
- Pharmaceuticals
- Ferritin Testing Market
Ferritin Testing Market Size, Share, and Growth Forecast 2026 - 2033
Ferritin Testing Market by Product (Reagents, Test Kits, Instruments), by Technology (Chemiluminescence Immunoassay, ELISA, Fluorescence Immunoassay, Rapid / Point-of-Care Tests), by Application (Iron Deficiency Anemia, Iron Overload Disorders, Pregnancy & Maternal Screening, Inflammatory & Chronic Diseases), by Regional Analysis, 2026 - 2033
Ferritin Testing Market Share and Trends Analysis
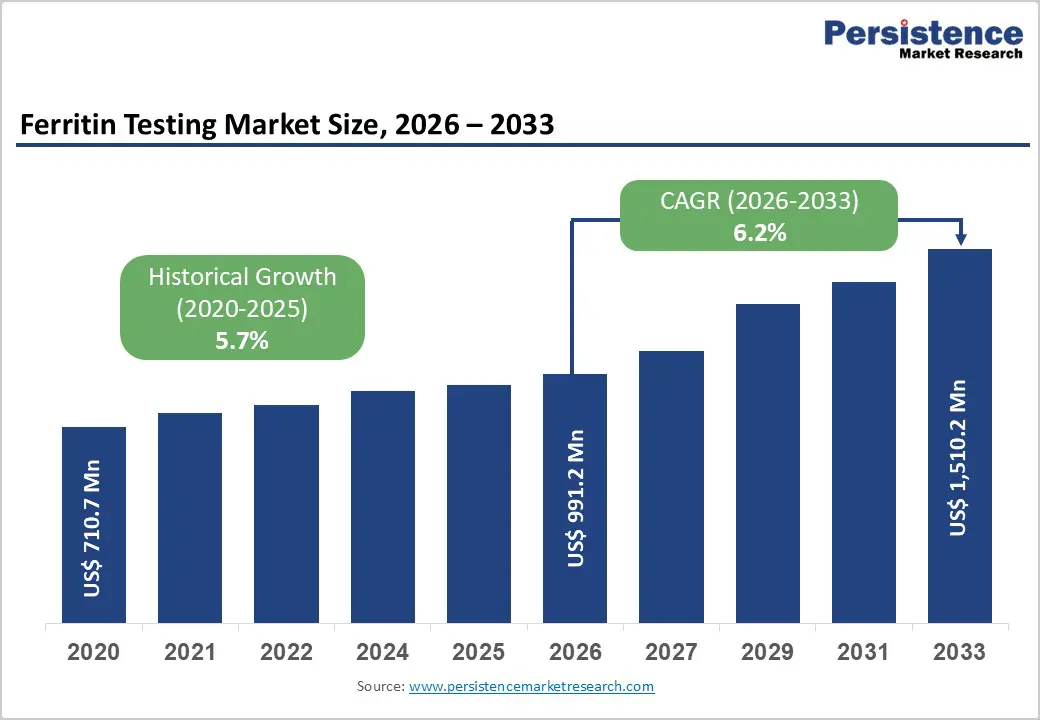
The global ferritin testing market size is expected to be valued at US$ 3.7 million in 2026 and projected to reach US$ 5.6 million by 2033, growing at a CAGR of 6.2% between 2026 and 2033.
This growth is driven primarily by the high and persistent global burden of iron deficiency anemia and the increasing recognition of ferritin as the preferred biomarker for assessing body iron stores. The market further benefits from the rising prevalence of chronic kidney disease-related anemia, expansion of maternal health and antenatal screening programs, and technological advances in high-sensitivity immunoassays and point-of-care platforms that improve diagnostic access and clinical decision-making.
