
- Biotechnology
- Therapeutic Enzymes Market
Therapeutic Enzymes Market Size, Share, and Growth Forecast, 2026 – 2033
Therapeutic Enzymes Market by Product Type (Asparaginase, Streptokinase, Lipase, Collagenase, Lysozyme, Others), Application (Leukemia, Antiviral Therapy, Stomach Disorders, Skin Ulcers, Others), End-user (Hospitals, Clinics, Research Laboratories, Others), and Regional Analysis for 2026-2033
Therapeutic Enzymes Market Share and Trends Analysis
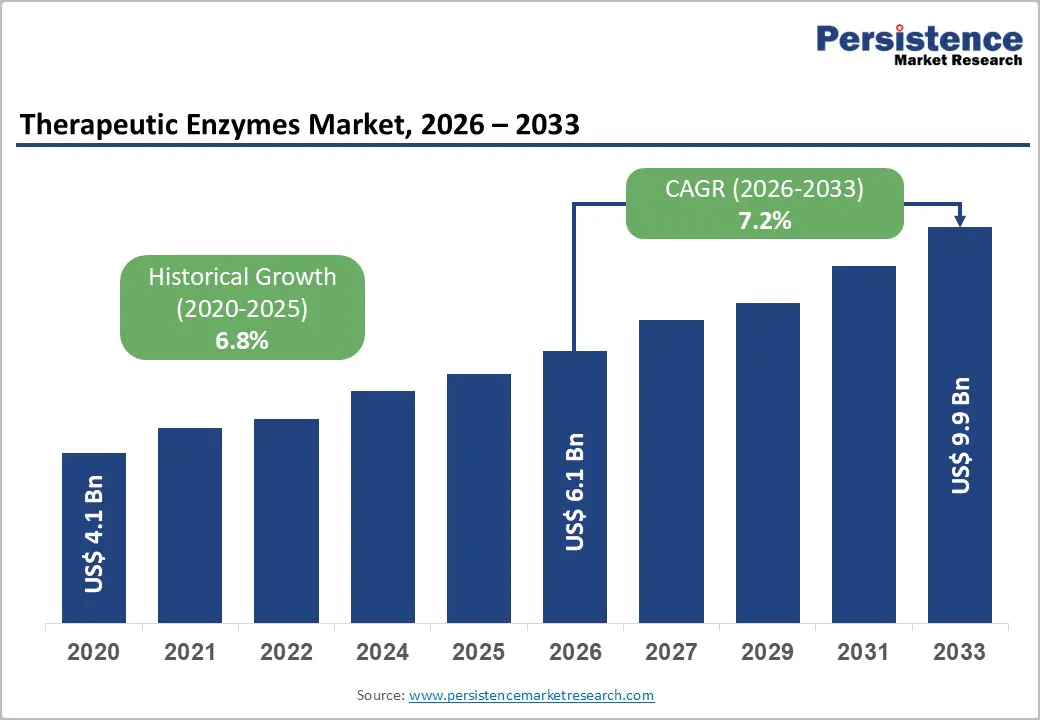
The global therapeutic enzymes market size is likely to be valued at US$ 6.1 billion in 2026, and is projected to reach US$ 9.9 billion by 2033, growing at a CAGR of 7.2% during the forecast period 2026−2033.
Market expansion is driven by increasing clinical adoption of enzyme-based therapies to address chronic and rare conditions. Rising prevalence of hematologic disorders and metabolic deficiencies accelerated demand for enzyme replacement and adjunct therapies. Technological integration in enzyme formulation and delivery platforms enhances bioavailability, efficacy, and patient adherence, reinforcing therapeutic outcomes. Enhanced awareness among healthcare professionals and patients has elevated diagnostic precision and early treatment initiation, improving treatment success and acceptance. Demographic shifts, including an aging population in developed regions, contribute to growing demand for supportive therapies that reduce morbidity and hospitalization. Expanding healthcare infrastructure and investment in specialized care centers facilitate broader access to enzyme therapies, enabling urban and semi-urban patient reach. Strategic collaborations between biotechnology firms, hospitals, and research institutions accelerated pipeline innovation, regulatory approval, and commercialization of novel enzymes.
