
- Medical Devices
- Single-Use Ultrasonic Probes Market
Single-Use Ultrasonic Probes Market Size, Share, and Growth Forecast, 2026 - 2033
Single-use Ultrasonic Probes Market by Product Type (Diagnostic Probes, Therapeutic Probes), Application (Cardiology, Gastroenterology, Urology, Obstetrics & Gynecology, Others), End-user (Hospitals, Others), and Regional Analysis for 2026 - 2033
Single-Use Ultrasonic Probes Market Size and Trends Analysis
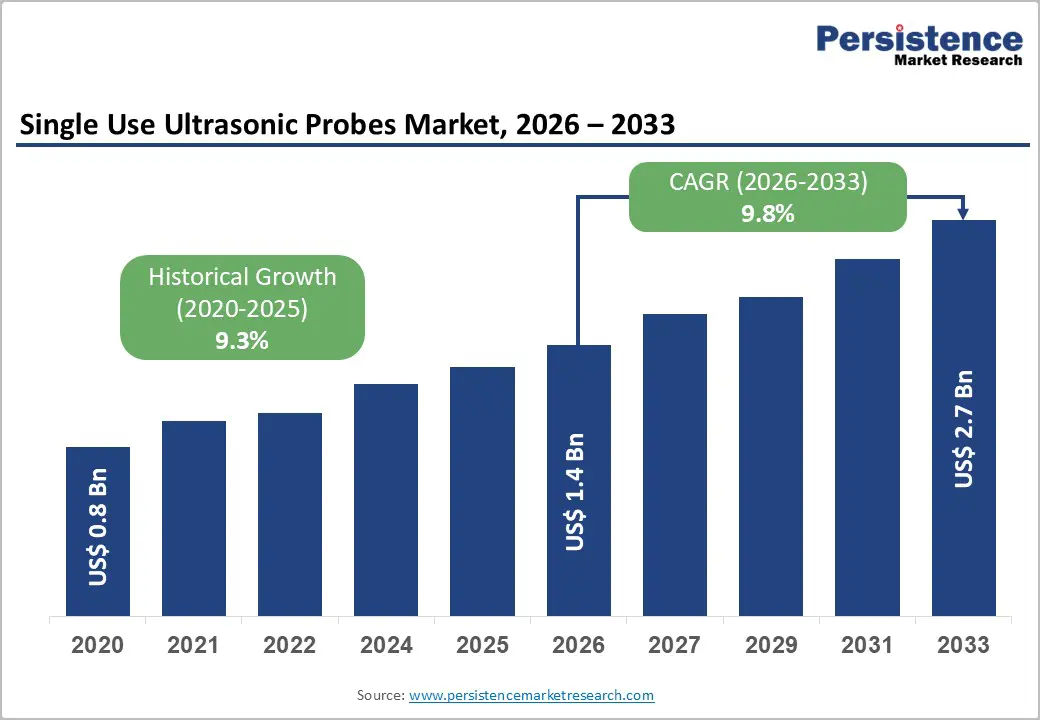
The global single-use ultrasonic probes market size is likely to be valued at US$1.4 billion in 2026, and is expected to reach US$2.7 billion by 2033, growing at a CAGR of 9.8% during the forecast period from 2026 to 2033, driven by the increasing prevalence of hospital-acquired infections, rising demand for infection control in diagnostic imaging, and growing adoption of disposable ultrasound probes in cardiology, urology, and obstetrics procedures.
Growing demand for high-resolution single-use diagnostic probes, especially in hospitals and ambulatory surgical centers, is accelerating adoption across healthcare facilities. Advances in piezoelectric materials, ergonomic designs, and cost-optimized manufacturing are further boosting uptake by offering better image quality and reduced cross-contamination risk. Increasing recognition of single-use ultrasonic probes as critical for patient safety, workflow efficiency, and regulatory compliance in emerging point-of-care and outpatient markets remains a major driver of market growth.
