- Biotechnology
- Metabolomics Biomarker Market
Metabolomics Biomarker Market Size, Share, and Growth Forecast, 2026 - 2033
Metabolomics biomarker market by Indication (Cardiovascular, Others), Application (Drug Designing, Others), End-user (Hospitals, Research Laboratories, Cancer Research Centers, Diagnostic Laboratories), and Regional Analysis for 2026 – 2033
Metabolomics Biomarker Market Size and Trends Analysis
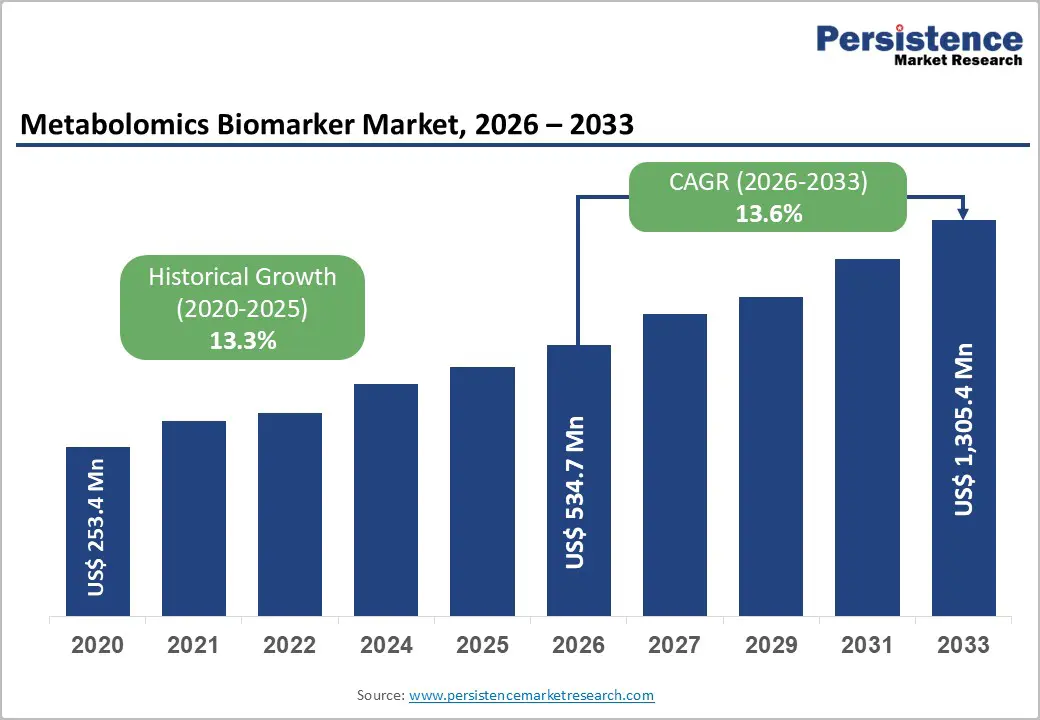
The global metabolomics biomarker market size is likely to be valued at US$534.7 million in 2026, and is expected to reach US$1,305.4 million by 2033, growing at a CAGR of 13.6% during the forecast period from 2026 to 2033, driven by the increasing prevalence of chronic diseases, rising demand for precision medicine, and growing adoption of metabolomics in early disease detection and drug response monitoring.
Rising demand for high-sensitivity metabolomics biomarkers, particularly in cancer and metabolic diseases, is driving adoption in diagnostics and research. Advances in mass spectrometry, AI analysis, and multi-omics integration enhance accuracy and therapeutic guidance, while their role in non-invasive disease stratification and personalized treatment fuels market growth.