Key Industry Highlights
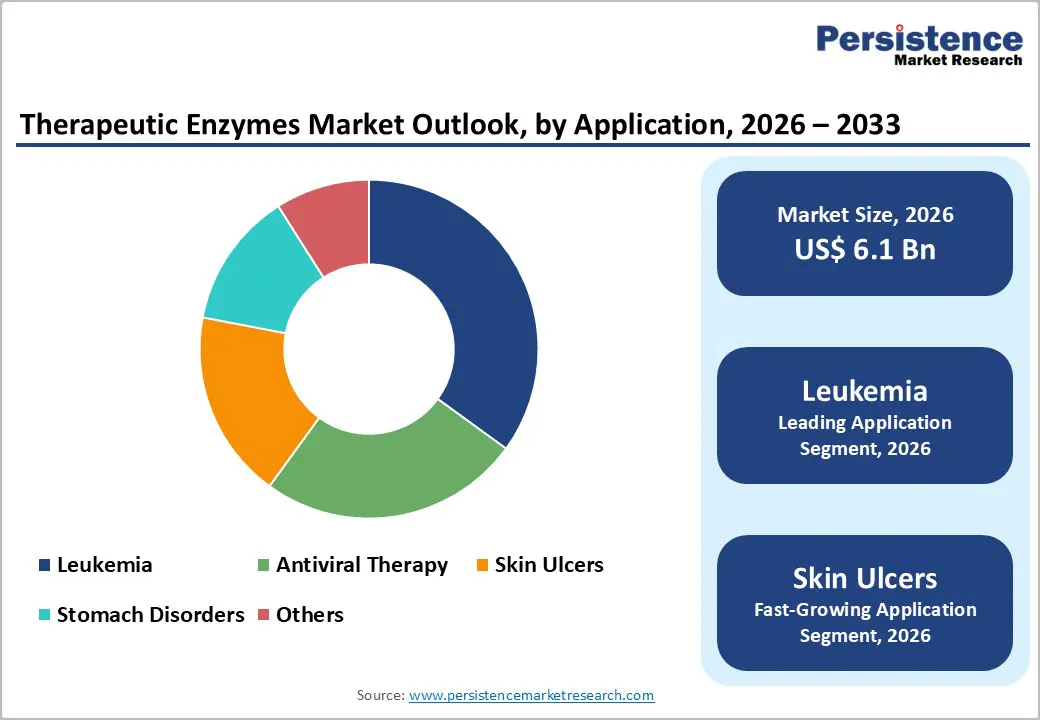
- Leading Application: Leukemia is expected to hold over 35% market share in 2026, driven by enzyme therapy effectiveness, physician familiarity, and insurance support.
- Fastest-growing Application: Skin ulcers are projected to be the fastest-growing between 2026 and 2033, fueled by rising chronic wound prevalence and extensive clinical adoption of enzymatic debridement.
