- Pharmaceuticals
- Multisystem Inflammatory Syndrome Market
Multisystem Inflammatory Syndrome Market Size, Share, and Growth Forecast, 2026 - 2033
Multisystem Inflammatory Syndrome Market by Diagnosis Type (Blood and urine test, Chest X-ray, CBC with differential, CRP, Others), Treatment (Antibiotics, Steroid Therapy, Intravenous Immunoglobulin (IVIG), Immunotherapy), and Regional Analysis for 2026 - 2033
Multisystem Inflammatory Syndrome Market Size and Trends Analysis
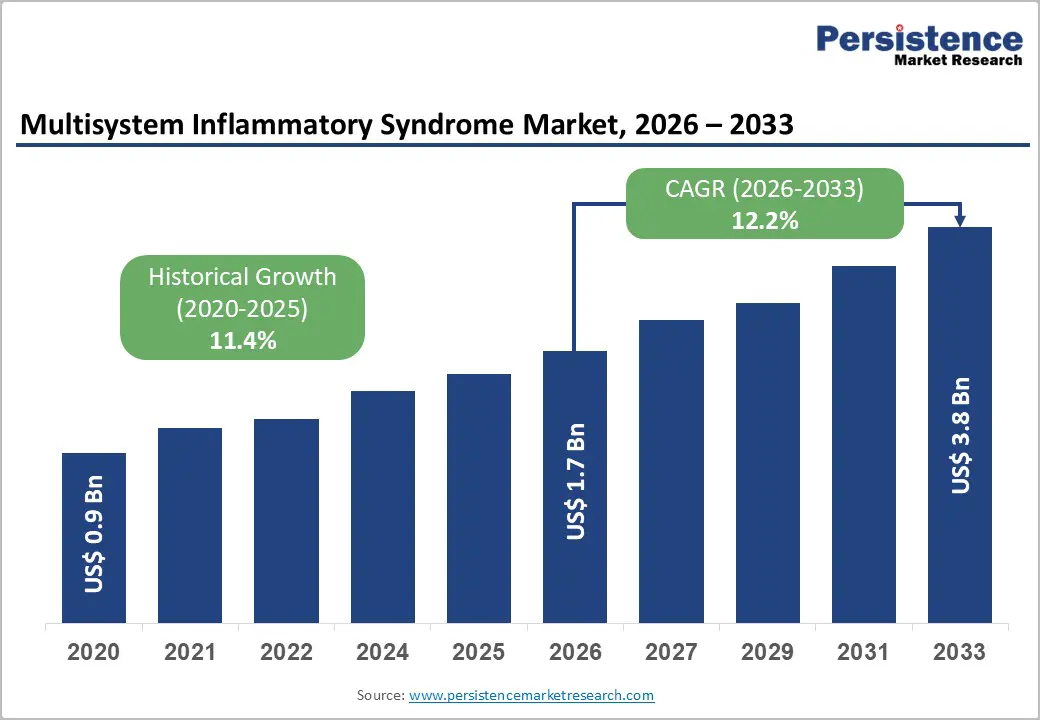
The global multisystem inflammatory syndrome market size is likely to be valued at US$1.7 billion in 2026 and is expected to reach US$3.8 billion by 2033, growing at a CAGR of 12.2% during the forecast period from 2026 to 2033, driven by increasing clinical recognition of hyper inflammatory conditions such as multisystem inflammatory syndrome in children (MIS-C) and multisystem inflammatory syndrome in adults (MIS-A).
These conditions are typically associated with post-infectious immune dysregulation, particularly following exposure to SARS-CoV-2, and are characterized by systemic inflammation affecting multiple organ systems, including cardiac, gastrointestinal, dermatologic, and neurological functions. Market growth is supported by advancements in early and accurate diagnostic approaches such as inflammatory biomarker profiling, cardiac imaging, and comprehensive laboratory testing, which enable prompt detection and risk stratification. Increased awareness among pediatricians, intensivists, and infectious disease specialists has significantly improved case identification and standardized management protocols.