Key Industry Highlights:
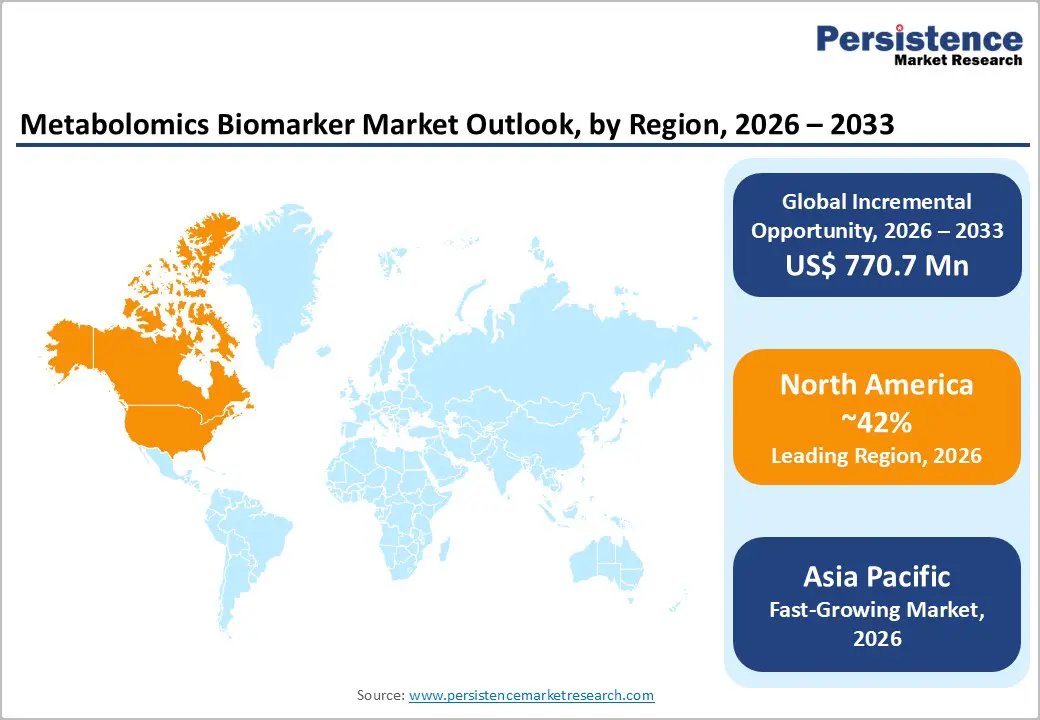
- Leading Region: North America, anticipated to account for a 42% market share in 2026, driven by strong research funding, advanced omics infrastructure, and high adoption of precision medicine in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by rising chronic disease burden, expanding clinical research, and growing investments in metabolomics platforms in China and India.
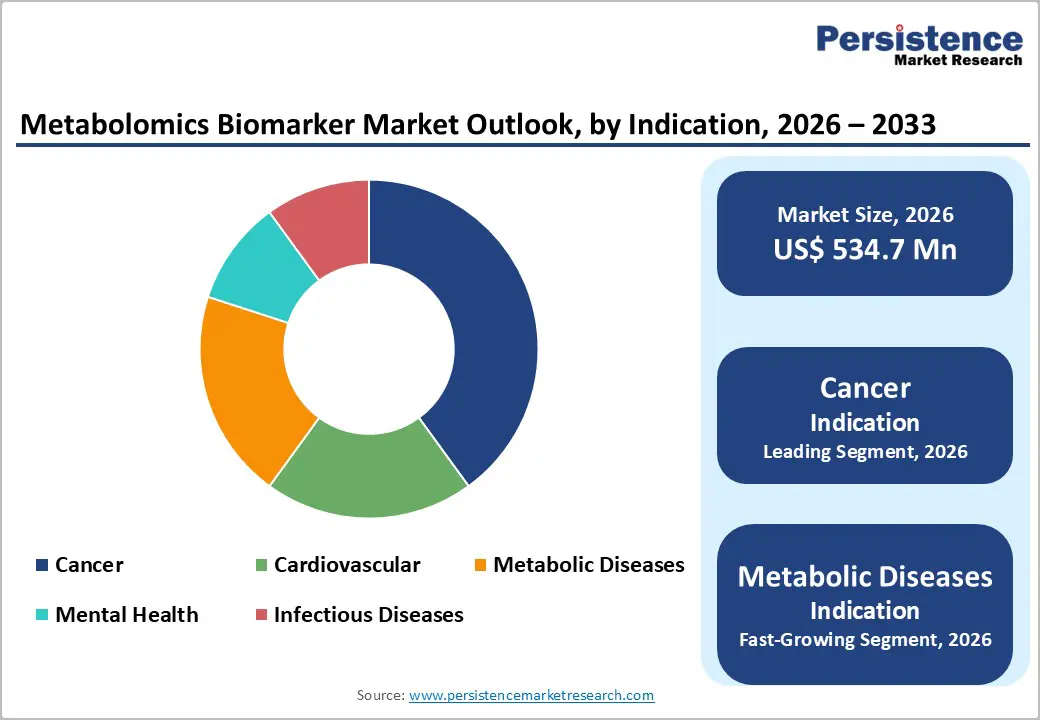
- Dominant Indication: Cancer, which accounts for approximately 38% of the market share, as it remains the primary focus of biomarker discovery and companion diagnostics.
