- Biotechnology
- Platelet Rich Plasma Market
Platelet Rich Plasma Market Size, Share, Growth, and Regional Forecast, 2026 - 2033
Platelet Rich Plasma Market by Product (Pure Platelet Rich Plasma (Pure PRP), Leukocyte-rich Platelet Rich Plasma (L-PRP), Platelet Rich Fibrin (PRF), and Others), by Application (Orthopedics, Neurology, Cosmetics, Wound Care, and Others) by End User (Hospitals, Specialty Clinics, Ambulatory Surgery Centers, and Academic and Research Institutes), and Regional Analysis from 2026 - 2033
Platelet Rich Plasma Market Share and Trend Analysis
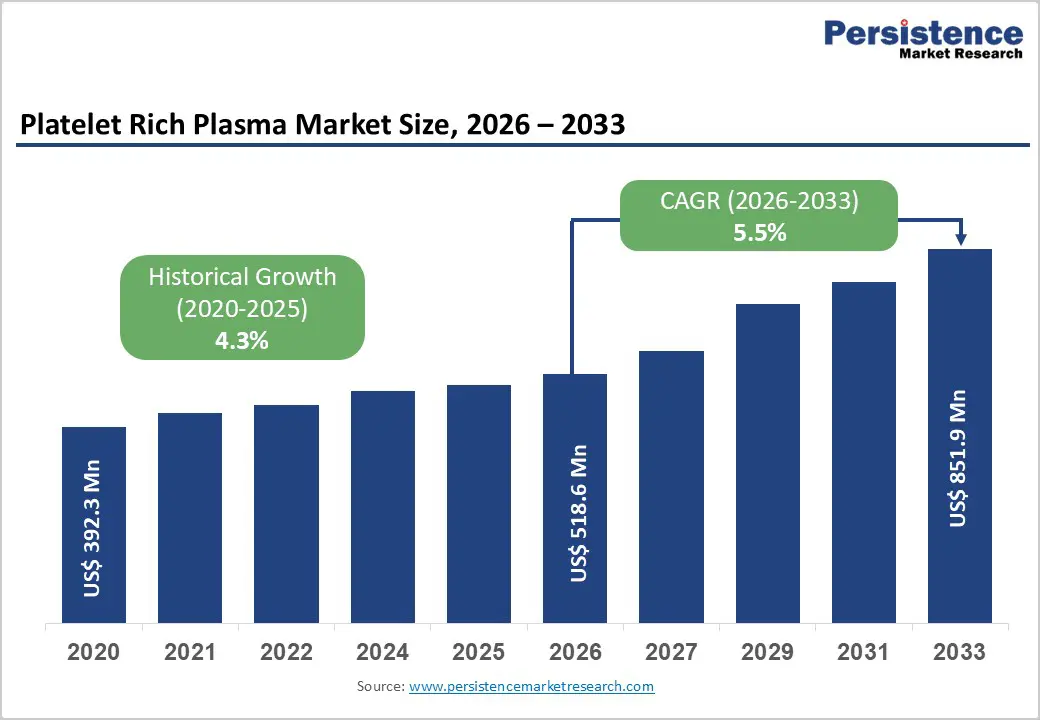
The global platelet-rich plasma market size is estimated to grow from US$ 518.6 Mn in 2026 to US$ 851.9 Mn by 2033. The market is projected to record a CAGR of 5.5% during the forecast period from 2026 to 2033. Adoption of platelet-based regenerative therapies is accelerating worldwide as healthcare providers increasingly incorporate biologic treatments that promote natural tissue repair and lead to faster recovery. Platelet-rich plasma procedures are gaining traction across orthopedics, dermatology, dentistry, and wound management due to their autologous nature and reduced risk of immune complications.