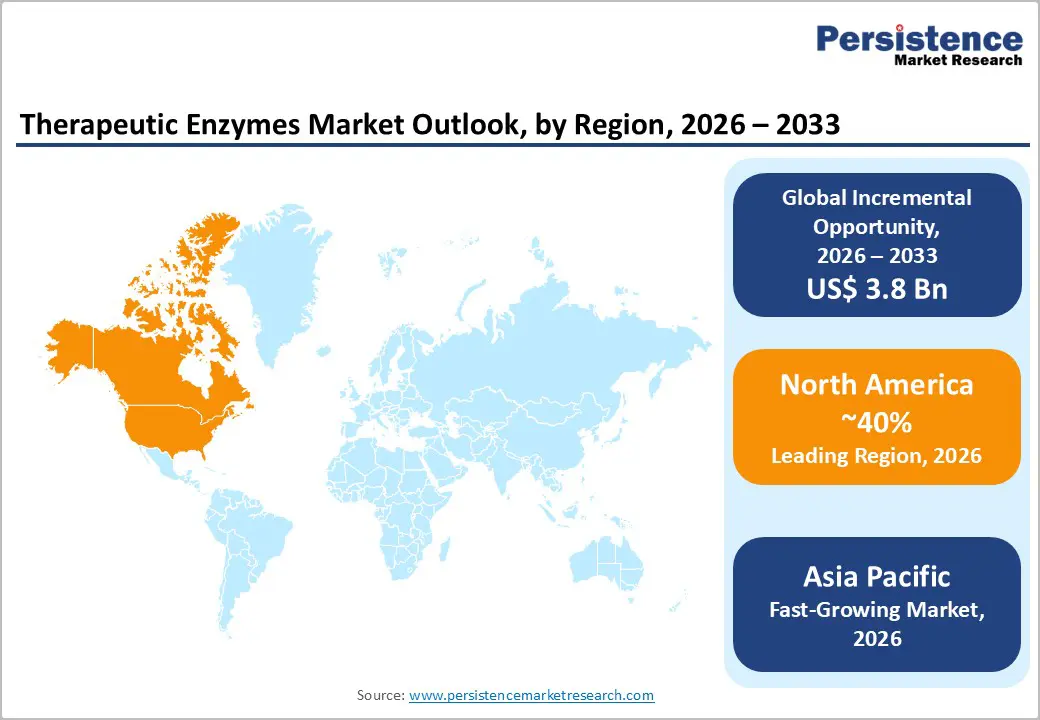
- Dominant Region: North America is projected to hold an estimated 40% market share by 2026, owing to advanced healthcare infrastructure, high disease prevalence, and widening adoption of enzyme therapies.
- Fastest-growing Market: The Asia Pacific market is expected to register the highest growth between 2026 and 2033, powered by rising chronic disease prevalence and improved treatment accessibility.
- February 2026: Researchers at the University of California, Irvine developed a powerful enzyme that can synthesize RNA quickly and accurately, which could accelerate RNA-based research and therapeutic development.
| Key Insights | Details |
|---|---|
|
Therapeutic Enzymes Market Size (2026E) |
US$ 6.1 Bn |
|
Market Value Forecast (2033F) |
US$ 9.9 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
7.2% |
|
Historical Market Growth (CAGR 2020 to 2025) |
6.8% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Rising Prevalence of Metabolic and Hematologic Disorders
The increasing incidence of metabolic and hematologic disorders expanded the pool of patients requiring targeted biochemical therapies, creating sustained clinical demand. Chronic metabolic conditions such as diabetes mellitus disrupt key physiological pathways, necessitating enzyme modulation to restore biochemical balance. Analysis from the World Health Organization (WHO) indicates that around 830 million people globally were living with diabetes as of 2025, with many lacking adequate treatment and early diagnosis, presenting a major chronic health challenge worldwide. Such widespread prevalence reflects a systemic shift in global health burdens, driven by aging populations, lifestyle changes, and rising obesity, all of which elevate metabolic stress on biological systems.
Hematologic disorders, including inherited enzyme deficiency conditions and dysregulated blood coagulation diseases, also contribute to elevated clinical demand for therapeutic enzymes. These conditions frequently require life long and precise enzyme supplementation or replacement to correct metabolic imbalances and prevent serious complications. Government health agencies emphasize early detection and treatment protocols for such conditions due to their potential to cause severe morbidity and long term health system strain if unmanaged. The aggregation of larger patient cohorts with chronic, enzymeimplicated conditions thus intensifies utilization of advanced therapeutic solutions.
