- Healthcare
- Vaccine Administration Market
Vaccine Administration Market Size, Share, and Growth Forecast, 2026 - 2033
Vaccine Administration Market by Product Type (Syringes, Jet Injectors, Intranasal Devices, Others), Route of Administration (Intramuscular, Subcutaneous, Intradermal, Others), Age Group (Pediatric, Adult, Geriatric), and Regional Analysis for 2026 - 2033
Vaccine Administration Market Share and Trends Analysis
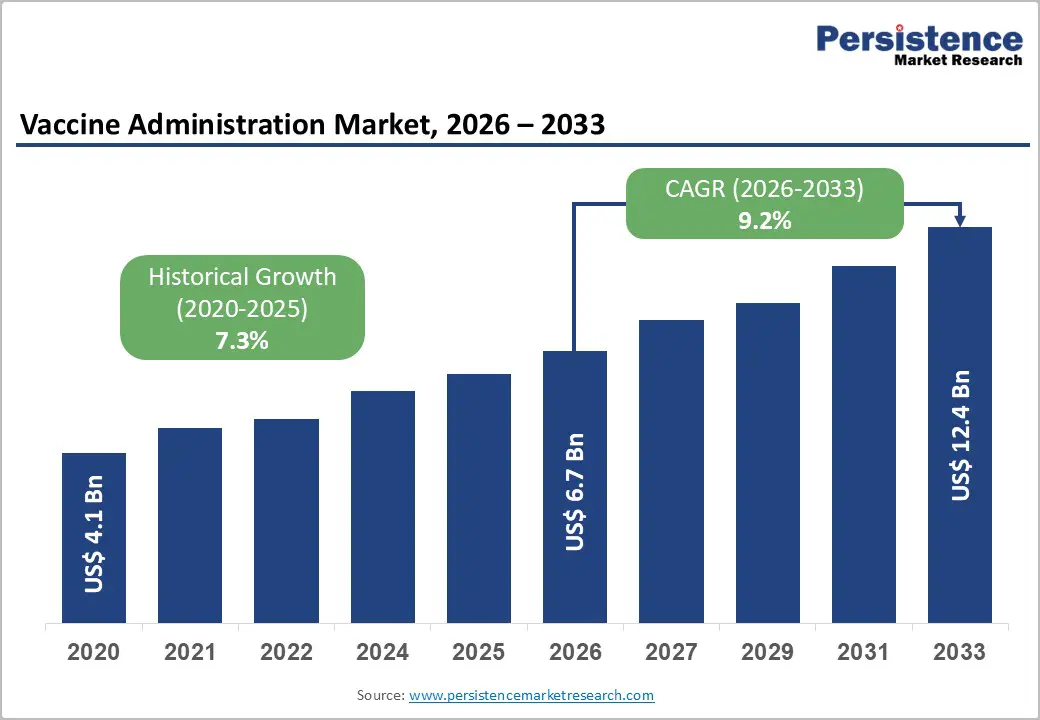
The global vaccine administration market size is likely to be valued at US$ 6.7 billion in 2026, and is projected to reach US$ 12.4 billion by 2033, growing at a CAGR of 9.2% during the forecast period 2026−2033. Sustained immunization demand, demographic transition, and healthcare system modernization position vaccine administration as a strategic segment of healthcare delivery with resilient, long-term growth prospects. Rising birth cohorts in emerging economies and expanding geriatric populations across developed regions increase routine and booster immunization volumes, directly elevating demand for safe and efficient administration devices. International health agencies, including the World Health Organization (WHO) and national immunization technical advisory groups, continue to reinforce vaccination schedules, strengthening clinical awareness and adherence.
Technology integration into delivery systems improves dose accuracy, reduces administration errors, and enhances infection control compliance, encouraging provider adoption across hospitals, retail pharmacies, and community clinics. Expansion of universal immunization programs and adult vaccination initiatives drives procurement through public tenders and private healthcare networks. Digital health records and traceability systems further support large-scale immunization tracking, improving operational efficiency.