Key Industry Highlights:
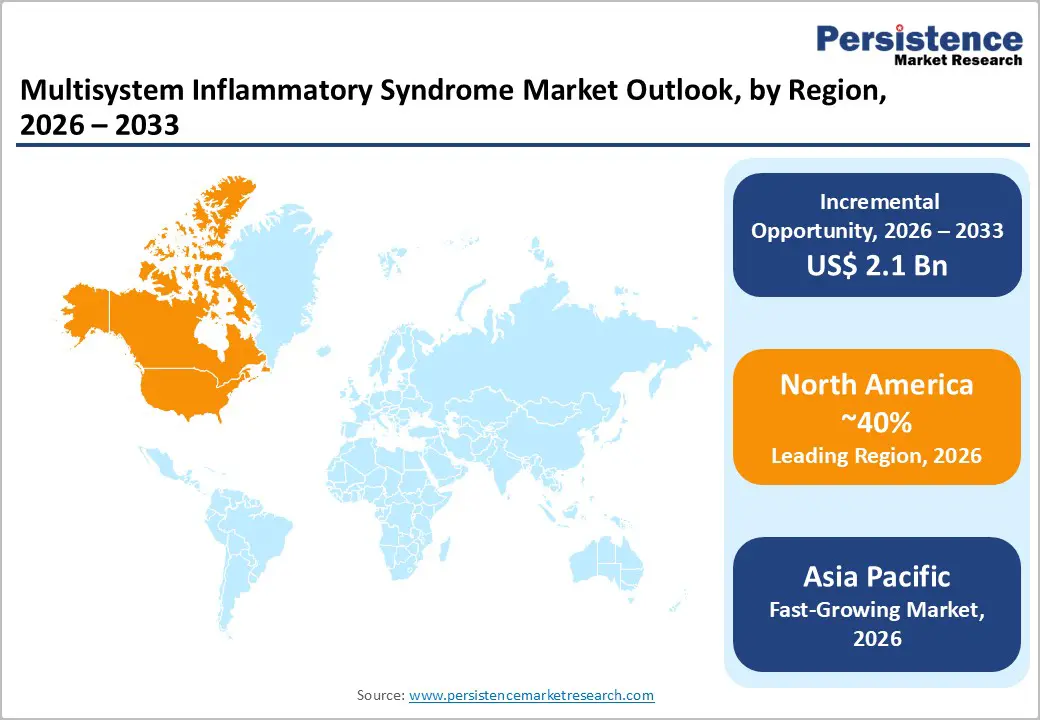
- Leading Region: North America, accounting for a market share of 40% in 2026, driven by strong surveillance systems, advanced diagnostics, supportive reimbursement policies, and established clinical treatment frameworks.
- Fastest-growing Region: Asia Pacific, supported by expanding healthcare infrastructure, rising disease awareness, and increasing regional manufacturing capabilities.