The rising prevalence of musculoskeletal disorders, sports injuries, and age-related degenerative conditions is expanding clinical utilization, while the growing demand for minimally invasive aesthetic procedures is further strengthening procedural volumes. Wider availability of standardized preparation kits and automated centrifugation systems is improving treatment consistency and enabling point-of-care applications across hospitals and specialty clinics. Continuous clinical research validating therapeutic benefits, along with increasing physician training programs, is supporting broader acceptance. In addition, the expansion of private healthcare infrastructure and outpatient treatment models is improving accessibility in emerging markets. Advancements in sterile processing workflows and simplified preparation technologies are helping providers deliver efficient, repeatable outcomes, contributing to sustained long-term growth across both developed and developing healthcare ecosystems.
Key Industry Highlights:
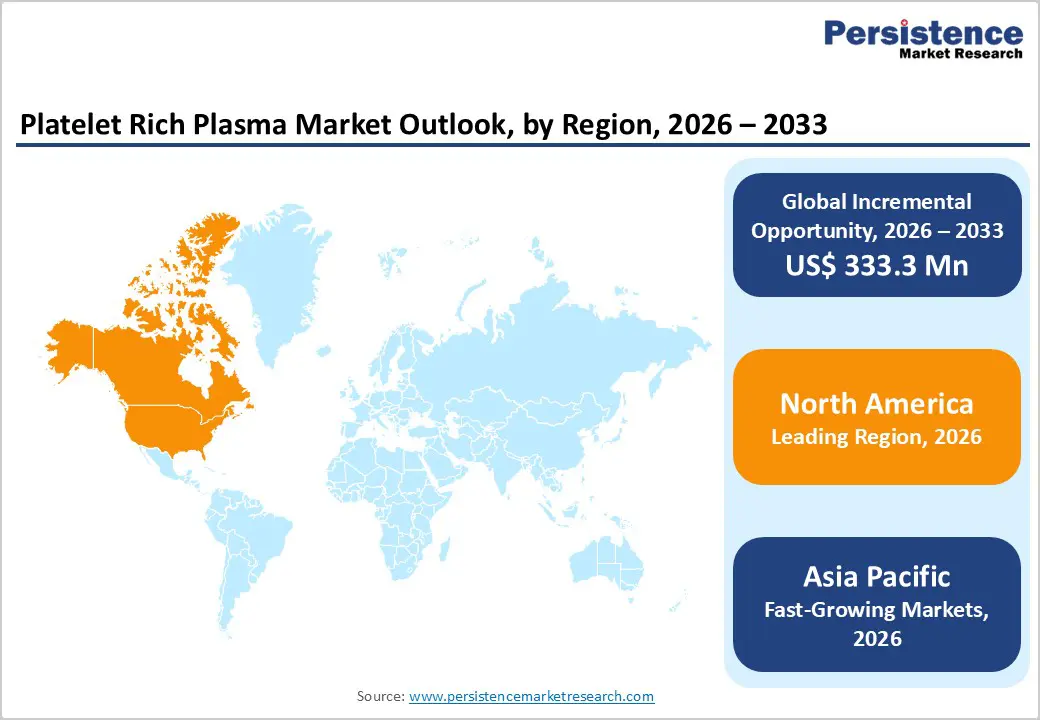
- Leading Region: North America accounts for 46.7% of the global market, supported by strong clinical adoption, advanced healthcare infrastructure, high awareness of regenerative therapies, and early integration of PRP procedures across orthopedic and aesthetic practices.
- Fastest-Growing Region: Asia Pacific is expanding at the fastest pace due to improving healthcare access, rising medical tourism, increasing disposable income, and growing acceptance of non-surgical regenerative and cosmetic treatments.