- Leading Application: Personalized medicine, to contribute nearly 52% of the market revenue, due to the highest clinical translation potential.
| Global Market Attributes | Key Insights |
|---|---|
| Metabolomics Biomarker Market Size (2026E) | US$534.7 Mn |
| Market Value Forecast (2033F) | US$1,305.4 Mn |
| Projected Growth CAGR (2026-2033) | 13.6% |
| Historical Market Growth (2020-2025) | 13.3% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Growth Analysis – Increasing Chronic Disease Burden and Precision Medicine Adoption
Chronic, non-communicable diseases (NCDs) represent a vast and escalating health challenge globally, with world-leading health authorities reporting that they accounted for about 43 million deaths in 2021, roughly 75% of all non-pandemic-related deaths worldwide. Cardiovascular diseases, cancers, chronic respiratory conditions, and diabetes together comprise most of these fatalities, including 18 million premature deaths before age 70 in the same year. These trends place immense pressure on health systems and amplify demand for more sophisticated diagnostic and treatment tools.
Government-led precision medicine initiatives illustrate how healthcare systems are adopting individualized approaches to address this burden. The U.S. National Institutes of Health’s All of Us Research Program has enrolled over 633,000 participants, building one of the largest diverse health and genomic data resources to inform tailored prevention and treatment strategies for chronic diseases. This type of initiative, supported by federal research funding, enables the discovery of novel biomarkers and disease mechanisms that are critical for targeted therapies and improved outcomes. Metabolomics biomarkers fit into this landscape by offering molecular insights into disease states and individual biological variation, thereby supporting the development and clinical adoption of precision medicine strategies amid a growing chronic disease burden.
Expanding Clinical Research and Multi-Omics Integration
Clinical research activity tracked on the U.S. ClinicalTrials.gov registry demonstrates the scale of modern clinical studies: the database maintained by the National Library of Medicine includes records for over 500,000 clinical trials and observational studies and receives more than 113,000 daily visitors to access study information. This reflects the extensive infrastructure supporting clinical investigation across therapeutic areas, including studies that increasingly incorporate molecular profiling data. To support deeper biological insights, government research agencies are investing directly in multi-omics approaches that integrate metabolomics with genomics, transcriptomics, proteomics, and other molecular layers.
The U.S. National Institutes of Health has committed US$50.3 million over five years to a Multi-Omics for Health and Disease Consortium to advance clinical research strategies that leverage integrated omics data. This funding supports high-throughput generation of genomic, epigenomic, transcriptomic, proteomic, and metabolomic profiles from study participants to characterize health and disease states. Government-supported resources such as the NIH’s Metabolomics Workbench house data from over 2,000 metabolomics studies, including tens of thousands of samples and known metabolites, thereby facilitating the integration of metabolite data with other biological datasets for clinical and translational research.
Barrier Analysis – High Cost of Advanced Metabolomics Platforms
Capital investment in advanced analytical systems, such as mass spectrometry and associated chromatographic platforms, constitutes a substantial barrier to the adoption of metabolomics biomarker workflows in research and clinical settings. Government funding programs acknowledge that state-of-the-art scientific instruments often have base costs exceeding $25,000, necessitating shared awards or consortium-level funding to enable acquisition by academic and biomedical institutions. For example, the NIH Shared Instrumentation Grant (SIG) Program is designed to support the purchase or upgrade of high-cost, specialized instruments, with awards ranging from US$50,000 to US$750,000 to make such systems accessible to NIH-supported research groups.
Per-sample analysis fees at government-supported facilities further illustrate the ongoing operational costs of advanced metabolomics workflows. NIH metabolomics cores charge up to US$175 per sample for untargeted metabolomics analysis due to the complexity of instrumentation and data processing involved. These figures reflect that both upfront capital outlay and recurring analytical expenses are high relative to many other laboratory technologies, limiting adoption by smaller labs and institutions that cannot justify or sustain such expenditures without significant external funding.