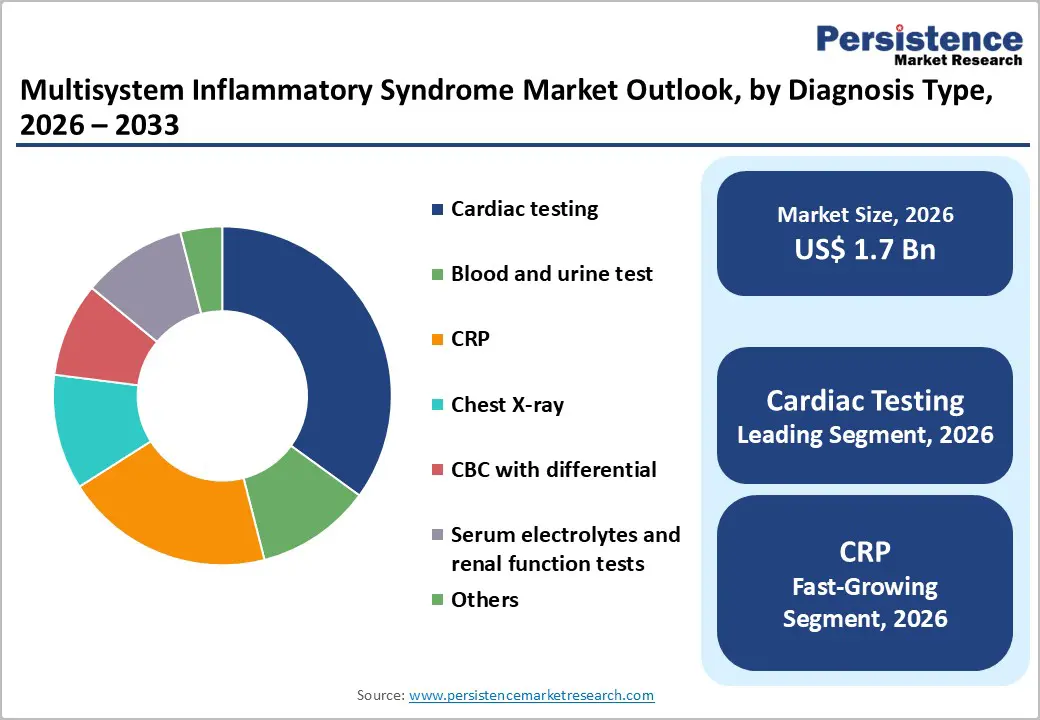
- Leading Diagnosis Type: Cardiac testing, accounting for 30% of the revenue share, driven by mandatory cardiac evaluation protocols and the high prevalence of cardiovascular complications in MIS cases.
- Leading Treatment Type: Intravenous immunoglobulin (IVIG), accounting for over 35% of the revenue share in 2026, supported by its established role as first-line therapy in clinical guidelines and strong efficacy in controlling immune-mediated inflammation in MIS patients.
| Key Insights | Details |
|---|---|
|
Multisystem Inflammatory Syndrome Market Size (2026E) |
US$1.7 Bn |
|
Market Value Forecast (2033F) |
US$3.8 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
12.2% |
|
Historical Market Growth (CAGR 2020 to 2025) |
11.4% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Growth Analysis - Increased Awareness and Diagnostic Advancements
Growing clinical awareness of multisystem inflammatory syndrome in children and multisystem inflammatory syndrome in adults has significantly strengthened the market landscape. Following the impact of SARS-CoV-2, healthcare systems enhanced surveillance programs and standardized screening protocols for hyperinflammatory syndromes. Pediatricians and critical care specialists are now more vigilant in recognizing early symptoms such as persistent fever, elevated inflammatory markers, and cardiac dysfunction. This heightened recognition has improved case detection rates and accelerated treatment initiation, directly increasing demand for diagnostic assays, cardiac imaging, and laboratory testing panels used in suspected MIS cases across hospital settings.
Advancements in diagnostic technologies reinforce this driver. Expanded use of biomarker testing, such as CRP, ferritin, D-dimer, troponin, and comprehensive metabolic panels, enables earlier and more accurate identification of systemic inflammation. Improvements in echocardiography and cardiac MRI enhance the assessment of myocardial involvement, a frequent complication in MIS patients. Integration of rapid laboratory turnaround systems and digital health records also supports timely clinical decision-making. These technological improvements reduce diagnostic delays, strengthen standardized care pathways, and expand the addressable patient pool.
Rising Adoption of Immunomodulatory Therapies
Multisystem inflammatory syndrome (MIS) is driven by immune dysregulation rather than direct viral injury; therefore, treatment approaches primarily aim to control and suppress systemic inflammation. First-line therapies such as intravenous immunoglobulin (IVIG) and corticosteroids have demonstrated strong clinical effectiveness in stabilizing patients and reducing complications. As consensus guidelines increasingly recommend early immunomodulation, hospitals are incorporating standardized treatment protocols, leading to consistent therapeutic demand and improved patient outcomes across pediatric and adult populations.