Technological Advancements in Enzyme Formulation and Delivery
Effective formulation and delivery systems directly determine therapeutic enzyme performance in patients. Native enzymes are inherently fragile in the human body, with rapid clearance and degradation limiting their circulation time and activity. Government linked biomedical research emphasizes that poor pharmacokinetics and stability often compromise enzyme therapy outcomes, necessitating frequent dosing and reducing clinical utility. Innovative formulation strategies such as polymer conjugation, encapsulation within protective carriers, and nanocarrier based systems protect enzymes from degradation, extend their half life in circulation, and improve bioavailability at the intended site of action, enabling more predictable therapeutic performance. This approach supports safer dose profiles and enhances in body persistence, addressing core pharmaceutical challenges associated with protein biologics.
Government health authorities also document progress in regulatory approvals of biologics as evidence of how delivery innovations are affecting the therapeutic landscape. For instance, U.S. regulatory data for 2025 show an uptick in biologics license approvals featuring recombinant proteins and biologically engineered agents, reflecting industry success in translating advanced biologic formulations into approved therapies. These approvals demonstrate that overcoming inherent limitations through formulation and delivery engineering is integral to advancing enzyme based treatments.
Growing Availability of Lower-Cost Alternatives
The widening presence of lower cost alternatives such as generics and biosimilar biologic therapies exerts significant pricing pressure on established enzyme based treatments. Regulators and health policymakers in 2025 are actively promoting these alternatives to contain overall drug expenditure and expand patient access to essential therapies. For example, the U.S. Food and Drug Administration (FDA) reported that expensive biologic medications accounted for only 5% of prescriptions but contributed 51% of total drug spending as of 2024, highlighting the disproportionate cost impact of complex biologic therapies on health systems; in this environment, streamlined pathways for lower cost biosimilars aim to increase competition and reduce costs for patients and payers.
Government initiatives focused on simplifying development and approval of cost effective alternatives also reshape competitive dynamics. By reducing unnecessary clinical testing and encouraging interchangeability of biosimilars with reference products, regulators are lowering barriers to market entry for affordable options, thereby increasing therapeutic choice and putting downward pressure on pricing. Health systems frequently prioritize treatments with favorable cost benefit profiles, and budget holders may favor proven lower cost alternatives over enzyme therapies that require complex manufacturing, specialized infrastructure, and higher reimbursement levels.
Limited Patient Awareness and Accessibility in Emerging Regions
Limited patient awareness and accessibility in emerging regions remains a major restraint due to structural and systemic shortcomings in health service delivery. The 2025 WHO World Bank Universal Health Coverage monitoring report states that an estimated 4.6 billion people globally still lack access to essential health services, a situation particularly severe in low and middle income countries where knowledge of advanced therapies is low and infrastructure gaps are significant. Healthcare systems in many emerging markets focus mainly on primary care and infectious disease control, leaving complex biologic therapies such as enzyme treatments lower on priority lists. Most rural and remote populations have minimal exposure to specialized care pathways, diagnostics, and treatment options, which limits both clinician and patient understanding of the value and availability of these therapies.
In many emerging regions, limited accessibility worsens the awareness challenge due to financial and logistical barriers that reduce engagement with healthcare systems. Out of pocket expenses, insufficient insurance coverage, and urban centric specialist facilities create travel and cost burdens that lower demand for high cost biologics and enzyme therapies. Without clear patient education campaigns, treatment guidelines, and subsidized access channels from public health agencies or insurers, decision pathways for patients and providers remain unclear, resulting in delayed diagnosis, low uptake of available treatments, and persistent unmet need.
Increasing Prevalence of Chronic and Rare Diseases
The global rise in chronic and rare diseases has created significant demand for targeted therapies that address underlying biological mechanisms rather than solely managing symptoms. Enzyme-based therapeutics provide precise intervention at the molecular level, enabling correction of specific metabolic dysfunctions or breakdown of pathogenic substrates. Conditions such as lysosomal storage disorders, metabolic syndromes, and enzyme-deficiency diseases demonstrate a high unmet need, highlighting the critical role of enzyme therapies in improving patient outcomes and offering treatment options where traditional modalities are limited. The prevalence of such diseases ensures ongoing clinical relevance and sustained interest from healthcare providers and research institutions in developing innovative solutions.