Key Industry Highlights
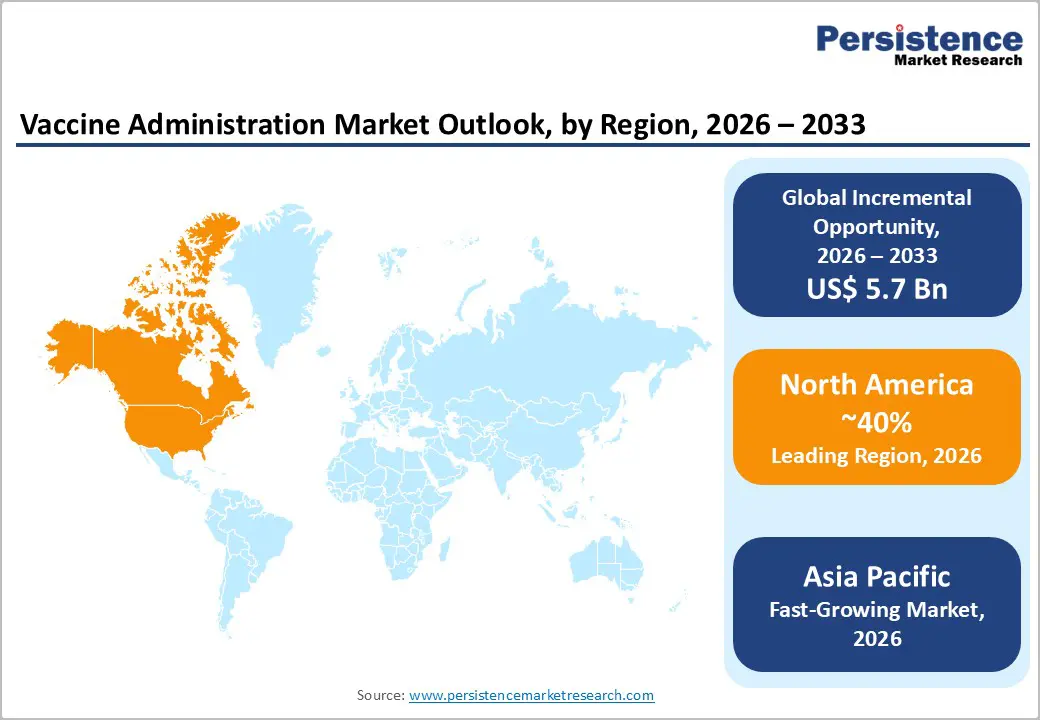
- Dominant Region: North America is expected to exceed 40% share by 2026, driven by strong immunization infrastructure in the United States and Canada.
- Fastest-growing Regional Market: Asia Pacific is projected to be the fastest-growing market between 2026 and 2033, propelled by large population demand and expanding immunization infrastructure.
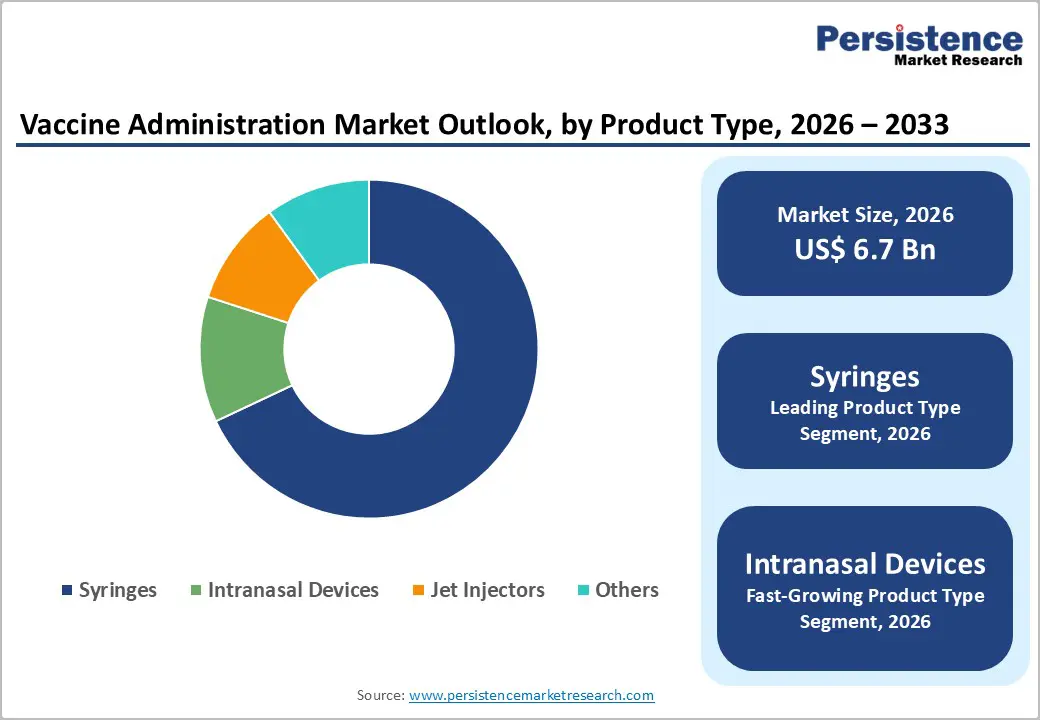
- Leading Product Type: Syringes are likely to hold nearly 68% share in 2026, owing to clinical standardization, safety designs, and large-scale procurement.
