- Executive Summary
- Global Viral Sensitizers Market Snapshot, 2026 and 2033
- Market Opportunity Assessment, 2026 - 2033, US$ Mn
- Key Market Trends
- Future Market Projections
- Premium Market Insights
- Industry Developments and Key Market Events
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Challenges
- Key Trends
- Macro-Economic Factors
- Global Sectorial Outlook
- Global GDP Growth Outlook
- COVID-19 Impact Analysis
- Forecast Factors - Relevance and Impact
- Value Added Insights
- Regulatory Landscape
- Product Adoption Analysis
- Value Chain Analysis
- Key Deals and Mergers
- PESTLE Analysis
- Porter’s Five Force Analysis
- Global Viral Sensitizers Market Outlook:
- Key Highlights
- Market Size (US$ Mn) and Y-o-Y Growth
- Absolute $ Opportunity
- Market Size (US$ Mn) Analysis and Forecast
- Historical Market Size (US$ Mn) Analysis, 2020-2025
- Market Size (US$ Mn) Analysis and Forecast, 2026-2033
- Global Viral Sensitizers Market Outlook: Product Type
- Introduction / Key Findings
- Historical Market Size (US$ Mn) Analysis, By Product Type, 2020 - 2025
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Attractiveness Analysis: Product Type
- Global Viral Sensitizers Market Outlook: Application
- Introduction / Key Findings
- Historical Market Size (US$ Mn) Analysis, By Application, 2020 - 2025
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Attractiveness Analysis: Application
- Global Viral Sensitizers Market Outlook: End User
- Introduction / Key Findings
- Historical Market Size (US$ Mn) Analysis, By End User, 2020 - 2025
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis: End User
- Key Highlights
- Global Viral Sensitizers Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Region, 2020 - 2025
- Market Size (US$ Mn) Analysis and Forecast, By Region, 2026 - 2033
- North America
- Europe
- East Asia
- South Asia and Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Viral Sensitizers Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2020 - 2025
- By Country
- By Product Type
- By Application
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2026 - 2033
- U.S.
- Canada
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis
- Europe Viral Sensitizers Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2020 - 2025
- By Country
- By Product Type
- By Application
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2026 - 2033
- Germany
- France
- U.K.
- Italy
- Spain
- Russia
- Rest of Europe
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis
- East Asia Viral Sensitizers Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2020 - 2025
- By Country
- By Product Type
- By Application
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2026 - 2033
- China
- Japan
- South Korea
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis
- South Asia & Oceania Viral Sensitizers Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2020 - 2025
- By Country
- By Product Type
- By Application
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2026 - 2033
- India
- Southeast Asia
- ANZ
- Rest of South Asia & Oceania
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis
- Latin America Viral Sensitizers Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2020 - 2025
- By Country
- By Product Type
- By Application
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2026 - 2033
- Brazil
- Mexico
- Rest of Latin America
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis
- Middle East & Africa Viral Sensitizers Market Outlook:
- Key Highlights
- Historical Market Size (US$ Mn) Analysis, By Market, 2020 - 2025
- By Country
- By Product Type
- By Application
- By End User
- Market Size (US$ Mn) Analysis and Forecast, By Country, 2026 - 2033
- GCC Countries
- Egypt
- South Africa
- Northern Africa
- Rest of Middle East & Africa
- Market Size (US$ Mn) Analysis and Forecast, By Product Type, 2026 - 2033
- Synthetic Viral Sensitizers
- Natural Viral Sensitizers
- Market Size (US$ Mn) Analysis and Forecast, By Application, 2026 - 2033
- Antiviral Drug Development
- Vaccine Development
- Oncolytic Viral Therapies
- Others
- Market Size (US$ Mn) Analysis and Forecast, By End User, 2026 - 2033
- Pharmaceutical Companies
- Research Institutes
- Biotechnology Companies
- Others
- Market Attractiveness Analysis
- Competition Landscape
- Market Share Analysis, 2025
- Market Structure
- Competition Intensity Mapping By Market
- Competition Dashboard
- Company Profiles (Details - Overview, Financials, Strategy, Recent Developments)
- Moderna
- Overview
- Segments and Products
- Key Financials
- Market Developments
- Market Strategy
- Pfizer
- AbbVie
- Bluebird Bio
- Merck & Co.