Healthcare systems are increasingly focused on interventions that can improve quality of life, reduce long-term care burdens, and offer cost-efficient disease management. Enzyme therapies can address disease pathophysiology directly, enabling improved efficacy and patient compliance. Governments and regulatory bodies support development and adoption of biologically targeted treatments through streamlined approval pathways and incentive programs, recognizing the potential to reduce healthcare pressures associated with chronic and rare diseases.
Advancement in Biotechnology and Recombinant Technology
Biotechnology and recombinant DNA engineering support reliable creation of therapeutic enzyme products through precision gene integration and controlled expression in host systems such as bacterial, yeast, or mammalian cells. These methods deliver consistent molecular structures and functional activity that align with regulatory quality expectations and clinical performance standards. Engineering platforms for expression reduce dependency on variable natural sources and streamline purification workflows that increase yield and reduce production risk. Government regulatory frameworks for biologics in 2025 show active translation of novel science into approved therapies, with the U.S. Food and Drug Administration approving 46 new novel drugs across small molecules and biologic classes during the year, signaling sustained regulatory throughput for innovative treatments in complex disease segments.
Recombinant platforms unlock cost efficiencies in scaling production and accelerate transitions from discovery to commercial launch through modular workflows that integrate synthetic biology tools, quality by design principles, and digital bioprocess monitoring. These capabilities reduce time to market relative to traditional extraction or chemical synthesis routes and support contract manufacturers and originator producers in meeting diverse demand profiles at scale. Government emphasis on modernizing biologics review processes and promoting advanced manufacturing technologies underscores a policy environment that supports uptake of engineered biologic approaches as core tools in therapeutic development.
Category-wise Analysis
Product Type Insights
Asparaginase is likely to be the leading segment with over 30% of the therapeutic enzymes market revenue share in 2026, due to its established role in leukemia therapy. Clinical acceptance is high, driven by demonstrated treatment effectiveness and inclusion in standard chemotherapy protocols. Hospitals and oncology centers implement structured dosing schedules to optimize therapeutic outcomes, while clinicians monitor enzyme activity levels to manage potential side effects. Provider preference favors asparaginase formulations with proven safety and efficacy profiles, supporting consistent prescription practices. Patient adherence benefits from long-standing clinical familiarity, reinforced by education programs and counseling. Accessibility is enhanced through hospital procurement networks, insurance reimbursement policies, and cold-chain logistics, ensuring continuous availability for treatment continuity.
Collagenase is expected to witness the fastest growth between 2026 and 2033, as demand for enzyme-based wound care and dermatology applications expands. Consumer confidence in enzymatic debridement increases due to clinical evidence demonstrating faster healing times and reduced infection risks in chronic wounds. Provider adoption accelerates with improved patient outcomes, reduced need for surgical interventions, and integration into multidisciplinary wound management protocols. Accessibility is broadened through outpatient clinics, specialized wound care centers, and home-based treatment models, enabling wider patient reach. Innovation in formulation stability, shelf-life extension, and ease of application enhances patient convenience and compliance, further driving market expansion.
Application Insights
Leukemia is poised to lead with a forecasted over 35% market share in 2026, owing to the critical role of enzyme therapy in treatment regimens. Clinical effectiveness drives adoption, supported by robust evidence of improved patient outcomes in both pediatric and adult populations. Physician familiarity with enzyme-based protocols reinforces prescription patterns and treatment consistency. Hospital-based administration ensures adherence and allows careful monitoring of side effects. Insurance coverage and reimbursement policies improve affordability, reducing financial barriers for patients. Preventive healthcare initiatives in high-risk groups contribute to early diagnosis and timely therapy initiation. Digital health platforms facilitate patient monitoring, allowing real-time tracking of response and long-term outcomes, while ongoing research and inclusion in clinical guidelines maintain market stability.