- Fastest-growing Product Type: Intranasal devices are projected as the fastest-growing segment from 2026 to 2033, driven by needle-free adoption and improved patient compliance.
- February 2026: The WHO Southeast Asia Regional Office held a virtual consultation to validate a draft immunization and vaccine research taxonomy framework.
| Key Insights | Details |
|---|---|
|
Vaccine Administration Market Size (2026E) |
US$ 6.7 Bn |
|
Market Value Forecast (2033F) |
US$ 12.4 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
9.2% |
|
Historical Market Growth (CAGR 2020 to 2025) |
8.5% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Expansion of National Immunization Programs and Adult Vaccination Policies
National immunization initiatives and clear adult vaccination policies strengthen vaccination infrastructure and delivery systems at scale by aligning public health objectives, funding, and service delivery across government agencies, state programs, and health care providers. The U.S. Vaccines National Strategic Plan 2021–2025 from the Department of Health and Human Services establishes a lifespan approach to vaccination, including immunization goals and measurable coverage targets that guide coordinated action across public and private sectors to increase access and use of recommended vaccines. This strategic focus ensures resources are allocated to vaccine access, provider engagement, and public education, which in turn improves the capacity to deliver vaccines and maintain high uptake rates across different age groups.
On the adult front, explicit policy support influences demand and coverage among populations that have traditionally shown lower vaccine uptake relative to children. Coverage tracking by the U.S. National Immunization Survey for the 2024–2025 season indicates COVID-19 vaccination coverage reached 44% among adults aged 65 years and older and 23% among adults aged 18 years and older, reflecting the impact of targeted adult immunization guidance and outreach efforts that prioritize high-risk groups and older age cohorts. These policies create structured schedules for adult vaccines, encourage health systems to stock and administer recommended immunizations, and enable insurers to cover preventive services.
Advancements in Vaccine Delivery Technologies
Government investment in innovative delivery platforms has materially reshaped immunization dynamics by enhancing accessibility, tolerability, and operational efficiency. The U.S. Centers for Disease Control and Prevention (CDC) reported that its collaboration on a novel microneedle patch for measles-rubella vaccines produced the first clinical data demonstrating safe and effective use in children, signaling the potential to reduce procedural barriers associated with traditional injections; this trial outcome is a tangible indicator of how technology changes can improve uptake and adherence in routine programs through reduced discomfort and simplified administration. Public sector endorsement of such technologies drives confidence among healthcare providers and payers, encouraging procurement and integration into national immunization schedules, which expands reach into underserved populations and bolsters overall coverage rates.
The business impact of delivery innovations extends beyond patient experience. Enhanced devices such as patches, micro-delivery systems and smart syringes streamline logistics by lowering reliance on highly specialized staff and reducing training costs while maintaining regulatory compliance. Real-time tracking and monitoring features embedded in next-generation delivery systems support inventory management and cold chain oversight by providing actionable data that aligns with governmental reporting standards. This operational visibility strengthens supply reliability and mitigates wastage, lowering the total cost of service delivery and improving return on investment for public health programs and private vaccination initiatives alike.
Training and Technical Complexity
Operational execution is constrained by the depth and breadth of training requirements for personnel administering injections, managing storage protocols and conducting patient assessments. The CDC stipulates that health care personnel must complete comprehensive, competency-based training prior to administering vaccines to ensure correct screening, route and site selection, handling procedures and documentation. This requirement increases staffing lead time, compliance oversight and internal audit obligations. As of 2025, the CDC’s Recommended Immunization Schedules include routine vaccines for children, adolescents and adults across multiple age groups, reflecting a complex matrix of dosing, timing and risk-based guidance that must be mastered by clinical teams.
