ID: PMRREP5441| 190 Pages | 9 Apr 2025 | Format: PDF, Excel, PPT* | Healthcare

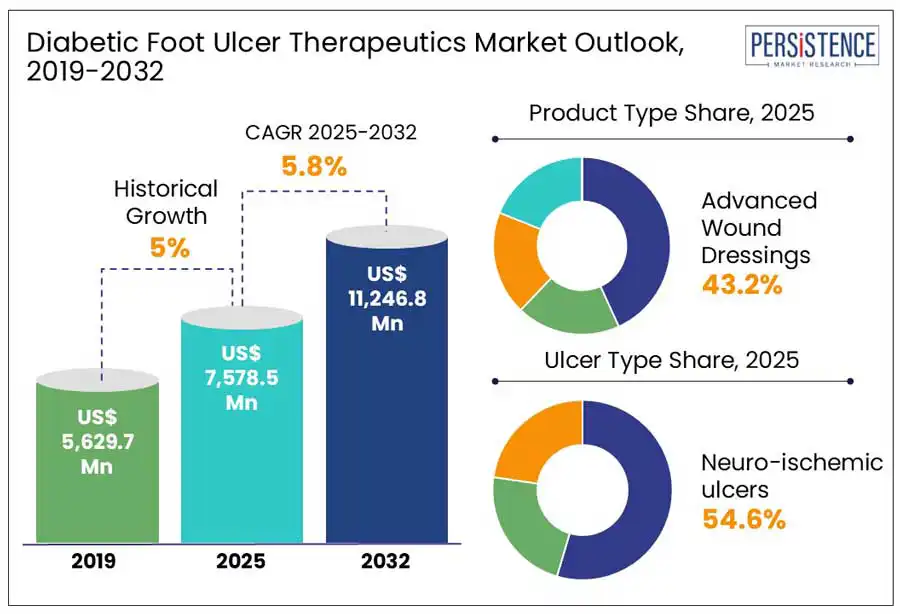
The global diabetic foot ulcer therapeutics market size is projected to rise from US$ 7,578.5 Mn in 2025 to US$ 11,246.8 Mn by 2032. It is anticipated to exhibit a CAGR of 5.8% during the forecast period from 2025 to 2032.
Diabetic Foot Ulcer (DFU) has become an important public health concern, leading to a lower quality of life, high healthcare expenditure, and long hospital stays for patients with diabetes. As per Persistence Market Research, the condition is considered a key complication in those living with type 2 diabetes, affecting around 20% of patients. Increasing global prevalence of diabetes, especially in middle- and low-income countries, has surged the cases of DFUs, bolstering demand for therapeutic interventions.
Key Industry Highlights
|
Global Market Attribute |
Key Insights |
|
Diabetic Foot Ulcer Therapeutics Market Size (2025E) |
US$ 7,578.5 Mn |
|
Market Value Forecast (2032F) |
US$ 11,246.8 Mn |
|
Projected Growth (CAGR 2025 to 2032) |
5.8% |
|
Historical Market Growth (CAGR 2019 to 2024) |
5.0% |
Increasing focus on the reduction of healthcare expenditure related to advanced diabetic foot conditions and limb preservation is projected to boost the global diabetic foot ulcer treatment market. Amputations significantly raise long-term medical costs in addition to causing psychological trauma and physical disability.
The average cost of a significant amputation in the U.S. can surpass US$ 70,000. Management of a diabetic foot ulcer with innovative wound care methods substantially reduces complications and costs. Hence, healthcare institutions and payers are constantly investing in the early-stage therapeutic interventions such as antimicrobial dressings, negative pressure wound therapy (NPWT), and bioengineered skin grafts.
A key factor estimated to limit the adoption of diabetic foot ulcer therapeutic strategies is patient non-compliance. Due to lifestyle restrictions or the lack of knowledge, diabetic patients often do not act in accordance with daily wound care instructions, follow-up appointments, or comply with offloading protocols.