- Iovance Biotherapeutics
- Bristol-Myers Squibb
- Cellectis
- Roche
- AstraZeneca
- Johnson & Johnson
- Alnylam Pharmaceuticals
- Regeneron Pharmaceuticals
- Gilead Sciences
- Others
- Moderna
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations
- Pharmaceuticals
- Viral Sensitizers Market
Viral Sensitizers Market Size, Share, Growth, and Regional Forecast, 2026 - 2033
Viral Sensitizers Market by Product Type (Synthetic Viral Sensitizers, Natural Viral Sensitizers), Application (Antiviral Drug Development, Vaccine Development, Oncolytic Viral Therapies, Others), End-user (Pharmaceutical Companies, Research Institutes, Biotechnology Companies, Others), and Regional Analysis from 2026 - 2033
Key Industry Highlights
- Dominant Segment: Synthetic Viral Sensitizers are increasingly important due to engineered design, reproducibility, and controllable activity, with 64.1% share in 2025. Optimized for specific viral pathways and targeted immune modulation, they enable precise antiviral therapies. Scalable production, integration with digital monitoring, and AI-driven optimization support consistent, effective treatments, enhancing applications in personalized medicine, immunotherapies, and next-generation antiviral strategies.
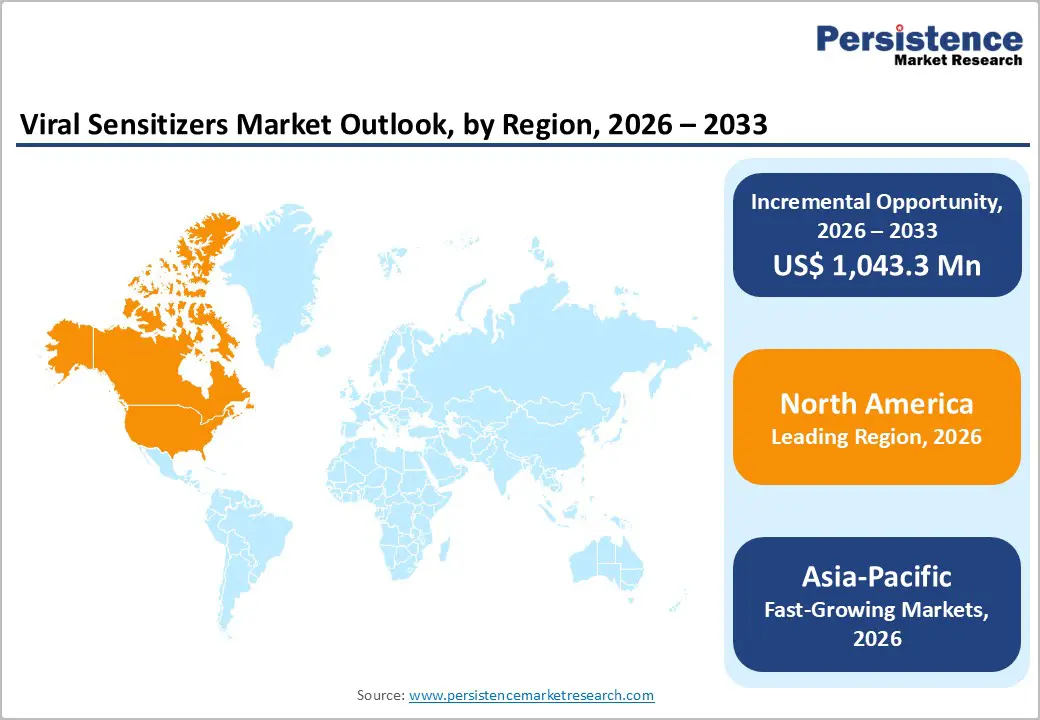
- Dominant Region: North America leads with approximately 39.8% share in 2025, supported by advanced biotech infrastructure, strong regulatory oversight, and early adoption of antiviral and immunomodulatory therapies. Asia Pacific is the fastest-growing region, fueled by expanding healthcare and biotech facilities, government initiatives, rising R&D investment, and scalable manufacturing adoption for novel viral sensitizers.
- Growth Indicators: Rising demand for enhanced antiviral treatments, increasing biopharmaceutical investments, technological advances in viral modulation, and the growing need for personalized and effective therapies propel growth.