Key Industry Highlights:
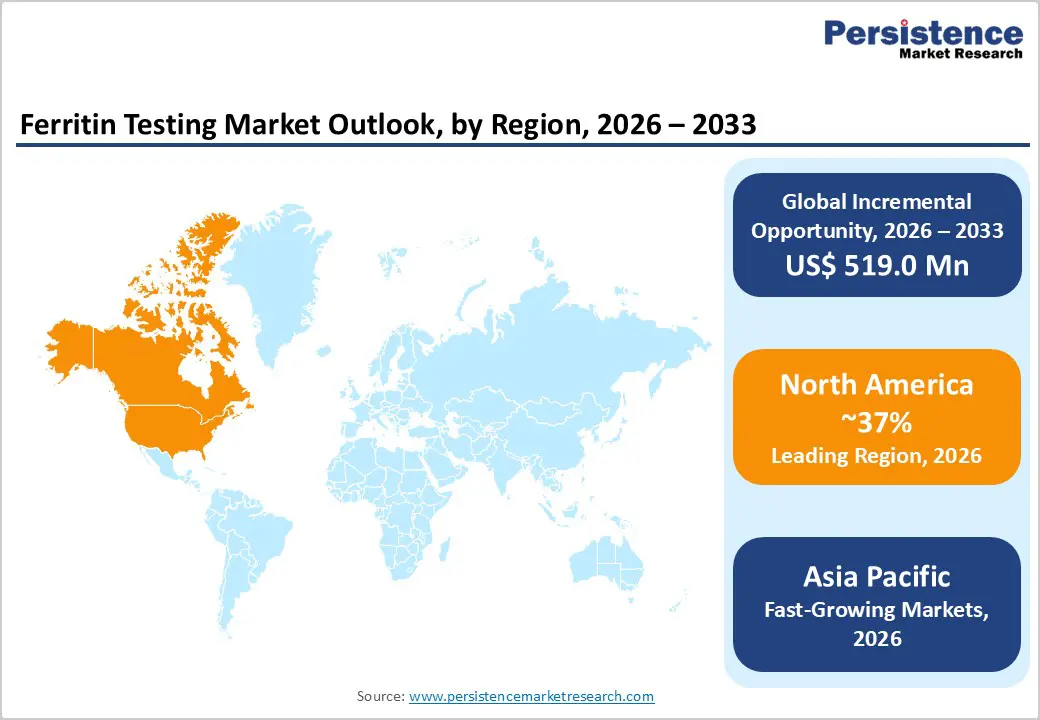
- North America leads the ferritin testing market with about 37% share in 2025, supported by advanced hospital and reference laboratory networks, strong guideline-driven use of ferritin in anemia and CKD management, and broad reimbursement that enables routine testing across the insured population.
- Asia Pacific is the fastest-growing region as high anemia prevalence intersects with rapid healthcare expansion, government-led programs such as Anemia Mukt Bharat, and increasing adoption of both automated immunoassay platforms and low-cost point-of-care tests that improve iron status assessment in underserved communities.
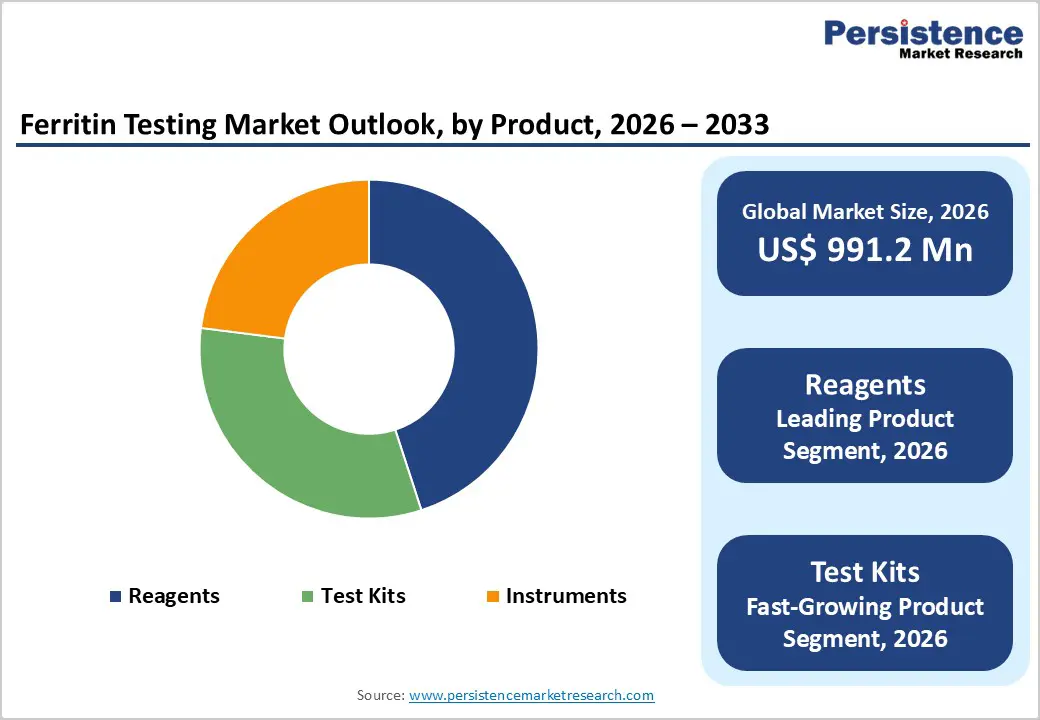
- On the product side, reagents dominate with around a 45% share in 2025, reflecting ongoing consumption tied to installed analyzers from major players such as Roche Diagnostics, Abbott Laboratories, and Siemens Healthineers, while test kits gain momentum in smaller laboratories and emerging markets.
- Test kits and rapid formats are the fastest-growing product segment, driven by demand from decentralized laboratories, diagnostic centers, and community settings that require standardized, ready-to-use solutions compatible with benchtop analyzers and point-of-care platforms, especially in resource-constrained regions.
| Key Insights | Details |
|---|---|
| Ferritin Testing Market Size (2026E) | US$ 991.2 Mn |
| Market Value Forecast (2033F) | US$ 1,510.2 Mn |
| Projected Growth (CAGR 2026 to 2033) | 6.2% |
| Historical Market Growth (CAGR 2020 to 2025) | 5.7% |
Market Dynamics
Drivers - Rising Global Burden of Iron Deficiency Anemia and Diagnostic Standardization
Iron deficiency anemia is one of the most common and impactful nutritional disorders globally, affecting an estimated 1.2 billion people and remaining a leading cause of years lived with disability, especially in low- and middle-income countries. The World Health Organization (WHO) reports that anemia affects about 40% of children aged 6-59 months and 36% of pregnant women worldwide, with roughly half of these cases attributable to iron deficiency, making ferritin-based assessment critical for prevention and control strategies. WHO guidance on assessing iron status and more recent global work