Technical complexity intensifies when evolving clinical guidelines, electronic immunization information systems and cold chain validation standards require continuous workforce upskilling. CDC training frameworks for 2025 outline structured education modules and competency verification processes for clinicians and immunization providers, reinforcing formal qualification pathways within public health programs. Certification tracking, periodic retraining and proficiency validation extend time to deployment for newly hired personnel and reduce scheduling flexibility during peak service cycles.
Cold-Chain Infrastructure Gaps and Logistical Complexities
Gaps in cold-chain infrastructure and logistical complexity impose material barriers to effective vaccination programs because most vaccines require strict temperature control from manufacturing until the moment of administration. Cold supply-chain systems must maintain temperatures typically between 2 °C and 8 °C, or much lower for certain products, through multiple handoffs, storage points, transport modes, and geographic environments. Any deviation outside these ranges can permanently degrade potency, rendering doses unusable in real-world delivery settings. Supply chain guidance from the World Health Organization underscores the need for functional, end-to-end logistics systems designed to protect vaccine quality and ensure uninterrupted availability of product at service points, while requiring significant investment in specialized equipment, continuous monitoring technologies, and trained personnel to manage variability in handling and transit conditions.
According to WHO immunization supply chain resources, significant cold-chain capacity limitations persist, particularly in regions with unreliable electricity, limited storage assets, and complex distribution networks. Insufficient temperature-controlled storage and handling infrastructure increases the risk of spoilage or loss during storage and transport, elevating operational costs and undermining delivery performance. For example, global estimates indicate that temperature-related failures contribute to inefficiencies in immunization efforts, with some analyses estimating up to half of vaccine doses affected by cold chain shortcomings in contexts lacking robust systems.
Strategic Public-Private Partnerships and Collaborations
Public and private sector collaboration amplifies the reach of vaccination efforts by aligning resources, expertise, and infrastructure to address gaps that neither sector can fully manage alone. Formal partnerships between federal agencies and private healthcare providers, pharmacies, and community organizations unlock access to broad networks capable of administering vaccines at scale and in diverse settings. For example, coordinated efforts that trained more than 295,000 community-level spokespersons and enabled administration of over 2.1 million vaccinations through localized outreach channels demonstrate the ability of joint initiatives to activate trusted messengers and service points that extend public health capacity beyond traditional channels.
Partnerships also serve as mechanisms to leverage private sector technological innovation, logistical systems, and workforce capabilities in support of public health objectives. By engaging network operators with existing clinical infrastructure and data systems, government agencies can improve service delivery efficiency and responsiveness during routine immunization campaigns and in response to emergent health threats. Public-private collaborations reduce duplication of effort, enhance capacity for rapid scale-up, and support information sharing that improves vaccine access and uptake in real time.
Digital Integration and Data-Driven Vaccine Management Tools
Integration of digital systems and data-centric vaccine management drives operational effectiveness and strategic planning. Integrated Immunization Information Systems (IIS) centralize individual and population vaccination data into a single, secure platform accessible by healthcare providers and public health authorities, eliminating fragmented record keeping and reducing redundant vaccinations. These systems empower clinicians with consolidated patient histories at the point of care, support automated scheduling and reminders through data exchange, and enhance surveillance by accurately tracking coverage and identifying under-served populations.
Real-time data exchange substantially enhances public health responsiveness and resource allocation. Systems such as the CDC’s Vaccine Tracking System (VTrckS) provide near real-time visibility into vaccine orders, shipments, and inventories across multiple levels of the healthcare supply chain, reducing administrative workload and improving order accuracy for providers. High-quality, timely data supports evidence-driven decisions for outbreak response and logistics planning, contributing to resilience during public health emergencies.
Category-wise Analysis
Product Type Insights
Syringes are anticipated to secure around 68% of the vaccine administration market revenue share in 2026, reflecting widespread clinical acceptance, standardized regulatory approval pathways, and compatibility with multiple vaccine formulations. Healthcare providers prioritize syringes due to procedural familiarity and established training protocols. Safety-engineered variants reduce occupational risk and align with infection prevention guidelines issued by public health authorities. Bulk procurement through national immunization programs supports high-volume manufacturing efficiencies. Accessibility across hospital, clinic, and outreach settings strengthens consistent demand.