Total contact casting is considered a highly effective in managing offloading plantar pressure. However, patients tend to discontinue it prematurely as it limits mobility. Regardless of the quality or accessibility of therapeutic interventions, this behavioral aspect is anticipated to lower their efficacy.
The ongoing development and commercialization of cell-based therapies and biologics to enhance wound regeneration in non-healing or complex ulcers is anticipated to create a new avenue. Treatments, including recombinant growth factors, mesenchymal stem cells, and Platelet-Rich Plasma (PRP) are showcasing promising results in clinical trials.
Increasing regulatory approval for regenerative medicine is another key factor anticipated to create opportunities for companies to launch new biologic therapies in the market. Anika Therapeutics, for instance, extends its presence in regenerative medicine by developing injectable hyaluronic acid-based therapies. These are extensively studied for wound healing applications such as diabetic ulcers by improving tissue regeneration and extracellular matrix formation.
Smart footwear and accessories are anticipated to gain traction in the diabetic foot ulcer therapeutics market in the foreseeable future. Such specialized footwear and accessories not only help with post-ulcer healing but also prevent ulcers from recurring or forming in high-risk patients. Key companies such as Siren Care have already created smart socks equipped with microsensors to constantly evaluate foot temperature in various zones.
High localized foot temperature is considered an early sign of tissue breakdown or inflammation, a precursor to ulcer development. The data gathered through smart socks is shared to a smartphone app, enabling healthcare professionals and patients to take immediate action. In-depth studies associated with similar smart footwear and accessories are projected to encourage companies to come up with new products for quick healing.
In terms of product type, the global market is divided into advanced wound dressings, skin substitutes, negative pressure wound therapy devices, and growth factors. Out of these, advanced wound dressings are estimated to account for a diabetic foot ulcer therapeutics market share of 43.2% in 2025.
It is attributed to their ability to smoothly manage non-healing, complex wounds, which are common among those living with diabetes. By encouraging angiogenesis and cellular migration, advanced dressings create a moist wound environment that speeds up healing.
Negative pressure wound therapy devices are projected to show a decent growth rate in the forecast period. Demand is anticipated to rise as these help lower the chances of amputations and the frequency of infections. These also help prevent external contamination by creating a sealed wound environment.
Based on ulcer type, the global market is trifurcated into neuropathic ulcers, ischemic ulcers, and neuro-ischemic ulcers. Among these, the neuro-ischemic ulcers segment will likely generate a share of around 54.6% in 2025.
Neuro-ischemic ulcers pose several risks such as amputation, infection, and even death. According to studies, neuro-ischemia is common in about 50% of diabetic population, but neuropathic ulcers affect nearly 35% of all ulcers. To prevent high fatality and infection rates, the need for therapeutic interventions is projected to rise among patients suffering from neuro-ischemic ulcers.
Neuropathic ulcers, on the other hand is set to exhibit a decent growth through 2032 due to increasing prevalence of peripheral neuropathy among diabetic individuals. The International Diabetes Federation mentioned that more than 50% of diabetic patients develop some form of peripheral neuropathy.
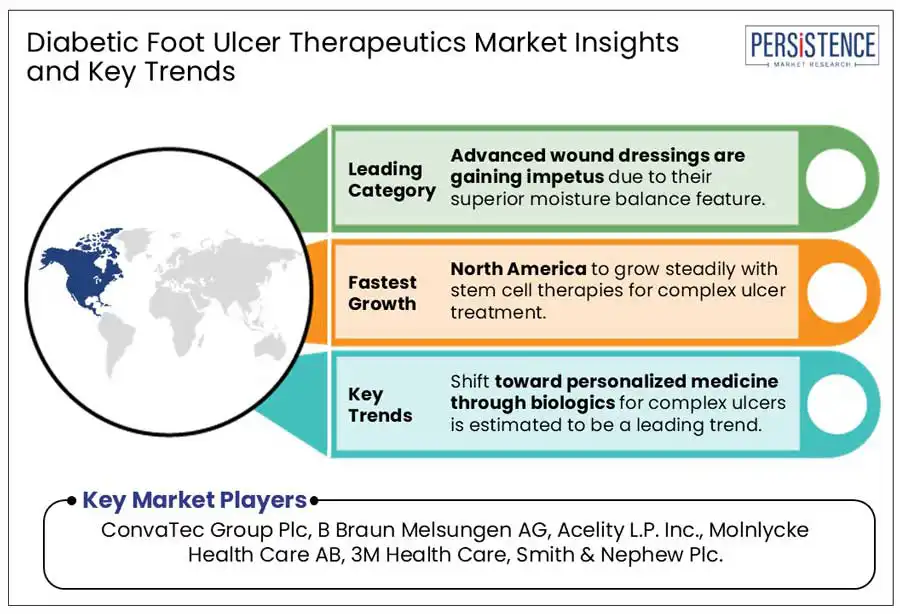
North America is projected to generate a share of 39.4% in 2025. The growth is propelled by rising prevalence of diabetes and the presence of several well-known advanced wound care therapy providers. The U.S. diabetic foot ulcer therapeutics market is anticipated to play a key role in boosting North America’s growth through 2032.