on physiologically based ferritin thresholds emphasize serum ferritin as the central biomarker for classifying iron deficiency and monitoring response to interventions, thereby directly increasing clinical and programmatic demand for ferritin testing. In parallel, large epidemiological datasets and new evidence from adult iron deficiency reviews underscore that ferritin cutoffs higher than the older 15 μg/L threshold (for example, <30-50 μg/L) more accurately capture latent deficiency, effectively enlarging the test-eligible population and reinforcing ferritin testing as a standard investigation in primary care, women’s health, and pediatrics.
Expansion of Maternal Health Programs and Pregnancy Screening Mandates
Maternal and perinatal health policies are increasingly integrating ferritin testing into routine antenatal care to detect iron deficiency before anemia becomes clinically overt, which strongly supports sustained test volumes. Evidence from clinical trials and systematic reviews shows that iron deficiency during pregnancy, even in the absence of frank anemia, is associated with higher risks of preterm birth, low birth weight, and impaired neurodevelopment in offspring, leading professional bodies and national programs to encourage earlier and more sensitive iron status assessment using serum ferritin. The U.S. Preventive Services Task Force and other expert groups highlight iron deficiency screening in pregnancy as an area of active policy interest, while operational guidelines such as India’s Anemia Mukt Bharat (AMB) under the National Health Mission link the use of intravenous iron therapy and monitoring directly to ferritin-based assessment in pregnant women and high-risk populations. Studies in obstetric populations indicate that using higher, physiologically informed ferritin cutoffs can identify substantially more women with depleted stores than hemoglobin alone, supporting the intensified use of ferritin assays to optimize dosing strategies and the follow-up of iron therapy. As governments expand antenatal screening coverage and quality, particularly across Asia and other high-burden regions, ferritin testing becomes embedded in standardized care pathways, driving recurring demand from hospitals, public health clinics, and private obstetric practices.
