- Biotechnology
- Europe Platelet Rich Plasma Market
Europe Platelet Rich Plasma Market Size, Share, and Growth Forecast 2026 - 2033
Europe Platelet Rich Plasma Market by Product Type (Pure Platelet Rich Plasma (P-PRP), Leukocyte-Rich Platelet Rich Plasma (L-PRP), Leukocyte-Platelet-Rich Fibrin (L-PRF), Pure Platelet Rich Fibrin (P-PRF), Others), by Origin, by Application, by Regional Analysis, 2026 - 2033
Europe Platelet Rich Plasma Market Size and Trend Analysis
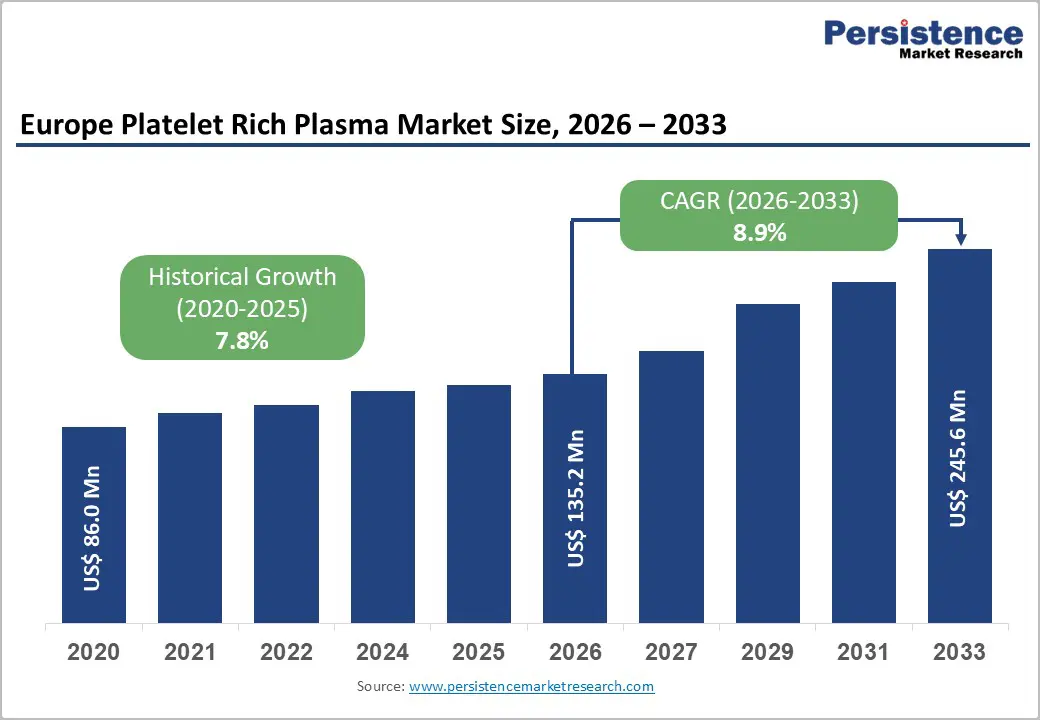
The European platelet-rich plasma market size is expected to be valued at US$ 135.2 million in 2026 and projected to reach US$ 245.6 million by 2033, growing at a CAGR of 8.9% between 2026 and 2033. The European platelet-rich plasma market growth is rising rapidly, driven by increasing demand for regenerative therapies across various medical fields such as orthopedics, sports medicine, cosmetics, and dentistry. PRP, which uses a patient's own platelet-rich plasma, has gained popularity for its ability to promote natural healing with minimal side effects.
The market growth is set to be fuelled by the increasing prevalence of musculoskeletal injuries, demand for aesthetic treatments, and rising cases of age-related degenerative conditions, which are common among Europe’s aging population. Trends in the region’s platelet rich plasma therapy industry indicate a growing preference for autologous PRP as it reduces infection risks and immune responses, aligning with stringent healthcare regulations.
Pure platelet-rich plasma (P-PRP) is also leading among product types due to its low leukocyte content, reducing inflammation and providing effective outcomes. Technological advancements in PRP preparation systems are simplifying procedures, making treatments highly accessible. As awareness and acceptance of PRP therapy continue to rise, Europe is anticipated to extend significantly, with an emphasis on minimally invasive and patient-specific treatment options.
Key Industry Highlights:
- Germany leads the Europe platelet rich plasma market due to advanced Orthopedic care, strong sports medicine demand, and strict EU MDR compliance.
- Italy is the fastest-growing PRP market in Europe, driven by expanding clinical research, rising Orthopedic adoption, and growing aesthetic regenerative treatments.