Key Industry Highlights:
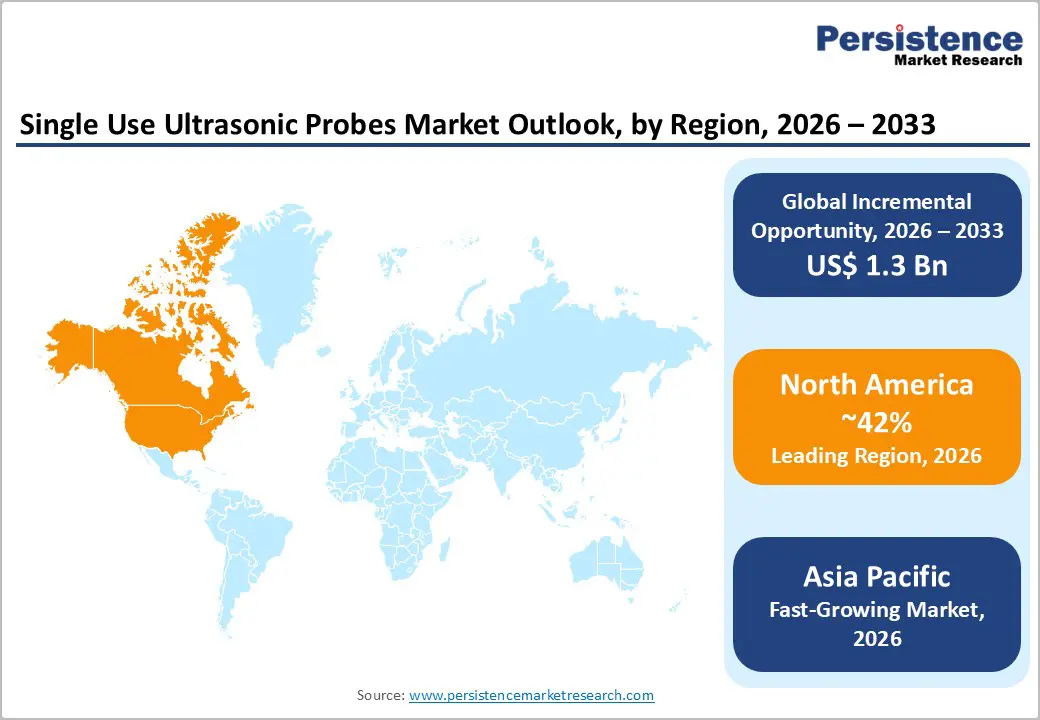
- Leading Region: North America, anticipated to account for a 42% market share in 2026, driven by stringent infection control regulations, high procedure volumes, and strong demand in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by rapid healthcare infrastructure expansion, increasing ultrasound utilization, and rising awareness of HAIs in China and India.
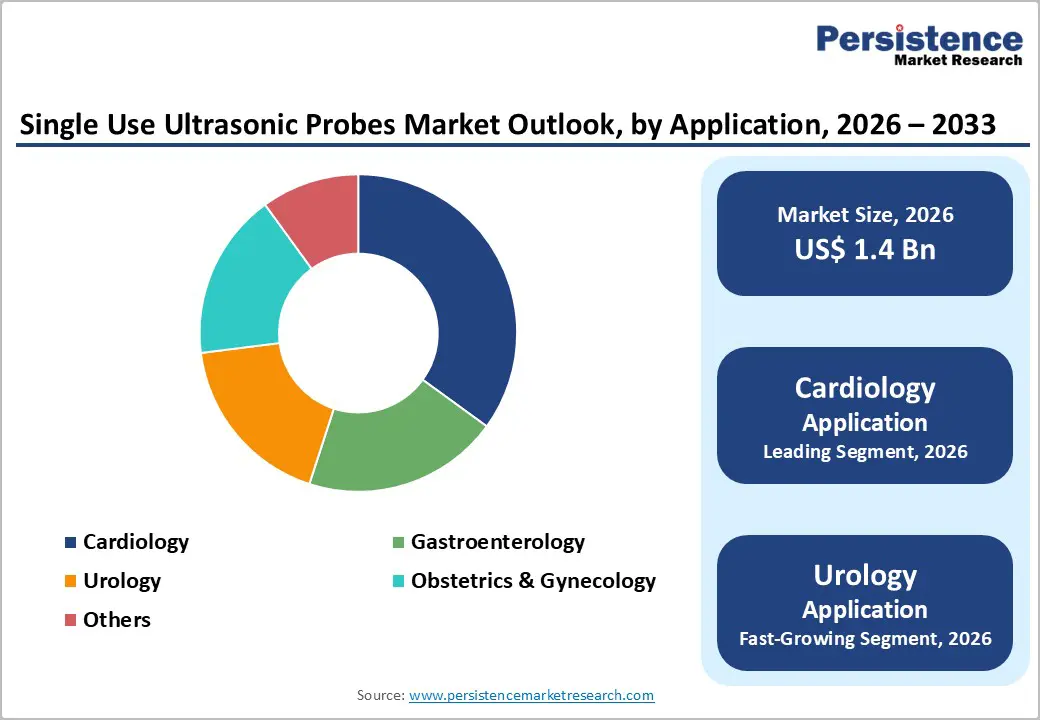
- Dominant Product Type: Diagnostic probes, to hold approximately 68% of the market share, as they remain the primary choice for routine imaging.