Restraints - Interpretation Challenges in Inflammatory and Chronic Disease Contexts
Despite its central role, serum ferritin is an acute-phase reactant, and concentrations can increase significantly in the presence of infection, inflammation, liver disease, malignancy, or metabolic syndrome, complicating interpretation and limiting its stand-alone diagnostic utility. In chronic kidney disease (CKD), for example, multiple studies have shown that elevated ferritin may reflect inflammation rather than iron repletion, making it difficult to distinguish functional iron deficiency from true iron overload and potentially leading to inappropriate treatment decisions if used without complementary markers such as transferrin saturation or C-reactive protein. Reviews of iron status evaluation in CKD populations emphasize that ferritin alone has limited sensitivity and specificity for body iron stores, and that large intra-individual variability over time reduces the reliability of single measurements for guiding long-term management. Similarly, in populations being evaluated for hereditary hemochromatosis or metabolic hyperferritinemia, elevated ferritin levels may be driven by obesity, alcohol-related liver disease, or inflammatory conditions rather than genetic iron overload, necessitating further biochemical and genetic testing and constraining reliance on ferritin assays as definitive diagnostics in these cohorts.
Limited Accessibility and Cost Constraints in Resource-Limited Settings
The regions with the highest burden of iron deficiency anemia, including sub-Saharan Africa and parts of South Asia, often have the weakest laboratory infrastructure and constrained financial resources, limiting access to ferritin testing where it is needed most. Traditional ferritin assays are typically carried out on automated chemistry or immunoassay analyzers requiring stable electricity, temperature-controlled reagent storage, trained laboratory personnel, and supply chains for consumables, conditions that can be challenging to maintain in rural or peripheral health facilities. Public health guidance from organizations such as the CDC has long noted that the cost and technical requirements of ferritin testing are key reasons why many population-based anemia control programs rely on simpler hemoglobin measurements, even though they provide less information on iron status. In some low-income settings, patients may need to travel long distances or pay out-of-pocket for laboratory-based ferritin testing, which can reduce uptake and adherence to recommended diagnostic algorithms, slowing the pace at which ferritin testing becomes routine in primary care or antenatal services. While point-of-care devices and card tests are emerging, their deployment at scale remains limited by procurement budgets, regulatory approvals, and training capacity, keeping overall penetration below potential in many high-burden markets.
Opportunity - Point-of-Care Testing Decentralization and Home-Based Diagnostic Platforms
The shift from centralized, laboratory-bound ferritin testing to decentralized point-of-care and near-patient solutions presents a major opportunity to expand access and reinforce demand. Recent validation work has demonstrated that point-of-care ferritin tests using finger-prick blood, including lateral flow-based card tests, can achieve strong agreement with standard laboratory methods across clinically relevant cutoffs, while delivering results within minutes and requiring minimal equipment or power. Research into smartphone-integrated biosensors and paper-based microfluidic devices has shown that ferritin can be measured across a wide concentration range using compact, low-cost hardware, with some platforms reporting detection windows from around 5-6 μg/L to well over 800 μg/L, covering both deficiency and overload scenarios. These innovations are particularly attractive for maternal health outreach, school health programs, and chronic disease management in community clinics, where rapid decisions on iron supplementation or referral can be made at the point of contact. In higher-income markets, home-sampling kits and digital health solutions that bundle ferritin testing with telemedicine follow-up also create new consumer-driven channels, complementing traditional lab-based services and supporting monitoring for athletes, individuals on restrictive diets, or patients managing chronic conditions.