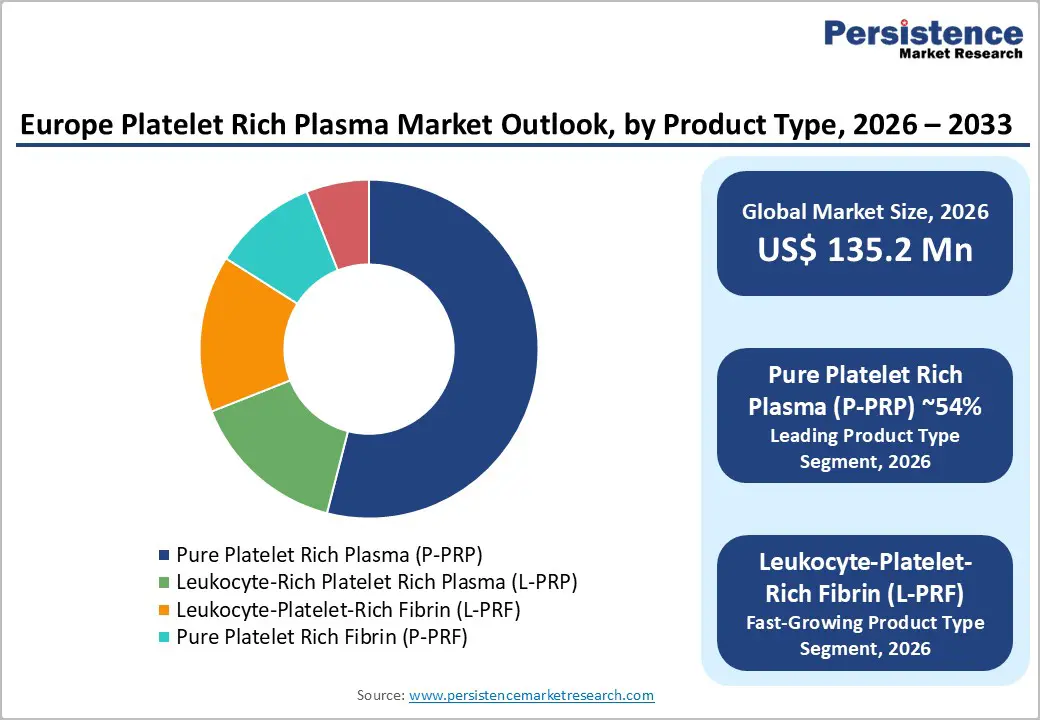
- Pure Platelet Rich Plasma (P-PRP) dominates the market with over half of total share, supported by clinical familiarity, safety profile, and CE-marked preparation systems.
- Orthopedic surgery holds the largest application share, as PRP is widely used for knee osteoarthritis, tendon injuries, sports medicine, and minimally invasive treatments.
| Key Insights | Details |
|---|---|
| Platelet Rich Plasma Market Size (2026E) | US$ 135.2 million |
| Market Value Forecast (2033F) | US$ 245.6 million |
| Projected Growth CAGR (2026 - 2033) | 8.9% |
| Historical Market Growth (2020 - 2025) | 7.8% |
Market Dynamics
Drivers - Growing Use of PRP in Dentistry, Dermatology, and Aesthetics to Boost Demand
The Europe platelet rich plasma market is set to witness a surging use across diverse therapeutic and clinical fields. It is mainly driven by the growing appeal of regenerative treatments that leverage the body’s own healing mechanisms. PRP’s applications have expanded beyond orthopedics and sports medicine to encompass aesthetics, dermatology, dentistry, wound care, and even hair restoration. This growing use is fueled by increasing demand for minimally invasive, natural therapies with fewer side effects from patients.
PRP aids in joint, ligament, and tendon repair in orthopedics, while in aesthetics, it is used for anti-aging treatments and scar reduction. Increasing prevalence of chronic conditions and surging aging population in Europe further support PRP adoption, as patients seek effective solutions to manage pain, heal injuries, and enhance quality of life. Continuous advancements in PRP preparation technologies and strong support from healthcare providers are additional drivers, positioning the Europe PRP market for significant growth across multiple medical fields.
High Demand for Minimally Invasive Cosmetic Treatments to Bolster Sales
Europe market is experiencing significant growth due to the rising demand for cosmetic and aesthetic treatments. Consumers are increasingly seeking natural, minimally invasive options to improve skin texture, reduce wrinkles, and promote hair growth. This is further leading to a rising adoption of PRP in beauty and anti-aging therapies.
