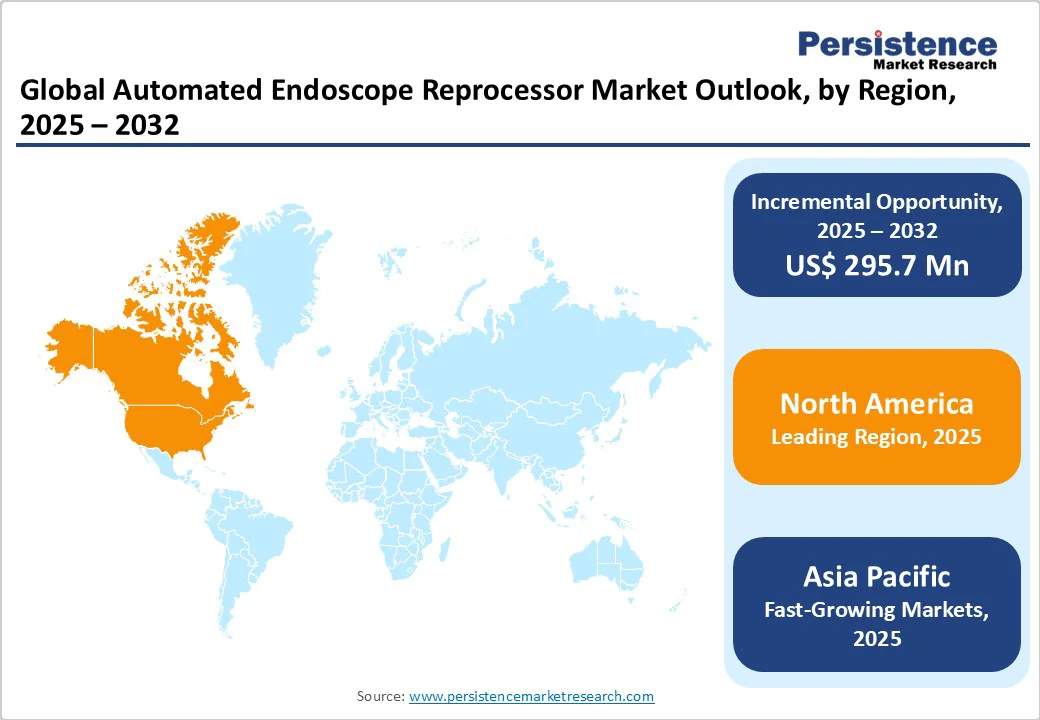
- Global Automated Endoscope Reprocessor Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Region, 2019-2024
- Market Size (US$ Mn) Analysis and Forecast, By Region, 2025-2032
- North America
- Europe
- East Asia
- South Asia and Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2019-2024
- By Country
- By Product
- By Modality
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2025-2032
- U.S.
- Canada
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis
- Europe Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2019-2024
- By Country
- By Product
- By Modality
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2025-2032
- Germany
- France
- U.K.
- Italy
- Spain
- Russia
- Türkiye
- Rest of Europe
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis
- East Asia Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2019-2024
- By Country
- By Product
- By Modality
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2025-2032
- China
- Japan
- South Korea
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis
- South Asia & Oceania Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2019-2024
- By Country
- By Product
- By Modality
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2025-2032
- India
- Southeast Asia
- ANZ
- Rest of South Asia & Oceania
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis
- Latin America Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2019-2024
- By Country
- By Product
- By Modality
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2025-2032
- Brazil
- Mexico
- Rest of Latin America
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis
- Middle East & Africa Automated Endoscope Reprocessor Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2019-2024
- By Country
- By Product
- By Modality
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2025-2032
- GCC Countries
- Egypt
- South Africa
- Northern Africa
- Rest of Middle East & Africa
- Market Size (US$ Mn) and Volume (Units) Analysis and Forecast, By Product, 2025-2032
- Single Basin Automated Endoscopic Reprocessors
- Dual Basin Automated Endoscopic Reprocessors
- Market Size (US$ Mn) Analysis and Forecast, By Modality, 2025-2032
- Stand-alone AER
- Table/Bench Top AER
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2025-2032
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Market Attractiveness Analysis
- Competition Landscape
- Market Share Analysis, 2024
- Market Structure
- Competition Intensity Mapping By Market
- Competition Dashboard
- Company Profiles (Details - Overview, Financials, Strategy, Recent Developments)
- Getinge
- Overview
- Segments and Product Types
- Key Financials
- Market Developments
- Market Strategy
- Steelco S.p.A.
- ASP India Private Ltd.
- STERIS
- Olympus America
- ECOLAB
- MMM Group
- SBSystem
- Belimed
- Cantel Medical Group of Companies
- Device Technologies Asia (Wassenburg Medical)
- HOYA Group (PENTAX Medical)
- OLIVE HEALTH CARE
- ARC Group of Companies Inc.
- Censis
- Hubei CFULL Medical Technology Co., Ltd.
- Medonica Co. LTD
- Detrox
- Huons Meditech
- MİXTA MEDİKAL
- SUN TECHNOLOGY JSC. (SUNTECH)
- JIAJING MEDICAL
- Nanosonics
- Getinge
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations

Loading page data
Please wait a moment