- Leading Application: Cardiology, contributing nearly 35% of the market revenue, due to the highest procedural volume.
| Key Insights | Details |
|---|---|
|
Single-Use Ultrasonic Probes Market Size (2026E) |
US$1.4 Bn |
|
Market Value Forecast (2033F) |
US$2.7 Bn |
|
Projected Growth CAGR (2026-2033) |
9.8% |
|
Historical Market Growth (2020-2025) |
9.3% |
Market Factors - Growth, Barriers, and Opportunity Analysis
Growth Analysis - Rising Hospital-Acquired Infections and Infection Control Mandates
Escalating rates of healthcare-associated infections and stricter infection-control mandates are shifting ultrasound workflows toward disposable probe use in acute and outpatient settings. Reusable probes and accessories introduce reprocessing risk across high-turnover environments such as emergency rooms, ICUs, and interventional suites, raising exposure to cross-contamination and compliance lapses. The Centers for Disease Control and Prevention estimated that 1 in 31 hospitalized patients in the U.S. has at least one healthcare-associated infection on any given day, signaling sustained operational risk for hospitals. Single-use ultrasonic probes reduce bioburden transfer, simplify workflow standardization, and lower audit exposure under hospital infection-prevention programs, supporting procurement shifts toward disposables to protect throughput and clinical outcomes.
Regulatory enforcement and facility accreditation elevate the cost of non-compliance across device reprocessing, documentation, and staff training, increasing the business case for disposable technologies. The Centers for Medicare & Medicaid Services reported that 1 in 4 U.S. hospitals had infection-control deficiencies cited during surveys in recent national oversight cycles, reinforcing compliance pressure for device handling protocols. Disposable probes align with mandated infection-prevention standards, reduce reprocessing labor and downtime, and support consistent service levels across high-volume ultrasound pathways.
Increasing Outpatient and Point-of-Care Ultrasound Adoption
Rising adoption of ultrasound in outpatient clinics and point-of-care settings is reshaping care delivery and expanding demand for single-use ultrasonic probes. Bedside imaging accelerates triage in emergency rooms, supports procedural guidance in ambulatory surgery centers, and enables rapid assessments in primary care and specialty clinics. Workflow pressure in high-turnover environments elevates the value of ready-to-use, disposable probes that minimize room downtime, simplify infection-prevention protocols, and support consistent throughput. Procurement teams favor standardized, single-use consumables to align with quality systems, reduce reprocessing labor, and stabilize per-procedure costs across decentralized care sites.
Point-of-care ultrasound integrates into care pathways across anesthesia, critical care, obstetrics, and interventional procedures, increasing probe utilization frequency outside traditional imaging departments. Clinical teams prioritize portability and immediate availability, which supports stocking disposable probes across multiple service lines and satellite clinics. Expanded outpatient reimbursement for image-guided procedures strengthens business cases for ultrasound adoption in community settings. As care shifts from inpatient wards to ambulatory and home-adjacent sites, disposable probe formats support scalable deployment, predictable operating costs, and compliance with infection-prevention policies across distributed networks.
Barrier Analysis - High Cost of Single-Use Probes
The high cost of single-use ultrasonic probes limits adoption despite their clinical and infection-control advantages. Compared with reusable probes, disposables carry a higher per-procedure expense, which can significantly increase operating costs for hospitals, diagnostic centers, and outpatient clinics performing frequent imaging procedures. Budget-constrained facilities, particularly in emerging markets, often face challenges balancing patient safety, infection prevention, and overall cost efficiency. Purchasing disposable probes at scale can strain procurement budgets, forcing administrators to prioritize select departments or procedures for implementation.
Cost sensitivity also affects adoption in ambulatory and small-clinic settings, where margins are tighter and ultrasound volumes are moderate. Reusable probes, despite requiring cleaning and sterilization, remain attractive due to lower upfront and recurring supply costs. Financial pressures encourage providers to assess utilization rates, explore hybrid models combining reusable and disposable probes, or negotiate bulk pricing agreements with manufacturers. The high unit cost of single-use probes continues to be a key restraint in widespread deployment.