Companion Diagnostics for Chronic Disease Management and Therapeutic Monitoring
Another significant opportunity lies in embedding ferritin testing more deeply into chronic disease pathways, especially in nephrology, oncology, and cardiology. Draft and finalized guideline documents from expert groups such as Kidney Disease: Improving Global Outcomes (KDIGO) and national kidney foundations recommend regular assessment of iron status, including ferritin and transferrin saturation, to optimize iron therapy and erythropoiesis-stimulating agent dosing in CKD, both in dialysis and non-dialysis patients. In these settings, ferritin is not only used to diagnose deficiency but also to avoid iron overload, guide intravenous iron administration, and track responses to complex treatment regimens over time, which translates into higher testing frequency per patient. Beyond CKD, ferritin is being evaluated as a prognostic and monitoring biomarker in inflammatory and infectious diseases and in conditions associated with hyperferritinemic syndromes, providing further scope to integrate ferritin into multiparameter panels and algorithm-based decision support tools. As laboratory middleware and clinical decision support systems become more sophisticated, vendors can create value by combining high-quality ferritin assays with software that flags risk scenarios (for example, discordant ferritin and hemoglobin patterns or possible hemochromatosis), potentially differentiating their offerings and capturing premium demand in advanced hospital markets.
Category-wise Analysis
Product Insights
Reagents hold a leading position in the ferritin testing market, accounting for roughly 45% of total revenues in 2025, reflecting their central role as recurring consumables across automated analyzers and ELISA platforms. Clinical laboratories performing high-throughput ferritin testing rely on a continuous supply of specific antibody-coated particles, calibrators, controls, and reaction buffers to maintain consistent assay performance, and these reagents are often sold in proprietary formats tied to installed instruments from major manufacturers. Companies such as Roche Diagnostics, Abbott Laboratories, and Siemens Healthineers provide dedicated ferritin reagent packs for systems like Cobas, Architect, and ADVIA, which are validated for defined analytical ranges and traceability, reinforcing brand loyalty and long-term reagent pull-through. Test kits, including ELISA and CLIA kits designed for batch processing or lower-throughput settings, represent the fastest-growing product subsegment as smaller laboratories, diagnostic centers, and emerging point-of-care platforms adopt standardized kits that simplify setup and reduce the validation burden. This growth is further aided by the availability of ready-to-use kits from specialist firms serving both clinical and research markets, often in flexible formats that can be deployed on generic microplate readers or benchtop analyzers.
Application Insights
Iron deficiency anemia is the dominant application area for ferritin testing, accounting for nearly 48% of total demand in 2025, because ferritin is widely recognized as the best single biomarker to confirm depleted iron stores in both clinical practice and public health programs. WHO and other global agencies emphasize iron deficiency anemia as a key target for reduction, and current estimates indicate that hundreds of millions of women of reproductive age, children, and adolescents require a comprehensive assessment of iron status, which in many settings now includes ferritin alongside hemoglobin. Clinical guidelines in primary care and internal medicine increasingly recommend ferritin measurement in adults with fatigue, unexplained anemia, heavy menstrual bleeding, gastrointestinal symptoms suggestive of occult blood loss, and in candidates for bariatric surgery or those following restrictive diets, further widening the test base.
Pregnancy and maternal screening is the fastest-growing application segment as more programs adopt ferritin rather than hemoglobin alone to detect iron deficiency early. Studies show that a substantial proportion of pregnant women have low ferritin despite acceptable hemoglobin values, and that iron supplementation guided by ferritin can improve maternal and fetal outcomes. Additional growth stems from its use in monitoring iron overload in hereditary hemochromatosis and transfusion-dependent anemias, as well as in inflammatory and chronic diseases (such as CKD and rheumatoid arthritis) where ferritin forms part of broader anemia of chronic disease work-ups.