Beyond conventional therapies, the growing use of biologics and cytokine-targeted treatments strengthens this driver. Agents targeting interleukin pathways and other inflammatory mediators are being utilized in severe or refractory cases. Ongoing clinical trials and real-world evidence continue to validate these approaches, encouraging broader adoption. Improved access to specialty care centers and trained immunology teams enhances safe administration of advanced therapies.
Barrier Analysis - Limited Incidence and Case Rarity
A major constraint in the MIS market is the relatively low incidence of confirmed cases compared to other inflammatory or infectious diseases. MIS remains a rare post-infectious complication, limiting the overall patient pool eligible for diagnosis and treatment. Although awareness has improved, sporadic occurrence patterns reduce consistent demand for specialized therapies. Healthcare providers may encounter limited case volumes outside major tertiary centers, which can slow investment in dedicated diagnostic infrastructure or targeted treatment programs in smaller healthcare facilities.
Declining rates of widespread viral outbreaks may reduce MIS incidence over time. As vaccination coverage and infection control measures improve, the frequency of severe post-viral inflammatory syndromes may stabilize or decrease. This epidemiological variability introduces uncertainty in long-term revenue projections. Pharmaceutical and diagnostic companies must therefore balance research investment with unpredictable case volumes. The rarity of MIS, while clinically significant, inherently restricts large-scale commercialization opportunities compared to more prevalent immune-mediated disorders.
Diagnostic Complexity and Heterogeneity
The condition shares overlapping clinical features with sepsis, Kawasaki disease, toxic shock syndrome, and other inflammatory disorders, making differential diagnosis difficult. Absence of a single definitive diagnostic test requires clinicians to rely on a combination of laboratory findings, imaging results, and clinical presentation. This heterogeneity can delay confirmation and occasionally lead to misclassification, affecting timely therapeutic intervention and consistent reporting across healthcare systems.
Variability in symptom presentation between children and adults complicates clinical pathways. Some patients primarily exhibit gastrointestinal symptoms, while others show significant cardiac or neurological involvement. Differences in severity and organ system impact require multidisciplinary evaluation, increasing reliance on specialized expertise. In regions with limited advanced diagnostic facilities, accurate identification may be delayed. These diagnostic challenges create barriers to standardized care adoption and may restrain broader market penetration in developing healthcare environments.
Opportunity Analysis - Technological Convergence in Diagnostics
Integration of advanced biomarker profiling, artificial intelligence–supported imaging analysis, and digital health monitoring tools enhances early detection capabilities. Machine learning algorithms applied to electronic health records can identify risk patterns and flag suspected inflammatory syndromes earlier in the clinical course. Improved multiplex testing platforms allow simultaneous measurement of inflammatory markers, cardiac enzymes, and immune mediators, increasing efficiency and diagnostic accuracy.
Point-of-care testing and portable imaging technologies also expand access in regional and community healthcare settings. Telemedicine platforms enable consultation with specialists, improving diagnostic confidence in complex cases. As healthcare systems increasingly invest in precision diagnostics and data-driven decision-making, MIS detection becomes more streamlined and standardized. This convergence of digital health, laboratory innovation, and imaging technologies creates scalable opportunities for diagnostic manufacturers and strengthens the overall ecosystem supporting MIS management.
Personalized Immunotherapies
Personalized immunotherapy represents a transformative opportunity within the MIS market. As understanding of immune pathway dysregulation deepens, treatment strategies are gradually shifting from broad immunosuppression to targeted cytokine modulation. Identification of patient-specific inflammatory profiles enables clinicians to tailor therapy based on severity, biomarker levels, and organ involvement. This precision approach can improve treatment outcomes, reduce adverse effects, and optimize resource utilization in critical care environments.
Emerging biologics targeting specific interleukins and immune mediators demonstrate promise in refractory cases. Ongoing research into genetic susceptibility and immune response variability refines patient stratification models. Collaboration between biotechnology firms and academic research institutions is accelerating innovation in targeted therapies. As personalized medicine gains momentum across immunology and inflammatory disease management, its application in MIS could enhance therapeutic precision and create new avenues for high-value, specialized treatment development.