Reprocessing Infrastructure and Regulatory Acceptance
The availability of reprocessing infrastructure and regulatory acceptance of reusable ultrasonic probes affect the adoption of single-use probes in healthcare facilities. Hospitals with established sterilization departments, automated cleaning systems, and trained staff can efficiently process reusable probes while meeting infection-control standards, reducing the immediate need for disposable alternatives. Regulatory frameworks in many regions, including CDC guidelines and hospital accreditation requirements, recognize properly reprocessed reusable probes as safe for patient use, reinforcing confidence in conventional workflows.
In facilities with limited reprocessing capacity or high patient throughput, single-use probes offer a simplified solution, but investment in disposables competes with existing capital and operational budgets. Acceptance by regulators and accreditation bodies also shapes procurement decisions, as institutions must demonstrate compliance with infection-prevention protocols regardless of probe type. Facilities often balance cost, workflow efficiency, and compliance risk, weighing whether the benefits of single-use probes justify transitioning from established reusable systems.
Opportunity Analysis - Demand in High-Resolution and Specialized Single-Use Probes
Specialty single-use probes allow healthcare providers to expand ultrasound applications in sensitive or high-risk patient populations, such as neonatal, pediatric, or immunocompromised patients. These probes deliver optimized frequencies and geometries for targeted imaging, improving diagnostic accuracy while eliminating the need for complex sterilization cycles. They also reduce downtime in high-throughput environments, as single-use probes are immediately available for consecutive procedures without cleaning delays.
Government-supported initiatives to improve patient safety and reduce hospital-acquired infections reinforce this demand. The Centers for Medicare & Medicaid Services has linked quality metrics and reimbursement adjustments to infection-control compliance, incentivizing the adoption of disposable, procedure-specific probes. Hospitals that integrate these devices benefit from lower cross-contamination risk, streamlined workflow, and consistent imaging quality across departments, creating a compelling business case for broad adoption in both routine and specialized ultrasound procedures.
Hospital and Clinic Workflow Efficiency
Improving hospital and clinic workflow efficiency is a significant driver for the adoption of single-use ultrasonic probes. In high-volume imaging environments such as emergency departments, operating rooms, and outpatient clinics, time is a critical factor. Reusable probes require thorough cleaning, sterilization, and quality checks between procedures, which can introduce delays, increase labor costs, and reduce patient throughput. Single-use probes are ready-to-use, eliminating reprocessing time and simplifying logistics, allowing clinicians to move quickly between patients without compromising safety or procedural quality. This streamlined workflow supports faster diagnostics, reduces procedure backlogs, and enhances overall operational efficiency.
Hospitals also benefit from reduced risk of human error associated with manual reprocessing protocols. Staff can focus on patient care instead of cleaning cycles, and facilities can maintain consistent turnaround times even during peak hours. Single-use probes minimize downtime caused by equipment maintenance or sterilization bottlenecks, enabling continuous operation across multiple departments.
Category-wise Analysis
Product Type Insights
Diagnostic probes are anticipated to dominate the market, accounting for approximately 68% of the market share in 2026. Their dominance is driven by their essential role in routine imaging and procedural guidance. They are widely used across hospitals, outpatient clinics, and emergency care settings for general-purpose ultrasound, including abdominal, cardiac, and obstetric imaging. Single-use formats reduce cross-contamination risk, simplify infection-control compliance, and support high patient throughput, making them the preferred choice for diagnostic workflows. GE HealthCare single-use diagnostic ultrasound probes in U.S. hospitals for routine abdominal, cardiac, and obstetric imaging. These disposable probes are integrated into emergency rooms, outpatient clinics, and point-of-care settings to reduce cross-contamination and streamline workflow.
Therapeutic probes represent the fastest-growing product type, due to increasing adoption in minimally invasive procedures, pain management, and targeted therapy delivery. These probes enable precise guidance for injections, ablations, and physiotherapy interventions while minimizing infection risk through single-use formats. Hospitals and outpatient centers favor disposable therapeutic probes to reduce sterilization requirements, streamline workflow, and maintain consistent performance across high-turnover procedures. HIFU systems produced by Haifu are used for non-invasive treatments such as tumor ablation and soft-tissue therapy. These systems include specialized ultrasound probes designed to deliver high-intensity focused ultrasound energy to target tissues, offering an alternative to surgical intervention in conditions like uterine fibroids and certain cancers.