Intranasal devices are expected to be the fastest-growing segment during the 2026-2033 forecast period, propelled by increasing acceptance of needle-free immunization approaches and enhanced patient compliance. Pediatric and adult populations with needle aversion demonstrate higher willingness to adopt non-invasive options. Mass immunization campaigns benefit from simplified administration processes requiring minimal clinical infrastructure. Ongoing research collaborations between biologics developers and device innovators support formulation compatibility. Retail pharmacy vaccination models further facilitate expansion of intranasal delivery platforms.
Route of Administration Insights
Intramuscular extracts are poised to dominate with a forecasted market share of 60% in 2026, powered by established clinical protocols, broad vaccine compatibility, and extensive provider training. National immunization frameworks across developed and emerging economies align with intramuscular administration for pediatric, adolescent, and adult immunization programs, reinforcing consistent utilization patterns. Regulatory approvals and product labeling for a majority of routine vaccines specify intramuscular delivery, strengthening institutional preference. Supply chains are optimized for syringes, safety needles, and auto-disable devices designed for this route, supporting procurement efficiency. Clinical guidelines emphasize standardized dosing depth and anatomical site selection, promoting uniformity, safety, and predictable post-vaccination monitoring across hospital networks and community health settings.
Intradermal is estimated to be the fastest-growing segment from 2026 to 2033, fueled by dose-sparing strategies, technological refinement in microinjection systems, and expanding research in targeted immune response stimulation. Advancements in precision-engineered delivery platforms enhance administration accuracy and reproducibility, increasing confidence among healthcare professionals. Policy discussions within public health agencies highlight antigen optimization strategies to extend vaccine supply during high-demand periods. Academic institutions and biotechnology firms continue evaluating dermal immunology pathways to strengthen efficacy outcomes. Procurement teams in specialty clinics and ambulatory centers assess compact, user-friendly intradermal devices that align with workflow efficiency objectives, supporting gradual integration into preventive care programs and travel immunization services.
Regional Insights
North America Vaccine Administration Market Trends
North America market is estimated to capture an approximate 40% of the vaccine administration market share by 2026, supported by advanced delivery infrastructure and high throughput across public and private channels. The U.S. is the largest contributor, driven by structured immunization governance under the Centers for Disease Control and Prevention, which standardizes national vaccine schedules and funds large-scale access programs such as Vaccines for Children. Broad retail pharmacy participation from chains such as CVS Health and Walgreens expands administration points beyond hospitals and physician offices, increasing accessibility and throughput. Canada strengthens overall share through provincially coordinated immunization registries and publicly funded vaccination programs that maintain high pediatric and adult coverage rates.
A structural advantage emerges from coordinated procurement and regulatory alignment across United States and Canada, enabling rapid deployment of newly approved vaccines into established delivery channels. Centralized federal purchasing contracts in the United States streamline distribution to state health departments, while Canadian provincial bulk purchasing agreements stabilize supply allocation. Advanced cold-chain logistics networks and temperature-monitoring technologies sustain product integrity across urban and rural facilities. Workforce standardization programs ensure trained pharmacists, nurses, and physician assistants follow consistent administration protocols, strengthening clinical reliability.
Europe Vaccine Administration Market Trends
Europe demonstrates stable expansion in vaccine administration services, supported by structured universal healthcare systems and coordinated supranational policy frameworks. Harmonized immunization strategies guided by institutions such as the European Centre for Disease Prevention and Control promote standardized vaccination schedules and cross-border surveillance alignment. High baseline immunization coverage across Germany, France, and Italy sustains consistent administration volumes through publicly funded primary care networks. Digital health card systems and interoperable electronic registries enhance dose tracking and minimize duplication across member states. Structured tender-based procurement models improve cost discipline while ensuring predictable supply allocation to hospitals, pharmacies, and community clinics. Strong pharmacovigilance frameworks reinforce public confidence, supporting stable uptake across pediatric and adult immunization categories.