- Opportunity: Opportunities include next-generation viral sensitizers for rare and infectious diseases, scalable manufacturing platforms, AI-enabled therapy optimization, integration with personalized medicine pipelines, and expansion in emerging biotech regions.
| Key Insights | Details |
|---|---|
|
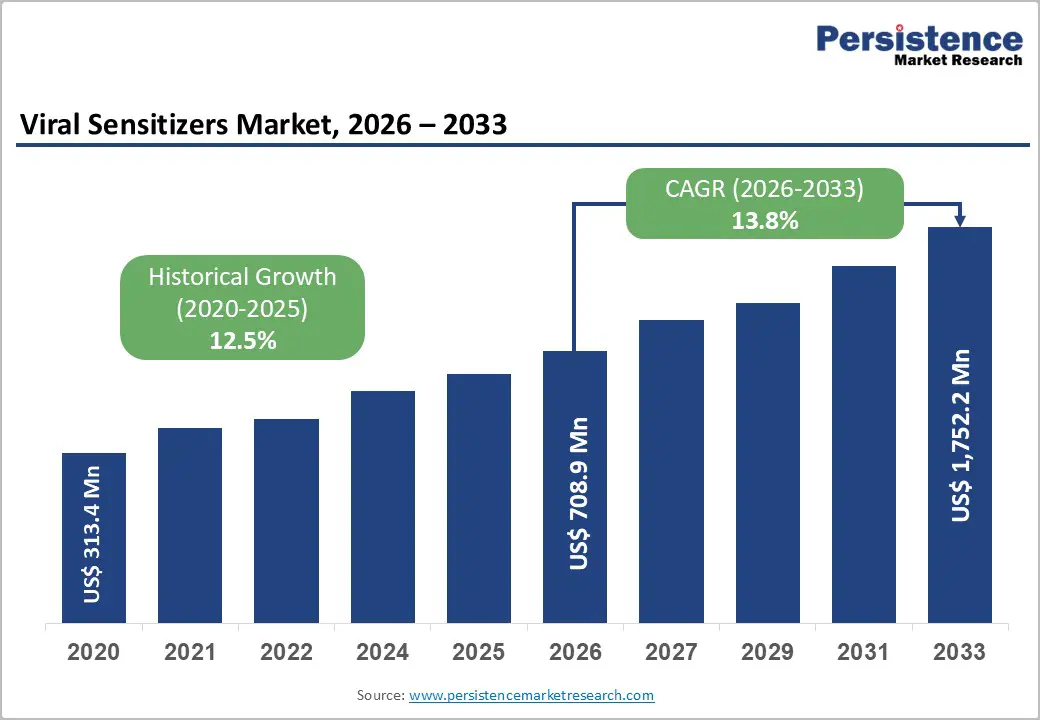
Global Viral Sensitizers Market Size (2026E) |
US$ 708.9 Mn |
|
Market Value Forecast (2033F) |
US$ 1,752.2 Mn |
|
Projected Growth (CAGR 2026 to 2033) |
13.8% |
|
Historical Market Growth (CAGR 2020 to 2025) |
12.5% |
Market Dynamics
Driver: Rising demand for antiviral and immunomodulatory therapies
The global burden of viral diseases remains high, sustaining demand for antiviral and immune-modulating treatments that often rely on viral sensitizers to improve therapeutic efficacy. According to the World Health Organization (WHO), nearly 39 million people were living with HIV in 2022, while hundreds of millions are chronically infected with hepatitis B or C, creating a persistent need for advanced therapies. Viral infections also contribute significantly to annual morbidity and mortality worldwide, with seasonal influenza alone causing 3–5 million severe cases each year, per WHO estimates.
Moreover, public health initiatives have expanded access to antivirals and improved treatment adherence, thereby increasing the use of adjunct technologies that enhance drug performance, including immunomodulatory sensitizers. Growth in antiviral prescriptions and clinical trials further underscores expanded therapeutic demand, reinforcing the role of sensitizers in supporting targeted immune responses and improving clinical outcomes.
Restraints: High cost of advanced viral sensitizers and production platforms
Manufacturing complex biologic and viral-targeted agents, which often incorporate multiple viral sensitizers, entails substantial costs and technical requirements. The Tufts Center for Drug Development reports the average cost to develop a new therapeutic remains near $897 million, with biologically based products often exceeding this due to sophisticated production and stringent quality demands. Such high R&D and manufacturing expenses must be recouped through pricing, limiting access and adoption, particularly in resource-limited settings.
In addition, specialized viral vector and cell-based manufacturing platforms are expensive: a typical viral vector gene therapy manufacturing run can cost close to $100 000 per dose at current scales, according to pharmaceutical engineering analyses, largely due to raw material inputs and low yields. These cost burdens constrain clinical adoption of advanced sensitizers and can deter smaller developers from investing in novel platforms.