Application Insights
Cardiology is expected to dominate the market, contributing nearly 35% of revenue in 2026, due to high demand for imaging in cardiac diagnostics and interventional procedures. Single-use probes reduce cross-contamination risk during echocardiography, intraoperative imaging, and catheter-guided interventions, supporting patient safety in high-risk cardiovascular care. Hospitals and outpatient cardiac centers prefer disposable formats to maintain workflow efficiency, minimize sterilization time, and comply with infection-prevention protocols. A disposable intracardiac echocardiography catheter developed by Shenzhen Sonosemi Medical, which was approved for marketing by China’s National Medical Products Administration (NMPA) in 2025. The device combines a catheter and ultrasound transducer to provide single-use intracardiac imaging of the heart and major cardiac vessels during electrophysiology and interventional procedures, enhancing safety by eliminating reuse and reducing infection risk in cardiac care settings.
Urology represents the fastest-growing application, propelled by increasing use of ultrasound in kidney, bladder, and prostate imaging, as well as in minimally invasive interventions such as biopsies and stone removal. Single-use probes reduce cross-contamination risk in high-risk urinary procedures and streamline infection-control compliance, which is critical in outpatient and hospital urology departments. The convenience of disposable probes supports high patient throughput, minimizes downtime for cleaning and sterilization, and ensures consistent image quality. EMS Electro Medical Systems produces disposable single-use ultrasound probes designed for urology applications, including sterile probes supplied for procedures such as kidney, bladder, and prostate imaging and interventions. These disposable urology probes are supplied sterile to help healthcare providers maintain infection-control standards in high-risk examinations and reduce downtime between patients, supporting patient safety and workflow efficiency in urology departments and clinics.
Regional Insights
North America Single-Use Ultrasonic Probes Market Trends
North America is projected to dominate, accounting for nearly 42% of the share in 2026, driven by the region’s stringent infection control regulations, high ultrasound utilization, and high public awareness of patient safety benefits. Distribution systems in the U.S. and Canada provide extensive support for single-use probe programs, ensuring wide accessibility across diagnostic probes, cardiology, and hospital populations. Increasing demand for high-resolution, convenient, and easy-to-deploy forms is further accelerating adoption, as these formats improve safety and reduce barriers associated with reprocessing.
Innovation in Single-Use Ultrasonic Probes technology, including stable therapeutic, improved wireless delivery, and targeted outpatient enhancement, is attracting significant investment from both public and private sectors. Government initiatives and CDC campaigns continue to promote use against HAI risks, workflow concerns, and emerging disposable threats, creating sustained market demand. The growing focus on urology grades and specialty uses, particularly for diagnostic probes and others, is expanding the target applications for single use ultrasonic probes.
Europe Single-Use Ultrasonic Probes Market Trends
Europe growth is supported by increasing awareness of infection prevention benefits, strong regulatory systems, and government-led healthcare quality programs. Countries such as Germany, the U.K., France, and the Netherlands have well-established ultrasound frameworks that support routine single-use probes and encourage the adoption of innovative diagnostic delivery methods. These high-safety formulations are particularly appealing for cardiology populations, regulation-conscious hospitals, and diagnostic users, improving outcomes and coverage rates.
Technological advancements in single-use probe development, such as enhanced high-frequency arrays, application-targeted delivery, and improved therapeutic grades, are further boosting market potential. European authorities are increasingly supporting research and trials for probes against both routine and specialized needs, strengthening market confidence. The growing emphasis on convenient, disposable options is aligned with the region’s focus on preventive HAI reduction and outpatient expansion. Public awareness campaigns and promotion drives are expanding reach in both hospitals and ASC segments, while suppliers are investing in advanced materials and novel variants to increase efficacy.
Asia Pacific Single-Use Ultrasonic Probes Market Trends
Asia Pacific is likely to be the fastest-growing market for single-use ultrasonic probes in 2026, driven by rising healthcare infrastructure awareness, increasing government initiatives, and expanding application programs across the region. Countries such as China, India, Japan, and South Korea are actively promoting probe campaigns to address imaging growth and emerging safety needs. Single-Use Ultrasonic Probes are particularly attractive in these regions due to their scalable administration, ease of adoption, and suitability for large-scale hospital and cardiology drives in both urban and semi-urban populations.