Category-wise Analysis
Diagnosis Type Insights
Cardiac testing is expected to lead the multisystem inflammatory syndrome market, accounting for approximately 30% of revenue in 2026, driven by the high prevalence of cardiovascular complications in both pediatric and adult cases. MIS frequently presents with myocardial dysfunction, coronary artery abnormalities, arrhythmias, and reduced ventricular function, making cardiac evaluation essential in suspected cases. For example, echocardiography is widely used in tertiary hospitals to monitor coronary artery dilation in MIS-C patients, supporting timely therapeutic decisions.
C-reactive protein (CRP) testing is likely to be the fastest-growing segment, supported by the expansion of its clinical utility in early screening and disease monitoring. CRP serves as a sensitive inflammatory biomarker, allowing physicians to rapidly assess systemic inflammation associated with MIS. Its widespread availability, rapid turnaround time, and cost-effectiveness make it accessible across hospitals, clinics, and diagnostic centers. For example, elevated CRP levels often trigger cardiac and immunological investigations in suspected MIS patients, accelerating diagnosis and intervention.
Treatment Type Insights
Intravenous immunoglobulin (IVIG) is projected to lead the market, capturing around 35% of the revenue share in 2026, supported by its established position as first-line therapy in MIS management. IVIG works by modulating immune system activity and reducing systemic inflammation, addressing the underlying immune dysregulation characteristic of the condition. Consensus treatment guidelines widely recommend IVIG as an initial intervention, either alone or in combination with corticosteroids, particularly in moderate to severe cases. For example, IVIG is routinely administered in pediatric intensive care units to manage MIS-C patients presenting with coronary involvement.
Steroid therapy is projected to be the fastest-growing treatment segment, supported by its increasingly prominent role in both first-line and adjunctive management approaches. Corticosteroids help suppress excessive immune activation and reduce inflammatory cytokine production, making them highly effective in severe or refractory MIS cases. Growing clinical evidence supports early combined use of steroids with IVIG to enhance recovery and reduce intensive care requirements. For example, methylprednisolone is frequently used in hospitalized MIS patients exhibiting persistent inflammation despite initial therapy.
Regional Insights
North America Multisystem Inflammatory Syndrome Market Trends
North America is anticipated to be the leading region, accounting for a market share of 38% in 2026, driven by strong healthcare infrastructure, advanced clinical practice, and extensive disease surveillance capabilities. The region benefits from well-established diagnostic networks that support early identification of MIS cases through widespread use of cardiac imaging, inflammatory biomarkers, and comprehensive laboratory testing. An example of corporate involvement in advancing care is Pfizer, which has collaborated on research initiatives exploring immunological responses and therapeutic targets following viral infections.
Another notable trend in North America is the growing focus on personalized and targeted treatment strategies for MIS. As clinicians recognize the heterogeneity of inflammatory responses among patients, there is increasing interest in tailored therapeutic regimens that go beyond traditional first-line treatments such as intravenous immunoglobulin (IVIG) and corticosteroids. Biologic therapies that target specific cytokine pathways are gaining traction in severe or refractory cases, with providers exploring combination approaches that optimize efficacy while minimizing adverse effects.
Europe Multisystem Inflammatory Syndrome Market Trends
Europe is likely to be a significant market for multisystem inflammatory syndrome in 2026, due to increasing clinical awareness and coordinated regional responses to post-infectious inflammatory conditions, particularly following the COVID-19 pandemic. Healthcare systems across the U.K., Germany, France, Spain, and other EU countries have adopted standardized protocols for early diagnosis and management of both pediatric and adult cases. National public health agencies and specialist societies emphasize rapid identification through inflammatory biomarkers, cardiac imaging, and comprehensive clinical assessment, improving overall care pathways.