Data Standardization and Regulatory Complexity
Standardization of metabolomics data remains a core technical barrier recognized by government research programs. NIH-funded initiatives, such as the Common Fund’s Metabolomics Program, explicitly aim to address data harmonization and quality control, underscoring the need for consensus on best practices in sample handling, study design, platform selection, and data annotation to enable reproducible use. Without widely adopted standards and interoperable data formats, datasets generated across different laboratories often cannot be directly compared or pooled, complicating the evidence base needed for regulatory review and clinical validation.
Regulatory processes for qualifying biomarkers are structured and multi-step, requiring extensive evidence of analytical and clinical utility before a biomarker can be accepted for use in drug development or diagnostics. The FDA’s Biomarker Qualification Program details formal stages of submission and review, reflecting inherent complexity in meeting regulatory standards for metabolomics markers. The lack of approved tests, challenges in data harmonization, and the absence of structured regulatory review pathways are concrete barriers that slow the standardization and clinical translation of metabolomics biomarkers.
Opportunity Analysis – Innovations in Cancer and Metabolic Disease Companion Diagnostics
Government cancer research data initiatives show how metabolomics is becoming integral to diagnostic and companion diagnostic discoveries. The U.S. National Cancer Institute’s Proteomic Data Commons (PDC) now supports mass spectrometry-based metabolomics and lipidomics datasets alongside proteomic and genomic data, enabling deeper metabolic characterization of tumors for different cancer types. This integrated resource includes multiple metabolome studies (e.g., ccRCC and GBM cohorts) that facilitate the identification of metabolic signatures linked to cancer biology and treatment response.
Public metabolomics repositories demonstrate scale and diversity of metabolic profiling efforts relevant for cancer and metabolic disease biomarkers. The National Metabolomics Data Repository (NMDR) encompasses over 3,000 publicly available metabolomics studies with tens of thousands of samples and thousands of known metabolites, covering disease states such as type 1 diabetes and cancer metabolic profiles. These datasets support translational research linking metabolic profiles to clinical outcomes and stratifying patients for targeted therapies.
Development in Personalized Medicine
The U.S. National Institutes of Health’s All of Us Research Program is a flagship precision medicine initiative gathering extensive health, genomics, and lifestyle data to fuel individualized approaches to diagnosis and treatment. To date, the program has made data available from more than 633,000 participants, offering one of the largest diverse biomedical datasets in existence for research into personalized health strategies. Results from this cohort include a growing collection of whole genome sequences, and expanded genomic releases include over 414,000 whole genome sequences for researchers to explore genetic variation and its links to disease risk and treatment responses.
Complementing cohort-scale collection efforts, the NIH’s Trans-Omics for Precision Medicine (TOPMed) Program has aggregated large molecular datasets, providing over 180,000 whole-genome sequences with associated phenotypic and clinical information for heart, lung, blood, and sleep disorders. These extensive, federally supported data resources enable researchers to identify molecular patterns such as metabolite profiles tied to individual disease trajectories and therapeutic outcomes.
Category-wise Analysis
Indication Insights
Cancer is anticipated to dominate the market, accounting for 38% of the market share in 2026, fueled by the central role of metabolic reprogramming in tumor initiation, progression, and treatment response. Cancer cells exhibit distinct metabolic signatures linked to rapid proliferation, immune evasion, and drug resistance, making metabolomics highly valuable for biomarker discovery. Rising use of precision oncology, targeted therapies, and immunotherapies increases demand for biomarkers that can stratify patients and monitor treatment efficacy. Metabolomic Technologies Inc. is a biotechnology company based in Edmonton, Alberta, Canada. Its urine-based test, PolypDx™, uses metabolic signatures to detect adenomatous polyps, the precursors to colorectal cancer, with significantly higher sensitivity than some traditional fecal screening methods, based on clinical validation in a cohort of nearly 1,000 patients.