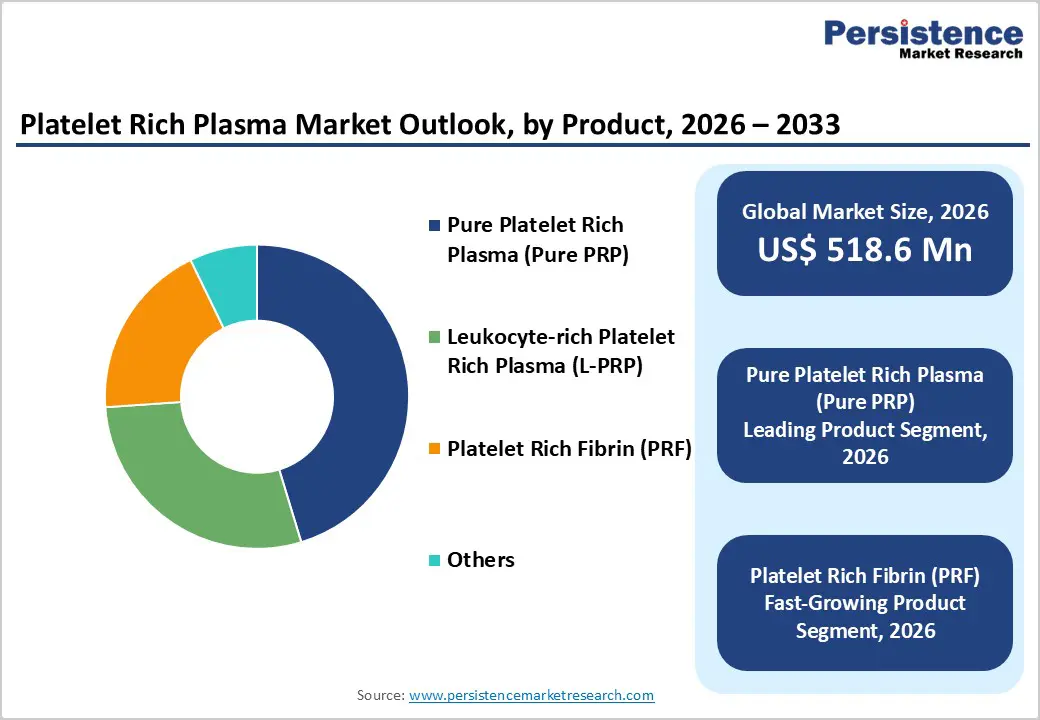
- Leading Product Segment: Pure Platelet Rich Plasma (Pure PRP) leads the market due to predictable therapeutic outcomes, a lower inflammatory response, and extensive use across dermatology, dentistry, and orthopedics.
- Fastest-Growing Product Segment: Platelet Rich Fibrin (PRF) is gaining rapid momentum as clinicians adopt fibrin-matrix formulations that enable sustained growth-factor release and enhanced tissue regeneration.
- Leading Application Segment: Orthopedics remains the dominant application area, driven by rising osteoarthritis cases, sports injury treatments, and a growing preference for minimally invasive pain management solutions.
- Fastest-Growing Application Segment: Cosmetics is witnessing accelerated growth driven by rising demand for hair restoration, facial rejuvenation, and non-surgical anti-aging procedures worldwide.
| Key Insights | Details |
|---|---|
|
Platelet Rich Plasma Market Size (2026E) |
US$ 518.6 Mn |
|
Market Value Forecast (2033F) |
US$ 851.9 Mn |
|
Projected Growth (CAGR 2026 to 2033) |
5.5% |
|
Historical Market Growth (CAGR 2020 to 2025) |
4.3% |
Market Dynamics
Driver - Growing Preference for Minimally Invasive Regenerative Therapies and Expanding Clinical Acceptance
The increasing global shift toward regenerative and autologous treatment approaches is substantially accelerating the adoption of platelet-based biologic therapies across multiple medical disciplines. Physicians and patients are increasingly favoring procedures that stimulate natural healing rather than relying solely on pharmaceuticals or invasive surgical interventions. Platelet-rich plasma has gained strong clinical traction due to its ability to deliver concentrated growth factors that promote tissue repair, angiogenesis, and inflammation modulation without introducing foreign biomaterials. The rising incidence of osteoarthritis, tendon injuries, spinal disorders, and sports-related trauma is creating sustained demand, particularly within orthopedic and sports medicine practices. Simultaneously, rapid growth in aesthetic medicine, including hair restoration, skin rejuvenation, and scar revision, has expanded procedural volumes in outpatient settings. Improvements in centrifugation technologies and standardized preparation kits have enhanced treatment consistency, improving physician confidence and patient outcomes. Worldwide, the aging population is further contributing to the prevalence of degenerative conditions.