Skin ulcers are anticipated to be the fastest-growing segment between 2026 and 2033, driven by increasing prevalence of chronic wounds, diabetic foot ulcers, and pressure sores. Clinical confidence in enzymatic debridement supports rapid adoption in hospital, outpatient, and home care settings, reducing the need for surgical interventions. Patient awareness and education improve acceptance of non-invasive enzymatic treatments, while retail penetration creates availability through over-the-counter and prescription channels. Integration of digital wound care monitoring systems enables remote tracking of healing progress, supports adherence to treatment schedules, and ensures accurate outcome documentation. Innovation in formulation stability and convenience further enhances patient compliance and expands market reach.
Regional Insights
North America Therapeutic Enzymes Market Trends
North America is expected to hold an estimated 40% of the therapeutic enzymes market share in 2026, reflecting the region's advanced healthcare infrastructure and concentration of clinical expertise. High prevalence of metabolic, hematological, and rare genetic disorders establishes a substantial addressable patient base, while widespread adoption of enzyme-based treatment protocols ensures consistent demand. Strategic integration of advanced biomanufacturing technologies, such as recombinant DNA production, pegylation, and stabilization techniques, enables high-purity and scalable enzyme supply. Regulatory alignment with accelerated approval pathways, facilitated by authorities such as the FDA, reduces time-to-market for innovative therapies, allowing earlier access to novel formulations. Robust insurance coverage and reimbursement mechanisms further drive utilization, enabling hospitals and specialized clinics to offer enzyme therapies with minimal financial barriers for patients.
Dominance is further reinforced by strong collaborative networks between academic institutions, clinical research centers, and commercial entities, enabling rapid translation of scientific discoveries into therapeutic applications. Digital health integration, including electronic health records, remote patient monitoring, and predictive analytics, enhances adherence tracking, outcome measurement, and personalized dosing strategies. High patient and provider trust in clinical protocols promotes early adoption, particularly for rare disease interventions with limited alternative treatments. Capital availability for biopharmaceutical ventures and strategic partnerships supports infrastructure expansion, manufacturing capacity, and distribution efficiency.
Europe Therapeutic Enzymes Market Trends
Europe maintains a stable position in the market for therapeutic enzymes due to structured healthcare systems, established clinical networks, and comprehensive reimbursement frameworks. High prevalence of metabolic, hematological, and chronic conditions in aging populations creates consistent demand for enzyme therapies. National health services prioritize early diagnosis and integration of enzyme treatments into standard care protocols, supporting widespread adoption in hospitals and specialty clinics. Regulatory alignment through the European Medicines Agency (EMA) provides predictable approval pathways, encouraging investment in clinical trials and innovative formulations. Strong presence of multinational and domestic pharmaceutical firms ensures continuous research, advanced manufacturing capabilities, and localized distribution networks.
Innovation adoption is reinforced by collaborative partnerships between academic institutions, biotechnology companies, and clinical research organizations. Investment in formulation stability, recombinant enzyme production, and targeted delivery enhances clinical effectiveness, safety, and patient compliance. Digital health initiatives, including electronic health records and telemonitoring, support treatment tracking and optimization of dosing protocols. Market accessibility is further strengthened through cross-border clinical collaborations and centralized procurement strategies. Capital allocation toward infrastructure modernization, combined with favorable reimbursement mechanisms, ensures operational efficiency and scalability.
Asia Pacific Therapeutic Enzymes Market Trends
The market in Asia Pacific is forecasted to experience the highest growth between 2026 and 2033, driven by rapid expansion of healthcare infrastructure and increasing accessibility to specialized treatment centers. Rising prevalence of metabolic and chronic disorders among urban and aging populations creates a substantial patient pool with unmet therapeutic needs. Strategic investment in hospital networks, outpatient clinics, and diagnostic facilities enhances early disease detection and facilitates timely administration of enzyme therapies. Expansion of domestic biopharmaceutical manufacturing capabilities reduces dependency on imports, shortens supply chains, and lowers treatment costs, supporting broader adoption. Policy initiatives by governments to improve rare disease management, combined with regulatory modernization, streamline approval of novel enzyme formulations, enabling faster market entry.