Opportunity: Development of synthetic and next-generation viral sensitizers
The evolution of synthetic and next-generation viral sensitizers presents a strong opportunity to meet clinical and manufacturing needs more efficiently. Advances in biologics and platform technologies, such as automated continuous manufacturing, are expanding capabilities to produce complex agents with greater consistency and scalability, addressing historic bottlenecks in biologic outputs. Continued innovation supported by increased R&D expenditure in virology and immunology enhances prospects for tailored sensitizers with improved safety and specificity.
Furthermore, digital tools and computational biology are enabling rational design of sensitizers that target viral pathways more effectively, with AI-aided optimization reducing experimental cycles and potentially lowering development timelines. These advances enable broader integration of synthetic sensitizers into personalized antiviral regimens and combination therapies across infectious and immune-mediated diseases.
Category-wise Analysis
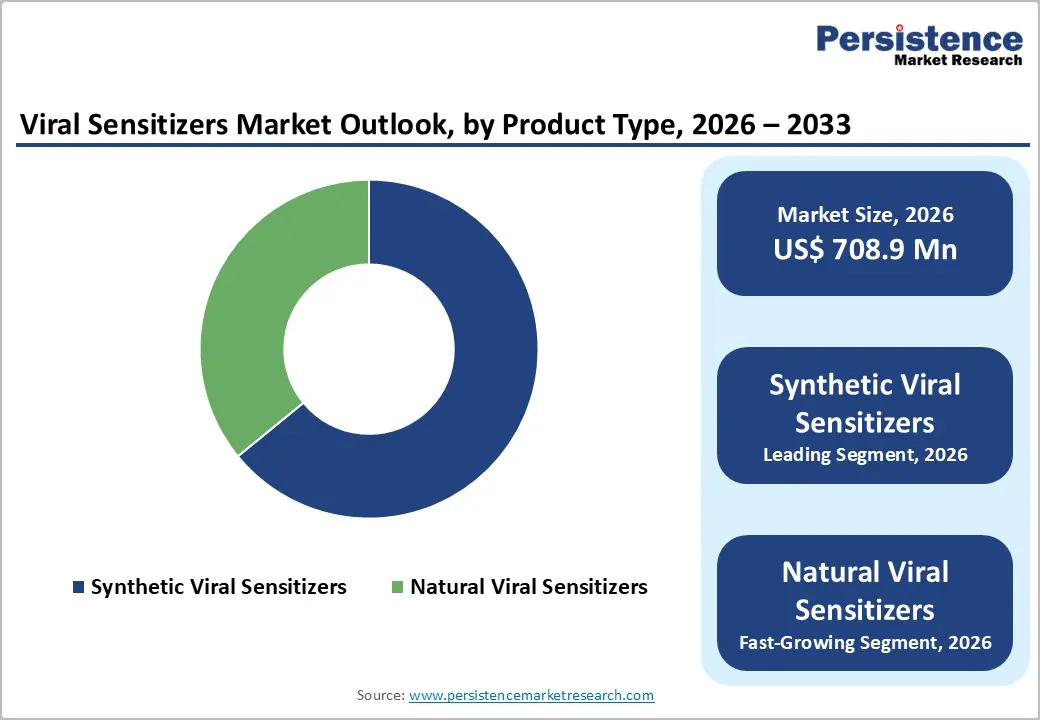
By Product Type Insights
Synthetic viral sensitizers occupies 64.1% share of the global market in 2025, because they offer engineered precision, reproducibility, and scalability compared with naturally derived alternatives. These sensitizers can be tailored for specific viral pathways, targeted immune modulation, and reduced off-target effects, improving therapeutic efficacy and safety. Their production is compatible with advanced biomanufacturing platforms, allowing consistent quality, large-scale output, and compliance with stringent regulatory standards. Furthermore, synthetic sensitizers integrate with digital monitoring and AI-driven optimization, enabling real-time assessment of patient response, dosage, and therapy effectiveness. Global adoption is increasing in antiviral therapies, immunomodulation, and personalized medicine, where controlled activity and predictable performance are critical for clinical success and large-scale deployment in hospitals and research facilities.
By Application Insights
Antiviral drug development dominates the market as viral infections remain a significant global health burden, necessitating constant innovation in therapeutics. Viral sensitizers enhance drug performance by improving viral targeting, boosting potency, and modulating immune responses, making them essential in developing effective antiviral agents. WHO reports approximately 254 million people with hepatitis B and 50 million with hepatitis C, while seasonal influenza affects millions annually, highlighting ongoing demand. Sensitizers are critical during preclinical and clinical development for screening, efficacy optimization, and combination therapy strategies. This focus drives pharmaceutical R&D, accelerating novel antiviral discovery and enabling safer, more potent therapies to treat emerging and chronic viral diseases worldwide.