As per the Centers for Disease Control and Prevention (CDC), in the U.S., more than 38 million individuals were living with diabetes in 2021. Among them, nearly 15% were estimated to develop a foot ulcer at some point in their lives. This rising incidence is projected to create a high demand for infection control therapeutics, bioengineered skin substitutes, and wound care products. The country’s higher-than-average rates of amputation and ulcer recurrence are also set to propel the adoption of innovative therapies.
In Asia Pacific, China is considered one of the most prominent countries with a high diabetes prevalence rate. As of 2023, more than 11% of adults in the country are diabetic. The field of DFU therapeutics is evolving in China with both domestically produced and imported products.
Local firms are focusing on offering cost-effective recombinant growth factors such as recombinant human epidermal growth factor (rhEGF). It is being extensively used across hospitals to enhance wound healing.
In India, lack of routine screening and poor glycemic control among diabetic individuals is anticipated to surge the development of foot ulcer complications. The market is at a nascent stage in the country. However, steady growth is estimated due to the adoption of antimicrobial foams and NPWT devices in both private and government hospitals.
Europe is predicted to be spearheaded by Germany as local firms such as Paul Hartmann and Lohmann & Rauscher focus on innovating exudate-control and antimicrobial dressings. The country is also active in terms of clinical trials, mainly those revolving around regenerative therapies and growth factors. Healthcare institutions such as the University Hospital of Cologne, for instance, are constantly conducting trials on tissue-engineered skin grafts, thereby opening the door to new opportunities.
National Health Service (NHS) in the U.K. plays a significant role in DFU management with a strong network of podiatric services. NHS England has also implemented the Diabetic Foot Care Pathways to include advanced wound care and early risk detection in tertiary settings.
The global diabetic foot ulcer market is competitive with the presence of several large- and small-scale companies. They are constantly striving to enhance their offerings by investing huge sums in research and development activities to launch new products.
Supportive government initiatives, product line expansions, partnerships, and acquisitions are a few key strategies of leading companies. Some of the other companies are focusing on gaining fast-track approvals from government bodies to market their products.
The market is projected to be valued at US$ 7,578.5 Mn in 2025.
The market is estimated to rise at a CAGR of 5.8% through 2032.
ConvaTec Group Plc, B Braun Melsungen AG, and Acelity L.P. Inc. are the leading providers.
The global market surged at a CAGR of 5.0% in the historical period.
Advanced wound dressings are the dominant product type in the market.
|
Report Attributes |
Details |
|
Historical Data/Actuals |
2019 - 2024 |
|
Forecast Period |
2025 - 2032 |
|
Market Analysis Units |
Value: US$ Bn/Mn, Volume: As Applicable |
|
Geographical Coverage |
|
|
Segmental Coverage |
|
|
Competitive Analysis |
|
|
Report Highlights |
|
|
Customization and Pricing |
Available upon request |
By Product Type
By Ulcer Type
By End-user
By Region
Delivery Timelines
For more information on this report and its delivery timelines please get in touch with our sales team.
About Author