Rapid adoption is further supported by increasing integration of digital health platforms into patient management and clinical workflows. Telemedicine services and mobile monitoring applications enable remote dosing oversight, adherence tracking, and real-time outcome measurement, particularly in semi-urban and rural areas. Rising awareness among healthcare providers and patients regarding the clinical effectiveness of enzyme therapies enhances acceptance, while educational initiatives on rare disease treatment improve patient engagement. Investments in research collaborations and pilot programs drive innovation in formulation stability, enzyme delivery, and targeted therapies, enabling local adaptation of global treatment standards. Capital inflows from public and private sectors, coupled with favorable reimbursement schemes, improve accessibility and affordability, positioning the market as a high-growth regional market.
Competitive Landscape
The global therapeutic enzymes market structure appears to be moderately consolidated, led by global players such as Pfizer Inc., Sanofi, and Takeda Pharmaceutical Company Limited. Leading firms collectively account for over 55% of revenue share, reflecting strong brand recognition, established distribution networks, and broad clinical adoption of enzyme-based therapies. These companies leverage extensive research and development (R&D) capabilities to innovate next-generation therapeutic enzymes targeting oncology, metabolic disorders, and chronic diseases. Regulatory compliance and adherence to international quality standards remain critical differentiators, enabling these firms to secure approvals for new therapies across multiple regions.
Regional and niche players complement the market by focusing on specialized therapeutic areas and localized manufacturing. Companies such as Novo Nordisk and AbbVie Inc. expand reach through tailored solutions and patient-centric programs that support adoption in specific disease segments. Competitive positioning emphasizes clinical support services, training for healthcare providers, and post-market monitoring to reinforce product credibility. Investment in advanced manufacturing processes improves scalability, supply consistency, and cost efficiency, allowing smaller firms to compete alongside global leaders.
Key Industry Developments
- In January 2026, Spruce Biosciences announced that long term clinical data on its tralesinidase alfa enzyme replacement therapy for Sanfilippo syndrome type B will be presented at the 22nd Annual WORLDSymposium™, highlighting durable reduction of disease biomarkers and stabilization of cognitive outcomes in treated patients.
- In July 2025, scientists at Chiba University reported that disabling the epigenetic enzyme SETD1B significantly slowed the growth of aggressive acute myeloid leukemia cells by reducing MYC oncogene activity, pointing to a promising new targeted treatment strategy.
- In June 2025, researchers at the University of Tokyo reported that targeting the herpes simplex virus1 (HSV1) enzyme uracil DNA glycosylase reactivated natural brain immunity and significantly reduced viral growth in mice, offering a potential new strategy to prevent fatal herpes encephalitis.
Companies Covered in Therapeutic Enzymes Market
- Pfizer Inc.
- Sanofi
- Takeda Pharmaceutical Company Limited.
- Novo Nordisk
- AbbVie Inc.
- Codexis Inc.
- Thermo Fischer Scientific
- Roche Holdings AG
- Novozymes
Frequently Asked Questions
The global therapeutic enzymes market is projected to reach US$ 6.1 billion in 2026.
Rising prevalence of chronic diseases and growing adoption of enzyme-based therapies in clinical treatments are driving the market.
The market is poised to witness a CAGR of 7.2% from 2026 to 2033.
Advancements in biotechnology and recombinant DNA technology present key opportunities for expanding R&D applications.
Some of the key market players include Pfizer Inc., Sanofi, Takeda Pharmaceutical Company Limited, Novo Nordisk, AbbVie Inc., Codexis Inc., and Thermo Fisher Scientific.