PRP’s ability to stimulate collagen production and accelerate tissue repair makes it highly effective for skin rejuvenation and scar reduction. It aligns with the preferences of Europe’s health-conscious and aging population. PRP treatments appeal to individuals looking for low-risk solutions, as they use the patient's own blood, reducing the risk of adverse reactions. This demand, combined with innovations in PRP application methods, is a strong driver for growth, solidifying Europe’s position as a key market for cosmetic PRP treatments.
Restraints - Stringent Regulatory Framework May Hamper Platelet Rich Plasma Therapy Demand
Regulatory delays stemming from the need for FDA approval are set to significantly hamper growth in the market. The stringent regulatory framework established by the FDA necessitates thorough evaluations and extensive clinical trials before products can gain market access.
Protracted approval process can deter innovation and slow down the introduction of new PRP therapies, limiting options for healthcare providers and patients. Hence, companies may hesitate to invest in research and development activities or face challenges in meeting regulatory requirements, ultimately affecting the competitiveness of Europe’s market.
Opportunities - Innovation in Leukocyte-Platelet-Rich Fibrin and Advanced Formulations
Significant opportunity exists in the development of advanced platelet concentrates, particularly leukocyte-platelet-rich fibrin (L-PRF) and pure platelet rich fibrin (P-PRF), which form fibrin matrices that may support sustained release of growth factors and improved tissue regeneration. Clinical reports suggest that L-PRF membranes can enhance bone and soft-tissue healing in dental, maxillofacial, and orthopedic procedures by providing some scaffold rich in platelets and leukocytes. As European surgeons gain experience with these second-generation platelet concentrates, demand for dedicated centrifugation systems and kits that can reliably produce L-PRF and P-PRF is expected to rise. Manufacturers that generate robust comparative data on outcomes versus traditional P-PRP, and that secure MDR-compliant device approvals, will be well-positioned to capture the fastest-growing niche within the platelet rich plasma market.
Popularity of Social Media Creates Opportunities for Cosmetic PRP Treatment Providers
The trend for significant skin enhancement through cosmetic PRP treatments represents a valuable opportunity in the Europe platelet rich plasma market. As consumers increasingly seek non-surgical solutions for improving skin texture, tone, and appearance, PRP therapy has emerged as an appealing option due to its natural and regenerative properties. The ability of PRP to stimulate collagen production and promote skin rejuvenation aligns with the growing trend toward minimally invasive cosmetic procedures.
The popularity of social media and influencer culture has surged awareness and demand for effective skincare treatments, further driving interest in PRP. This trend offers manufacturers and practitioners an opportunity to broaden their offerings, educate potential clients, and capitalize on the burgeoning aesthetic market in Europe.
Category-wise Insights
By Product Type
Pure platelet rich plasma (P-PRP) is leading in terms of product type due to its broad applicability and high safety profile. P-PRP, containing a high concentration of platelets without leukocytes minimizes inflammation and risk of immune response. This makes it ideal for regenerative treatments in orthopedics, sports medicine, and aesthetics.
Its efficacy in accelerating tissue healing and repair has gained the trust of healthcare providers and patients seeking minimally invasive options. Additionally, P-PRP’s versatility allows it to be easily integrated into various therapeutic procedures, supporting its dominance in Europe’s platelet rich plasma shots industry.
By Origin
By origin, the autologous segment is anticipated to lead with a high platelet rich plasma market share in Europe. Growth is primarily attributed to its high safety and compatibility.
Derived from the patient’s own blood, autologous PRP significantly reduces the risk of immune reactions, infections, and transmission of diseases. These features make it the preferred choice in clinical applications across Europe.
Superior compatibility of autologous PRP ensures better patient outcomes, especially in treatments like orthopedics, sports injuries, and cosmetic procedures, where rapid healing and minimal side effects are essential. Furthermore, Europe’s regulatory standards favor autologous treatments for their high safety. This encourages widespread adoption among local healthcare providers, driving market growth in the region.
Regional Insights
Germany Platelet Rich Plasma Market Trends and Insights
Germany is the leading country market within Europe for platelet rich plasma, accounting for an estimated 29% share of regional revenues in 2025. The country hosts a dense network of orthopedic and sports medicine centers, many of which have been early adopters of PRP injections for knee osteoarthritis, tendinopathies, and ligament injuries. German hospitals and private clinics have also integrated PRP into aesthetic dermatology offerings, including hair restoration and facial rejuvenation procedures, reflecting strong consumer demand for non-surgical regenerative treatments.