Metabolic diseases are expected to be the fastest-growing indication, driven by the rising global prevalence and increasing clinical focus on early detection and disease management. Metabolomics enables detailed profiling of small molecules, offering insights into altered metabolic pathways that underlie these conditions. Personalized intervention strategies, lifestyle monitoring, and targeted therapeutics rely on such molecular data to optimize outcomes. Metabolic disease biomarkers are Human Metabolome Technologies, Inc. (HMT). HMT’s advanced metabolomics platforms (CE-MS and LC-MS) can quantify over 100 polar metabolites involved in metabolic disorders from clinical samples such as blood and urine, supporting biomarker discovery and therapeutic monitoring in conditions including diabetes and other metabolic diseases.
Application Insights
Personalized medicine is expected to dominate the market, contributing nearly 52% of revenue in 2026, due to its focus on tailoring treatments to individual biological profiles. Metabolomics enables detailed analysis of a patient’s unique metabolic state, revealing biomarkers that guide therapy selection, monitor drug response, and predict adverse effects. Increasing integration of genomics, proteomics, and metabolomics in clinical practice supports targeted interventions for complex diseases such as cancer, cardiovascular disorders, and metabolic conditions. Metabolon, Inc., is a biotechnology company that uses advanced metabolomics profiling to support precision health approaches in clinical research and care. Metabolon’s platforms generate detailed metabolic signatures from biological samples that help identify biomarkers linked to disease states and individual responses to therapies, enabling clinicians and researchers to tailor treatment strategies based on a patient’s unique biochemical profile.
Drug designing is likely to be the fastest-growing application, driven by its ability to provide detailed insights into cellular metabolism and disease pathways. Metabolomics identifies specific metabolites and biochemical signatures that reveal potential drug targets, mechanisms of action, and off-target effects. This molecular-level understanding allows pharmaceutical companies to design more effective and safer drugs, optimize lead compounds, and predict patient response. Evotec SE provides high-resolution metabolomics profiling to pharmaceutical partners to understand disease-related metabolic changes, identify compound-induced metabolic pathway alterations, and support the selection and optimization of drug candidates across the research pipeline, from target identification through preclinical evaluation.
Regional Insights
North America Metabolomics Biomarker Market Trends
North America is projected to dominate, accounting for nearly 42% share in 2026, driven by the region’s strong NIH/NCI funding, advanced omics infrastructure, and high public awareness of precision medicine benefits. Diagnostic systems in the U.S. and Canada provide extensive support for metabolomics biomarker programs, ensuring wide accessibility across cancer, personalized medicine, and diagnostic laboratory populations. Increasing demand for high-sensitivity, convenient, and easy-to-interpret forms is further accelerating adoption, as these formats improve early detection and reduce barriers associated with conventional diagnostics.
Innovation in metabolomics biomarker technology, including stable multi-omics, improved AI interpretation delivery, and targeted cancer enhancement, is attracting significant investments from both public and private sectors. Government initiatives and FDA campaigns continue to promote use against disease risks, therapy selection concerns, and emerging companion diagnostic threats, creating sustained market demand. The growing focus on metabolic disease grades and specialty uses, particularly for personalized medicine and others, is expanding the target applications for metabolomics biomarkers.
Europe Metabolomics Biomarker Market Trends
Europe's growth is fueled by increasing awareness of precision medicine benefits, strong regulatory systems, and government-led biobanking and cohort programs. Countries such as Germany, the U.K, France, and the Netherlands have well-established research and diagnostic frameworks that support routine metabolomics biomarker use and encourage adoption of innovative biomarker delivery methods, including multi-metabolite panels. These high-accuracy formulations are particularly appealing for cancer populations, regulation-conscious laboratories, and personalized medicine users, improving outcomes and coverage rates.
Technological advancements in metabolomics biomarker development, such as enhanced LC-MS/MS, application-targeted delivery, and improved AI-grade platforms, are further boosting market potential. European authorities are increasingly supporting research and trials for biomarkers against both routine and specialized needs, strengthening market confidence. The growing emphasis on convenient, non-invasive options is aligned with the region’s focus on preventive disease management and reducing healthcare burden. Public awareness campaigns and promotion drives are expanding reach in both diagnostic and research segments, while suppliers are investing in validation and novel variants to increase efficacy.