Additionally, growing awareness through clinical studies and physician training programs is strengthening acceptance among healthcare providers. As healthcare systems emphasize faster recovery, reduced hospitalization, and cost-effective therapies, biologic treatments such as PRP are increasingly integrated into mainstream treatment protocols, thereby reinforcing long-term market expansion.
Restraints - Lack of Standardized Protocols and Variability in Clinical Outcomes
Despite rising adoption, several structural challenges continue to limit the broader penetration of platelet-based therapies. One of the most significant barriers is the absence of universally standardized preparation and administration protocols. Variations in centrifugation speed, platelet concentration levels, leukocyte content, and activation techniques can lead to inconsistent therapeutic outcomes, creating skepticism among clinicians and payers. Differences between commercially available preparation systems further complicate comparability across clinical studies, slowing regulatory alignment and evidence-based guideline development. Reimbursement limitations also restrict adoption in many regions, as PRP procedures are often categorized as elective or experimental, requiring patients to pay out of pocket.
Additionally, clinical efficacy remains highly dependent on practitioner expertise and patient-specific biological factors, which introduces variability in treatment success rates. Limited long-term clinical data for certain applications, particularly neurology and advanced regenerative indications, continues to affect acceptance among conservative healthcare systems. Training requirements and the need for sterile handling environments can also pose operational challenges for smaller clinics. Collectively, these factors create uncertainty regarding treatment predictability and economic value, moderating adoption despite strong interest in regenerative medicine solutions.
Opportunity - Expansion into Aesthetic Medicine, Sports Rehabilitation, and Next-Generation Biologic Combinations
Significant growth opportunities are emerging as platelet-based therapies expand beyond traditional orthopedic applications into high-growth medical and wellness segments. Aesthetic dermatology represents a particularly strong avenue, with increasing consumer demand for non-surgical anti-aging treatments and hair restoration procedures driving adoption in specialty clinics worldwide. Integration of PRP with complementary regenerative technologies, including stem cell therapies, microneedling, and biomaterial scaffolds, is opening new therapeutic pathways to enhance tissue regeneration outcomes. Sports rehabilitation programs are also incorporating biologic injections to accelerate recovery timelines for professional and amateur athletes, creating recurring treatment demand. Technological advancements such as automated preparation devices, closed sterile systems, and point-of-care processing platforms are improving workflow efficiency and enabling broader clinical accessibility.
Emerging markets present additional opportunities as private healthcare infrastructure expands and patients' awareness of regenerative medicine increases. Furthermore, ongoing clinical research exploring PRP applications in chronic wound management, dentistry, and nerve repair may unlock new revenue streams. As innovation continues to improve treatment consistency and clinical validation strengthens, PRP therapies are positioned to evolve from niche procedures into widely accepted components of personalized regenerative healthcare.
Category-wise Analysis
By Product Insights
Pure Platelet Rich Plasma (Pure PRP) is projected to retain market leadership in 2026, accounting for a 45.3% revenue share. Its dominance is primarily linked to predictable biological performance and reduced inflammatory response compared with leukocyte-rich formulations. Clinicians widely prefer Pure PRP in dermatology, dentistry, and aesthetic medicine, where controlled healing and minimal post-procedure irritation are critical. The technology enables consistent platelet concentration while limiting leukocyte activity, improving procedural reproducibility across outpatient environments. Increasing adoption of closed-system preparation kits and automated centrifugation platforms has further strengthened utilization by minimizing contamination risks and operator variability. In addition, expanding demand for hair restoration, facial rejuvenation, and soft-tissue regeneration procedures supports recurring procedural volumes. Continuous innovation in disposable kits, simplified preparation workflows, and point-of-care biologic processing systems ensures scalability for both hospitals and specialty clinics, reinforcing Pure PRP as the most commercially viable and clinically versatile formulation segment worldwide.