Regional Insights
North America Viral Sensitizers Market Trends
North America dominates the viral sensitizers market with 38.9% share in 2025, due to its advanced biopharmaceutical and healthcare ecosystem, strong R&D investment, and robust regulatory infrastructure. The region contributed roughly 38–40 % of the infectious disease therapeutics market, reflecting deep capabilities in developing and deploying novel antiviral agents and adjunct technologies. Higher healthcare spending and infrastructure enable rapid adoption of innovative modalities that enhance antiviral performance. Government and private funding support extensive clinical trial activity and early adoption of complex therapies, including those using viral sensitizers. The substantial concentration of biotech companies, research institutions, and venture capital further accelerates innovation. This depth of scientific and commercial capability ensures North America retains leadership in emerging viral technologies and therapeutic enhancements.
Europe Viral Sensitizers Market Trends
Europe is an important region for viral sensitizers because of its coordinated public health infrastructure, strong regulatory frameworks, and collaborative healthcare policies. Agencies like the European Centre for Disease Prevention and Control (ECDC) and EU health programs such as EU4Health (€5.1 billion budget for 2021–2027) enhance disease surveillance, preparedness, and therapeutic development. These platforms support scientific collaboration and data sharing across EU member states, strengthening readiness for viral outbreaks and fostering innovation in therapeutics and adjunct technologies. Europe’s integrated health systems facilitate clinical adoption of advanced antiviral treatments and adjunct sensitizing agents within standardized care pathways. The region’s commitment to real-world evidence (RWE) and regulatory harmonization helps streamline the evaluation of novel therapies. This environment ensures Europe remains strategically significant for commercial and clinical deployment of viral sensitization technologies.
Asia Pacific Viral Sensitizers Market Trends
Asia Pacific is the fastest-growing market for viral sensitizers due to expanding healthcare infrastructure, increasing government investment, and rising infectious disease burden across populous countries. The region has experienced dramatic growth in biopharmaceutical innovation and clinical activity, with Asia’s share of the global innovative drug pipeline increasing from 28% to 43% over five years, outpacing both the United States and Europe. Rapid expansion of domestic R&D, manufacturing capacities, and clinical research activities, particularly in China and South Korea drives adoption of advanced therapeutic technologies, including sensitizers. Population factors also contribute: Asia-Pacific remains home to large segments affected by infectious diseases that require ongoing innovations in treatment and supportive technologies. Combined with supportive policies and improving healthcare access, these dynamics fuel the region’s rapid growth trajectory.
Competitive Landscape
Leading viral sensitizers companies focus on advanced antiviral and immunomodulatory technologies, scalable production, and regulatory compliance. Investments target process optimization, AI-driven design, and high-throughput testing to ensure efficacy and consistency. Strategic collaborations with biotech, academia, and regulators, coupled with stringent quality control and integrated supply chains, accelerate global adoption of viral sensitizers in therapeutic and research applications.
Key Industry Developments:
- In September 2025, Tozaro secured Canada-UK research and development funding to advance tools for lentiviral production, in partnership with ViricaBiotech Inc. and the Centre for Process Innovation (CPI). The funding, awarded in 2025, aimed to accelerate development of scalable, high-quality lentiviral manufacturing technologies to support gene and cell therapy applications.
- In May 2025, AbbVie announced a collaboration and license option agreement with ADARx Pharmaceuticals to develop next-generation siRNA therapies across multiple therapeutic areas. The agreement, finalized in 2025, allowed AbbVie to access ADARx’s proprietary RNA-editing and siRNA technology platforms, aiming to advance treatments for genetic, metabolic, and viral diseases.
Companies Covered in Viral Sensitizers Market
- Moderna
- Pfizer
- AbbVie
- Bluebird Bio
- Merck & Co.
- Iovance Biotherapeutics
- Bristol-Myers Squibb
- Cellectis
- Roche
- AstraZeneca
- Johnson & Johnson
- Alnylam Pharmaceuticals
- Regeneron Pharmaceuticals
- Gilead Sciences
- Others
Frequently Asked Questions
The global viral sensitizers market is projected to be valued at US$ 708.9 Mn in 2026.
Rising viral infections, demand for antiviral therapies, technological advances, and expanding healthcare infrastructure drive market growth.
The global viral sensitizers market is poised to witness a CAGR of 13.8% between 2026 and 2033.
Opportunities include synthetic viral sensitizers, next-generation therapies, AI optimization, scalable manufacturing, and emerging biotech markets.