Asia Pacific Metabolomics Biomarker Market Trends
Asia Pacific is likely to be the fastest-growing market for metabolomics biomarkers in 2026, driven by rising chronic disease awareness, increasing government initiatives, and expanding application programs across the region. Countries such as China, India, Japan, and South Korea are actively promoting biomarker campaigns to address cancer and metabolic disease growth and emerging precision needs. Metabolomics biomarkers are particularly attractive in these regions due to their scalable administration, ease of adoption, and suitability for large-scale diagnostic laboratory and hospital drives in both urban and rural populations.
Technological advancements are supporting the development of stable, effective, and easy-to-deploy metabolomics biomarkers, which can withstand challenging validation conditions and minimize cost dependence. These innovations are critical for reaching domestic laboratories and improving overall disease coverage. Growing demand for cancer, personalized medicine, and diagnostic laboratory applications is contributing to market expansion. Public-private partnerships, increased healthcare expenditure, and rising investment in biomarker research and validation capacity are further accelerating growth. The convenience of biomarker delivery, combined with improved accuracy and reduced risk of late diagnosis, positions it as a preferred choice.
Competitive Landscape
The global metabolomics biomarker market is highly competitive, featuring a mix of established life-science instrument leaders and emerging biomarker discovery specialists. In North America and Europe, Agilent Technologies, Inc. and Thermo Fisher Scientific, Inc. dominate through robust R&D capabilities, extensive distribution networks, and strong ties with diagnostic laboratories, supported by advanced mass-spectrometry and multi-omics programs. Companies such as Chenomx Inc., Creative Proteomics, Biocrates Life Sciences AG, and Metabolon, Inc. focus on specialized metabolomics solutions for biomarker discovery, enhancing analytical depth and translational research applications. Bruker Corporation and Metabolomic Diagnostics Ltd. provide integrated platforms for high-resolution metabolite profiling, supporting clinical and research workflows.
In the Asia Pacific region, local players leverage cost-competitive solutions to improve accessibility and adoption. Strategic partnerships, collaborations, and acquisitions across these players accelerate portfolio expansion, technological innovation, and commercialization. Cancer-focused metabolomics applications enhance early detection, reduce late-stage risks, and personalized-medicine formulations improve patient stratification, driving penetration in precision oncology and chronic disease research.
Key Industry Developments:
- In June 2025, Thermo Fisher Scientific Inc. (NYSE: TMO), a global leader in scientific solutions, launched the Thermo Scientific™ Orbitrap™ Astral™ Zoom and Orbitrap™ Excedion™ Pro mass spectrometers. These instruments provided unmatched analytical performance and speed, enabling breakthroughs in biological processes. They supported advancements in precision medicine and offered insights into complex diseases such as Alzheimer’s and cancer. Integrated into Thermo Fisher’s comprehensive portfolio, these innovations bolstered success in proteomics, biopharmaceutical development, and scientific research.
- In May 2025, Thermo Fisher launched the MxP® Quant 1000 kit to address challenges in complex chronic diseases through metabolomics. The kit’s metabolites and pathways were meticulously selected, and its practical applications were demonstrated. Researchers leveraged the kit to generate actionable metabolomic data, supporting disease profiling, biomarker discovery, and targeted studies.
Companies Covered in Metabolomics Biomarker Market
- Agilent Technologies, Inc.
- Chenomx Inc.
- Creative Proteomics
- Thermo Fisher Scientific, Inc.
- Biocrates Life Sciences Ag
- Metabolon, Inc.
- Bruker Corporation
- Metabolomic Diagnostics Ltd.
- Biomark Diagnostics Inc.
- Human Metabolome Technologies America, Inc.
- Merck KGaA
Frequently Asked Questions
The global metabolomics biomarker market is projected to reach US$534.7 million in 2026.
The increasing incidence of cancer, diabetes, cardiovascular, and metabolic disorders drives demand for metabolomics biomarkers to enable early detection, monitoring, and personalized treatment strategies.
The metabolomics biomarker market is poised to witness a CAGR of 13.6% from 2026 to 2033.
Rising prevalence of metabolic disorders and the use of metabolomics for drug discovery, mechanism elucidation, and early disease detection create opportunities for new biomarker panels, diagnostic platforms, and companion diagnostics across chronic and complex diseases.
Metabolon, Inc., Thermo Fisher Scientific, Inc., Agilent Technologies, Inc., Biocrates Life Sciences AG, and Bruker Corporation are the key players.