Market performance is further shaped by demographic aging trends and policy emphasis on adult booster programs. Rising elderly population cohorts increase demand for influenza, pneumococcal, and shingles vaccinations delivered through general practitioners and retail pharmacies. Expansion of pharmacist-administered vaccination authority in countries such as the U.K. and Portugal diversifies administration channels and improves accessibility outside traditional hospital settings. Cross-border health data exchange initiatives enhance outbreak preparedness and enable coordinated response strategies. Investment in advanced cold-chain logistics and temperature-monitoring technologies safeguards product integrity across decentralized delivery systems.
Asia Pacific Vaccine Administration Market Trends
Asia Pacific is forecasted to be the fastest-growing market for vaccine administration through 2033, propelled by expanding public immunization mandates, demographic scale, and structural healthcare modernization. Large population bases in India, China, and Indonesia create sustained demand across pediatric, adolescent, and adult vaccination programs, supporting high administration volumes. Governments are integrating digital immunization registries with national health identification systems, improving beneficiary tracking and dose compliance across multi-year schedules. Capital allocation toward primary healthcare centers and district-level cold-chain infrastructure enhances last-mile distribution efficiency. Public funding commitments for universal immunization coverage increase procurement predictability, enabling providers to expand service capacity with lower inventory risk exposure.
Growth acceleration is further supported by policy alignment that prioritizes preventive healthcare within national development agendas. Bulk procurement frameworks across India and China reduce per-dose acquisition costs, improving affordability for large-scale administration programs. Expansion of private hospital chains and corporate healthcare networks broadens access points beyond public facilities, capturing demand from rising middle-income populations seeking adult and travel immunizations. Investment in workforce certification programs enhances competency in advanced delivery techniques, facilitating adoption of intradermal and microinjection platforms.
Competitive Landscape
The global vaccine administration market structure displays moderate concentration, with leading multinational device manufacturers shaping competitive structure through scale, regulatory expertise, and diversified technology platforms. Key players include BD, Terumo Corporation, PharmaJet, West Pharmaceutical Services, Inc., and Nipro Europe Group Companies. These companies maintain diversified portfolios spanning conventional syringes, safety-engineered injection systems, and alternative delivery technologies, enabling alignment with national immunization procurement frameworks and hospital network requirements. Global manufacturing footprints, quality certifications, and regulatory approvals across multiple jurisdictions strengthen contract retention and tender competitiveness.
Competitive differentiation centers on safety innovation, precision engineering, and scalable supply chain integration. Investment in needle-free and intradermal delivery platforms supports expansion into high-efficiency immunization campaigns and specialty clinical segments. Procurement authorities prioritize device reliability, sterility assurance, and compatibility with temperature-controlled logistics, elevating entry barriers for smaller manufacturers. Automation in production facilities and vertical integration of components improve cost discipline and margin stability.
Key Industry Developments
- In September 2025, Pakistan launched a nationwide human papillomavirus (HPV) vaccination campaign targeting girls aged 9–14 to protect them from cervical cancer, joining more than 150 countries that have introduced the WHO-prequalified vaccine into their immunization schedules.
- In August 2025, the Ministry of Health and Family Welfare reported that 11 routine vaccines were administered free to children under the national vaccination campaign, with intensified outreach and strategic interventions deployed to improve immunization coverage across rural and urban districts
- In July 2025, Terumo Corporation announced the commercial launch of its Immucis Intradermal Injection System designed to deliver vaccines and other approved drugs into the dermal layer of skin, offering potential immunogenic advantages and significant antigen dose reduction compared with traditional injection methods.
Companies Covered in Vaccine Administration Market
- BD.
- Terumo Corporation
- PharmaJet.
- West Pharmaceutical Services, Inc.
- Nipro Europe Group Companies
- Hindustan Syringes & Medical Devices Ltd
- Retractable Technologies, Inc.
- Cardinal Health.
Frequently Asked Questions
The global vaccine administration market is projected to reach US$ 6.7 billion in 2026.
Expanding national immunization programs, rising preventive healthcare adoption, technological advancements in delivery systems, and strengthened public health infrastructure investment are driving the market.
The market is poised to witness a CAGR of 9.2% from 2026 to 2033.
Advancement of needle-free and intradermal delivery technologies, integration of digital immunization tracking systems, expansion of adult vaccination programs, and growth in emerging healthcare systems are unlocking high-value market opportunities.
Some of the key market players include BD, Terumo Corporation, PharmaJet, West Pharmaceutical Services, Inc., and Nipro Europe Group Companies.