Germany’s position is reinforced by strict adherence to EU MDR 2017/745 and national implementations of blood and tissue directives, which ensure that PRP preparation devices are CE-marked and that blood handling follows high quality and safety standards. Recent court enforcement actions against non-compliant PRP tubes at major medical trade fairs underscore the emphasis on regulatory compliance and device quality. Combined with advanced healthcare infrastructure and high per-capita spending on orthopedic and aesthetic procedures, these factors sustain Germany’s leadership in the Europe platelet rich plasma market.
Italy Platelet Rich Plasma Market Trends and Insights
Italy is among the fastest-growing platelet rich plasma markets in Europe, supported by strong clinical research activity and increasing use of PRP in both public and private healthcare settings. Italian orthopedic research centers have contributed significantly to the global evidence base on PRP for knee osteoarthritis, leading multicenter randomized trials and meta-analyses that document meaningful improvements in pain and function at 6-12 months follow-up. This research visibility has encouraged broader adoption of PRP injections in sports medicine, trauma, and degenerative joint disease across Italian hospitals and specialist clinics.
In parallel, Italy has a vibrant aesthetic medicine sector where PRP is used for facial rejuvenation, scar remodeling, and hair restoration, often marketed as a natural, autologous alternative to synthetic fillers or pharmacologic therapies. Compliance with EU MDR and national rules on blood-derived products ensures that PRP preparation devices meet stringent safety and performance criteria, supporting practitioner and patient confidence. As awareness of regenerative therapies grows among clinicians and consumers, Italy is expected to outpace many peer markets in PRP procedure growth over the forecast period.
Competitive Landscape
The competitive landscape of Europe platelet rich plasma market is characterized by a mix of established companies and emerging players, each striving to innovate and capture high shares. Key industry participants include biotechnology firms, medical device manufacturers, and specialized clinics offering PRP treatments across various applications. These applications include orthopaedics, aesthetics, and wound healing.
Competitive strategies often focus on enhancing product efficacy, optimizing processing techniques, and broadening distribution channels. Additionally, collaborations and partnerships between companies and healthcare providers are becoming increasingly common, aiming to enhance research and development efforts.
The presence of several clinical studies supporting the effectiveness of PRP therapy also fosters competition. It is attributed to the validation of treatment protocols and the emergence of new entrants seeking to explore this evolving market. As a result, the regional market is evolving rapidly, driven by innovation, regulatory advancements, and rising consumer awareness.
Key Market Developments
- In December 2025, the Düsseldorf District Court issued a preliminary injunction against non-CE-marked PRP tubes exhibited at MEDICA 2025, reinforcing strict enforcement of EU MDR 2017/745 rules for PRP preparation devices and signaling higher compliance expectations across Europe.
- In August 2024, the Swiss Agency for Therapeutic Products (Swissmedic) revised its norms by replacing the preparations of platelet-rich fibrinogen and platelet-rich plasma with the preparation group ‘platelet products for allogeneic or autologous use.’ Additionally, the agency included information regarding establishment licenses for the production and distribution of non-standardized pharmaceuticals.
- In February 2024, Dynamic Regenerative Medicine, based in Birmingham, announced the introduction of its advanced Platelet Rich Plasma (PRP) injection treatment with dynamic regenerative medicine. The clinic aims to provide long-lasting and safe treatment options for those suffering from orthopedic and musculoskeletal conditions.
Companies Covered in Europe Platelet Rich Plasma Market
- Arthrex, Inc.
- Stryker Corporation
- Johnson and Johnson Ltd.
- Zimmer Biomet Holdings Inc.
- Terumo Corporation
- Glofinn Oy
- Medira Ltd.
- Regen Lab S.A.
- Others
Frequently Asked Questions
The Europe platelet rich plasma market size is expected to reach around US$ 135.2 million in 2026.
Demand is primarily driven by growing clinical use of PRP in knee osteoarthritis and sports injuries, rising adoption in aesthetic dermatology and hair restoration, and a supportive regulatory framework for MDR-compliant PRP preparation devices that ensures safety and quality.
Germany currently leads the Europe platelet rich plasma market, benefiting from advanced orthopedic and sports medicine infrastructure, high patient awareness of regenerative therapies, and strict enforcement of EU MDR 2017/745 requirements for PRP preparation devices.
Key opportunities include the development of advanced platelet concentrates such as Leukocyte-Platelet-Rich Fibrin (L-PRF) and the expansion of autologous and emerging allogeneic PRP into new indications like chronic wounds and complex orthopedic conditions, supported by robust European clinical research.
Key players include Arthrex, Inc., Stryker Corporation, Johnson and Johnson Ltd., Zimmer Biomet Holdings Inc., Terumo Corporation, Glofinn Oy, Medira Ltd., Regen Lab S.A., and Dr. PRP Europe, among other regional device manufacturers.