Regional Insights
North America Ferritin Testing Market Trends and Insights
North America is the leading regional market, accounting for approximately 37% share in 2025, underpinned by strong healthcare infrastructure, high testing capacity, and well-established clinical guidelines that incorporate ferritin into diagnostic algorithms. In the United States, national survey data indicate notable anemia prevalence among specific groups, and public health authorities such as the CDC highlight iron deficiency as a significant contributor, reinforcing the need for comprehensive assessment of iron status in women, children, and patients with chronic disease. Large integrated health systems and commercial laboratories operate extensive networks of automated immunoassay analyzers from vendors like Abbott Laboratories, Roche Diagnostics, and Siemens Healthineers, ensuring that ferritin testing is widely available and often bundled into standard anemia and CKD panels.
Asia Pacific Ferritin Testing Market Trends and Insights
Asia Pacific is the fastest-growing regional market for ferritin testing, driven by a combination of high anemia burden, expanding healthcare infrastructure, and active government programs targeting maternal and child health. Countries such as India and China account for a large proportion of the world’s anemic population, with surveys and research articles documenting substantial prevalence of iron deficiency anemia among children, adolescents, and women of reproductive age, making ferritin a crucial tool for more accurate assessment of iron status as programs shift beyond hemoglobin-only screening. In India, the Anemia Mukt Bharat initiative and associated operational guidelines for intravenous iron therapy in pregnancy explicitly highlight the need for robust diagnosis and monitoring, providing a policy foundation to support broader adoption of ferritin testing across the public and private sectors.
Competitive Landscape
The ferritin testing market shows a moderately consolidated competitive landscape, driven by strong demand for anemia diagnosis and chronic disease monitoring. Competition is primarily based on test accuracy, sensitivity, automation compatibility, and turnaround time. Manufacturers focus on expanding reagent portfolios and developing easy-to-use test kits to support high-volume laboratories and decentralized testing settings. Technological differentiation through chemiluminescence and rapid testing formats plays a key role in gaining market share. Pricing competitiveness and long-term reagent supply agreements influence buyer decisions, especially in hospital laboratories.
Key Developments:
- In August 2025, Daye launched a comprehensive at-home hormone testing service in the UK, using what it described as pain-free blood-collection technology. The service addressed conditions such as PCOS, thyroid disorders, and fertility concerns through upper arm blood collection instead of traditional finger-prick methods. The testing panels include vitamin D and ferritin levels, which the company describes as often-overlooked nutrients essential for reproductive health.
Companies Covered in Ferritin Testing Market
- Abbott Laboratories
- Thermo Fisher Scientific
- Roche Diagnostics
- Siemens Healthineers
- Bio-Rad Laboratories
- DiaSorin
- Beckman Coulter
- QuidelOrtho Corporation
- Abnova Corporation
- bioMérieux
- Eurolyser Diagnostica
- Cortez Diagnostics
- Others
Frequently Asked Questions
The global ferritin testing market is expected to be valued at US$ 3.7 million in 2026, and is projected to reach about US$ 5.6 million by 2033, reflecting a forecast CAGR of approximately 6.2% between 2026 and 2033.
Major demand drivers include the high and persistent global burden of iron deficiency anemia affecting hundreds of millions of women and children, greater emphasis on accurate iron status assessment in clinical and public health guidelines, expansion of maternal and antenatal screening programs, and rising use of ferritin in chronic disease pathways such as chronic kidney disease and hereditary hemochromatosis.
North America is the leading region, supported by advanced diagnostic infrastructure, strong presence of major IVD manufacturers, guideline-driven use of ferritin in anemia and CKD management, and reimbursement systems that facilitate routine testing in both hospital and outpatient settings.
The most significant opportunity is the expansion of point-of-care and decentralized ferritin testing, including lateral flow card tests and smartphone-based biosensors that provide rapid, low-cost results from capillary blood. These technologies can greatly improve access in low-resource settings and support large-scale screening in maternal health, school health, and community anemia programs.
Key players include Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, and Thermo Fisher Scientific, along with Bio-Rad Laboratories, DiaSorin, Beckman Coulter, QuidelOrtho Corporation, bioMérieux, Eurolyser Diagnostica, Abnova Corporation, and Cortez Diagnostics, which together provide a wide spectrum of automated assays, reagents, test kits, and point-of-care platforms serving clinical and research customers worldwide.