By Application Insights
The orthopedics segment is expected to remain the largest application area in 2026, capturing 32.8% of the global market. Growth is closely tied to the increasing prevalence of osteoarthritis, tendon injuries, ligament damage, and age-related degenerative joint conditions. PRP therapy has gained strong physician acceptance as a minimally invasive alternative to corticosteroids and surgical interventions, particularly for knee osteoarthritis and chronic tendon pathologies. Sports medicine programs increasingly incorporate PRP injections to accelerate tissue repair and reduce recovery timelines for athletes, further strengthening procedural demand. Advancements in imaging-guided injection techniques have improved treatment accuracy and clinical confidence, encouraging wider adoption across orthopedic practices. Additionally, growing patient preference for regenerative therapies that utilize autologous biological material aligns with global healthcare trends favoring natural healing approaches. As healthcare systems emphasize outpatient musculoskeletal management and cost-effective pain reduction strategies, orthopedic applications continue to represent the most stable and procedure-driven revenue contributor.
By End-user Insights
Hospitals are anticipated to hold a 47.4% market share in 2026, maintaining their position as the primary delivery setting for platelet-rich plasma therapies. Their leadership stems from access to multidisciplinary specialists, advanced diagnostic imaging, and established surgical ecosystems that support complex orthopedic and wound-care procedures. Hospitals also manage higher patient inflow involving trauma cases, chronic joint disorders, and post-operative recovery treatments where PRP is increasingly integrated into care pathways. Institutional adoption benefits from standardized protocols, trained clinical staff, and strict sterility controls, enabling consistent biologic preparation and administration. Furthermore, collaborations between hospitals and regenerative medicine researchers are accelerating clinical validation studies, strengthening physician confidence and reimbursement discussions. Investments in biologics processing units and integrated centrifugation systems allow efficient same-day preparation, improving workflow efficiency. As healthcare providers continue transitioning toward regenerative and minimally invasive therapies, hospitals remain central to the global utilization of PRP.
Regional Insights
North America Platelet Rich Plasma Market Trends
North America is expected to retain market leadership in 2026, accounting for a 46.7% value share, supported by strong clinical adoption and a mature regenerative medicine ecosystem. The United States drives regional expansion through high awareness among physicians and patients regarding biologic therapies for orthopedic pain management and aesthetic enhancement. Favorable reimbursement scenarios in selected orthopedic indications, combined with the growth of private-pay cosmetic procedures, have accelerated procedural volumes.
The region benefits from early commercialization of PRP preparation technologies, widespread availability of FDA-cleared systems, and strong physician training networks that promote standardized treatment protocols. Sports medicine institutes and professional athletic organizations actively utilize PRP to shorten rehabilitation timelines, reinforcing clinical credibility. Additionally, increasing demand for hair restoration and anti-aging procedures across dermatology clinics continues to expand outpatient adoption. Continuous innovation in automated preparation devices and closed sterile kits, along with ongoing clinical research validating efficacy, ensures North America maintains technological and commercial leadership within the global PRP landscape.
Europe Platelet Rich Plasma Market Trends
Europe’s platelet-rich plasma market is projected to expand steadily in 2026, supported by advanced healthcare infrastructure and robust regulatory oversight across Germany, France, Switzerland, and Italy. Clinical adoption in the region is strongly influenced by evidence-based medicine, encouraging standardized PRP preparation protocols and controlled therapeutic applications. Orthopedic rehabilitation and dental implantology remain major contributors to demand, particularly as aging populations increase the incidence of degenerative musculoskeletal conditions.
European practitioners demonstrate high adoption of leukocyte-controlled formulations designed to optimize healing while minimizing inflammation. Sustainability and cost-efficiency also influence purchasing decisions, leading healthcare providers to favor reusable centrifugation platforms combined with certified sterile consumables. Academic hospitals and research institutes actively conduct clinical trials exploring PRP use in chronic wound management and regenerative dentistry, expanding the application scope. Integration of digital patient monitoring and outcome tracking further strengthens treatment validation, allowing Europe to maintain a stable, high-value position driven by clinical precision and long-term therapeutic reliability.