Technological advancements are supporting the development of stable, effective, and easy-to-use single use probes, which can withstand challenging clinical conditions and minimize infection dependence. These innovations are critical for reaching domestic facilities and improving overall diagnostic coverage. Growing demand for diagnostic probes, cardiology, and hospital applications is contributing to market expansion. Public-private partnerships, increased healthcare expenditure, and rising investment in probe research and production capacity are further accelerating growth. The convenience of probe delivery, combined with improved safety and reduced risk of HAIs, positions it as a preferred choice.
Competitive Landscape
The global single-use ultrasonic probes market is highly competitive, featuring established imaging giants alongside emerging specialists focused on infection-control solutions. In North America and Europe, GE HealthCare and Philips Healthcare maintain leadership through robust R&D capabilities, extensive hospital networks, and strong ties with clinical procedures, supported by innovative diagnostic and high-resolution probe programs. Their solutions enhance imaging accuracy while minimizing cross-contamination risk in high-turnover healthcare settings. In Asia Pacific, local manufacturers advance cost-competitive disposable probes, improving accessibility in emerging markets and enabling broader adoption across hospitals, outpatient clinics, and point-of-care facilities.
Diagnostic single-use probes deliver consistent image quality, streamline workflow, and reduce infection risk, allowing large-scale integration across multiple departments. Strategic partnerships, acquisitions, and co-development initiatives enable companies to combine technical expertise, expand product portfolios, and accelerate commercialization timelines. Therapeutic probe formats address procedural guidance in interventional cardiology and urology, improving clinical outcomes while penetrating high-growth specialty segments, reinforcing overall market expansion globally.
Key Industry Developments
- In June 2025, Royal Philips announced the global launch of the Flash Ultrasound System 5100 POC, a portable point-of-care ultrasound designed for anesthesia, critical care, emergency medicine, and musculoskeletal imaging. The system combined exceptional image clarity, smart automation, and intuitive touchscreen controls, enabling clinicians to perform and document exams in real time. Built on Philips’ established cardiology and general imaging technology, Flash 5100 POC supported rapid, confident decision-making in high-pressure settings such as the ER, ICU, and trauma units, streamlining workflows and enhancing efficiency for users of all experience levels.
- In January 2025, Samsung Medison, a global medical equipment company and affiliate of Samsung Electronics, launched the all-new Samsung Z20 in the U.S., a premium AI-powered ultrasound system designed for advanced OB-GYN applications. The Z20 made its U.S. debut at the Society for Maternal-Fetal Medicine 2025 conference in Denver, Colorado. The system introduced innovations to address diagnostic challenges in high-BMI patients and reduce variability caused by operator experience, while helping mitigate the impact of staffing shortages on exam quality and workflow efficiency.
- In September 2024, GE HealthCare announced the launch of enhanced Venue ultrasound systems and introduced a new point-of-care solution, Venue Sprint. The Venue Sprint, designed for maximum portability, combined the established Venue software with AI-enabled tools, delivered uncompromised image quality, and included wireless probe capability compatible with Vscan Air handheld ultrasound systems. Other devices in the Venue family received upgrades with wireless probe connectivity, additional advanced clinical tools, and the latest ViewPoint™ 6 updates, improving workflow efficiency and optimizing clinical operations across hospitals and point-of-care settings.
Companies Covered in Single-Use Ultrasonic Probes Market
- GE HealthCare
- Philips Healthcare
- Siemens Healthineers
- Canon Medical Systems
- Hitachi Medical Systems
- Mindray Medical International
- Fujifilm Holdings Corporation
- Esaote
- Samsung Medison
- Hologic
- Boston Scientific.
- Others
Frequently Asked Questions
The global single-use ultrasonic probes market is projected to reach US$1.4 billion in 2026.
Growing use of bedside and decentralized ultrasound in emergency, critical care, and outpatient clinics increases demand for single-use probes due to workflow efficiency and infection control.
The single-use ultrasonic probes market is poised to witness a CAGR of 9.8% from 2026 to 2033.
AI-enabled, portable, and wireless probe systems support rapid diagnostics, enhance procedural accuracy, and expand adoption across hospitals, ambulatory centers, and emerging markets.
GE HealthCare, Philips Healthcare, Siemens Healthineers, Canon Medical Systems, and Mindray Medical are the key players.