Therapeutic innovation is another key trend shaping the European MIS market as treatment strategies evolve beyond conventional approaches. There is growing interest in advanced immunomodulatory therapies, including biologics and cytokine-targeted agents that address specific inflammatory pathways. For example, Roche/Genentech has been involved in clinical and real-world evaluation of IL-6 inhibitors that may benefit patients with severe inflammatory responses, contributing to expanded therapeutic options within the region.
Asia Pacific Multisystem Inflammatory Syndrome Market Trends
The Asia Pacific region is likely to be the fastest growing region, driven by countries enhance disease awareness, diagnostic capabilities, and pediatric care infrastructure. Nations such as China, Japan, India, Australia, and ASEAN members are strengthening clinical surveillance systems, enabling earlier identification and management of MIS cases. Regional healthcare initiatives increasingly emphasize the importance of inflammatory biomarker testing, cardiac evaluation, and comprehensive laboratory panels to support accurate diagnosis.
With rising demand for IVIG, corticosteroids, and targeted immunotherapies, regional pharmaceutical companies, along with multinational partners, are increasing their focus on localized supply chains and expanded access programs. For example, Takeda Pharmaceutical has been instrumental in scaling up plasma-derived immunoglobulin products in the Asia Pacific region, strengthening the availability of essential therapeutics used in first-line MIS treatment. Training programs and clinical workshops aimed at enhancing physician expertise in managing post-infectious hyper-inflammatory syndromes support long-term market sustainability.
Competitive Landscape
The global multisystem inflammatory syndrome market exhibits a moderately fragmented structure, driven by a mix of large pharmaceutical and biotechnology companies, regional diagnostics firms, and specialized healthcare providers focused on post-infectious inflammatory conditions. Continued clinical demand for rapid and accurate diagnostics, combined with expanding immunomodulatory treatment protocols, has attracted investment from diverse industry participants. Players are increasingly advancing cardiac testing technologies, inflammatory marker assays, and immunotherapy solutions to address the complex needs of MIS presentations in both pediatric and adult populations.
With key leaders including Pfizer-BioNTech, Novartis AG, Roche/Genentech, Takeda Pharmaceutical, Merck & Co., Inc., and AbbVie Inc., the competitive landscape reflects a balance between established multinational firms and emerging specialized innovators. These players compete through product portfolio expansion, strategic collaborations with clinical research organizations, and investment in advanced diagnostic platforms that enable earlier detection and improved patient stratification.
Key Industry Developments:
- In July 2025, a Phase 2a clinical trial reported that larazotide, an oral drug originally developed for celiac disease, was safe and showed potential benefit in treating children with Multisystem Inflammatory Syndrome in Children (MIS-C). According to findings published in Science Translational Medicine, children with MIS-C who received larazotide alongside standard care experienced faster resolution of gastrointestinal symptoms, quicker clearance of SARS-CoV-2 spike protein antigen from the bloodstream, and earlier return to usual activities compared with placebo recipients, with no larazotide-related safety concerns reported.
Companies Covered in Multisystem Inflammatory Syndrome Market
- Mesoblast Limited
- Alliance for Cell Therapy Now
- Sanford Health
- IBA Life sciences
- Pfizer-BioNTech
- Novartis AG
- GlaxoSmithKline plc (GSK)
- Merck & Co., Inc.
- Eli Lilly and Company
- AbbVie Inc.
- Sanofi S.A.
Frequently Asked Questions
The global multisystem inflammatory syndrome market is projected to reach US$1.7 billion in 2026.
Rising awareness and early diagnosis of post-viral hyper inflammatory conditions, along with increasing adoption of advanced immunomodulatory therapies and standardized treatment protocols.
The multisystem inflammatory syndrome market is expected to grow at a CAGR of 12.2% from 2026 to 2033.
Key market opportunities lie in the development of targeted immunotherapies, advanced diagnostic technologies, and personalized treatment approaches for managing severe immune-mediated inflammatory responses.
Mesoblast Limited, Alliance for Cell Therapy Now, Sanford Health, and IBA Life Sciences are the leading players.