Asia Pacific Platelet Rich Plasma Market Trends
Asia Pacific is projected to be the fastest-expanding regional market, registering a CAGR of approximately 7.4% between 2026 and 2033. Rapid development of healthcare infrastructure across China and India is significantly improving access to regenerative therapies in both urban hospitals and private specialty clinics. Rising disposable incomes and increasing acceptance of aesthetic procedures, including hair restoration and skin rejuvenation, are accelerating PRP adoption among younger patient populations. Governments across the region are investing in modernizing orthopedic care and sports medicine facilities, supporting broader use of biologic treatments for injury recovery.
Cost-efficient local manufacturing of centrifugation systems and consumables has improved affordability, enabling smaller clinics to introduce PRP services. Furthermore, the expansion of medical tourism in countries such as South Korea, Thailand, and India is driving demand for minimally invasive cosmetic procedures. Growing physician training programs and awareness initiatives are improving clinical confidence, positioning Asia Pacific as the most influential growth engine shaping the next phase of global PRP market expansion.
Competitive Landscape
The global platelet-rich plasma market is highly competitive, with strong participation from Johnson & Johnson, Arthrex, Inc., EmCyte Corporation, Dr PRP USA LLC, and Juventix Regenerative Medical, LLC. These players leverage strong clinical networks, physician training programs, and innovation in centrifugation systems, closed-loop preparation kits, and sterile processing technologies to support diverse regenerative applications.
Rising demand for minimally invasive therapies, orthopedic and aesthetic procedures, and faster tissue healing is accelerating innovation, with manufacturers emphasizing automated PRP systems, standardized protocols, digital workflow integration, and expansion into emerging healthcare markets to enable scalable and consistent biologic treatment outcomes.
Key Industry Developments:
- In October 2025, Regenerative Medical Technologies Group, Inc. expanded its CELLGENIC brand into the Dominican Republic through Global Stem Cells Group, increasing training and certification programs in regenerative medicine. As PRP therapies are commonly introduced as entry-level biologic procedures due to safety and accessibility, expanding practitioner education and clinic networks contributes to higher PRP adoption across emerging healthcare markets.
- In September 2025, CollPlant Biotechnologies expanded its European distribution network for Vergenix™ STR in Poland, a tendon-repair solution aligned with orthopedic regenerative treatments. Since PRP is widely used alongside collagen-based biologics to enhance tendon healing and tissue regeneration, the expansion supports increased adoption of orthobiologic treatment protocols, indirectly strengthening PRP procedure demand within sports medicine and musculoskeletal care.
Companies Covered in Platelet Rich Plasma Market
- Johnson & Johnson
- Arthrex, Inc.
- EmCyte Corporation
- Dr PRP USA LLC
- Juventix Regenerative Medical, LLC
- Terumo Corporation
- Zimmer Biomet
- Stryker
- Apex Biologix
- Celling Biosciences, Inc.
- Regen Lab SA
- Exactech, Inc.
- Isto Biologics
- Royal Biologics
- Nuo Therapeutics, Inc.
- Others
Frequently Asked Questions
The global platelet rich plasma market is projected to be valued at US$ 518.6 Mn in 2026.
Rising demand for minimally invasive regenerative therapies, increasing sports and orthopedic injuries, aging population, and growing adoption of PRP in cosmetic and musculoskeletal treatments are primarily driving the global platelet-rich plasma market.
The global platelet rich plasma market is poised to witness a CAGR of 5.5% between 2026 and 2033.
Expansion into aesthetic medicine, integration with regenerative therapies (e.g., stem-cell combinations), technological advancements in PRP preparation systems, and growing adoption across emerging healthcare markets create major growth opportunities globally.
Johnson & Johnson, Arthrex, Inc., EmCyte Corporation, Dr PRP USA LLC, and Juventix Regenerative Medical, LLC are some of the key players in the platelet rich plasma market.








