- Executive Summary
- Global Primary Care POC Diagnostics Market Snapshot 2026 and 2033
- Market Opportunity Assessment, 2026 - 2033, US$ Bn
- Key Market Trends
- Industry Developments and Key Market Events
- Demand Side and Supply Side Analysis
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definitions
- Market Dynamics
- Driver
- Restraint
- Opportunities
- Trends
- Macro-Economic Factors
- Global GDP Outlook
- Global Healthcare Expenditure
- Forecast Factors - Relevance and Impact
- COVID-19 Impact Assessment
- Value Added Insights
- Value Chain analysis
- Key Market Players
- Product Adoption Analysis
- Key Promotional Strategies by key players
- PESTLE Analysis
- Porter's Five Forces Analysis
- Regulatory and Technology Landscape
- Global Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
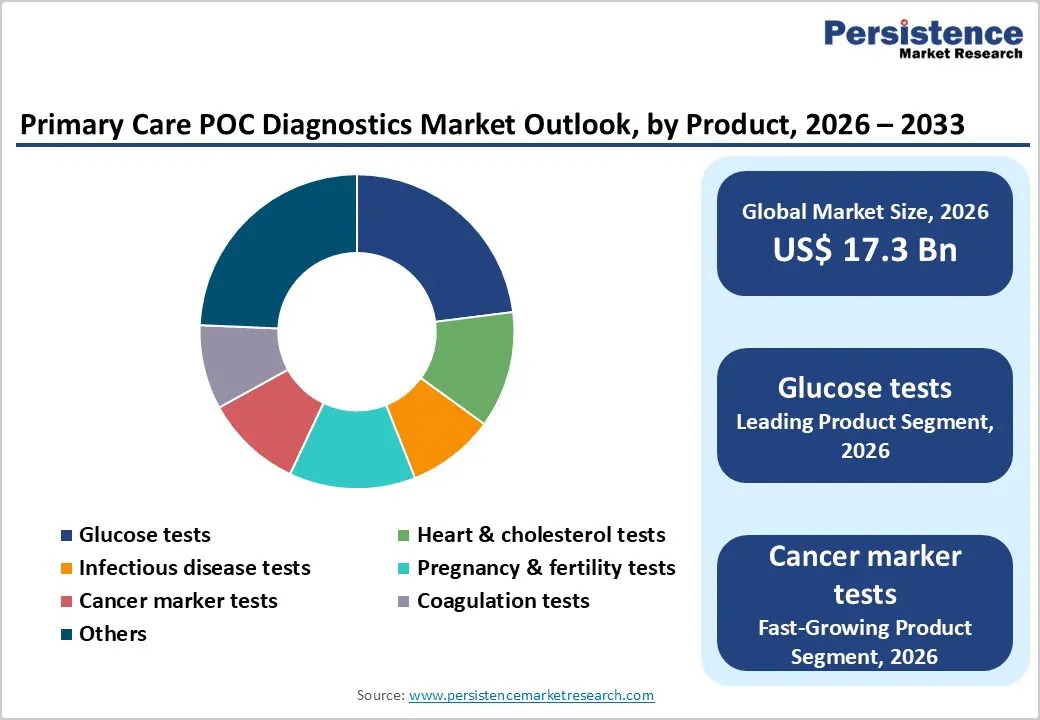
- Global Primary Care POC Diagnostics Market Outlook: Product
- Introduction/Key Findings
- Historical Market Size (US$ Bn) Analysis by Product, 2020-2025
- Current Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- Market Attractiveness Analysis: Product
- Global Primary Care POC Diagnostics Market Outlook: End User
- Introduction/Key Findings
- Historical Market Size (US$ Bn) Analysis by End User, 2020-2025
- Current Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- Market Attractiveness Analysis: End User
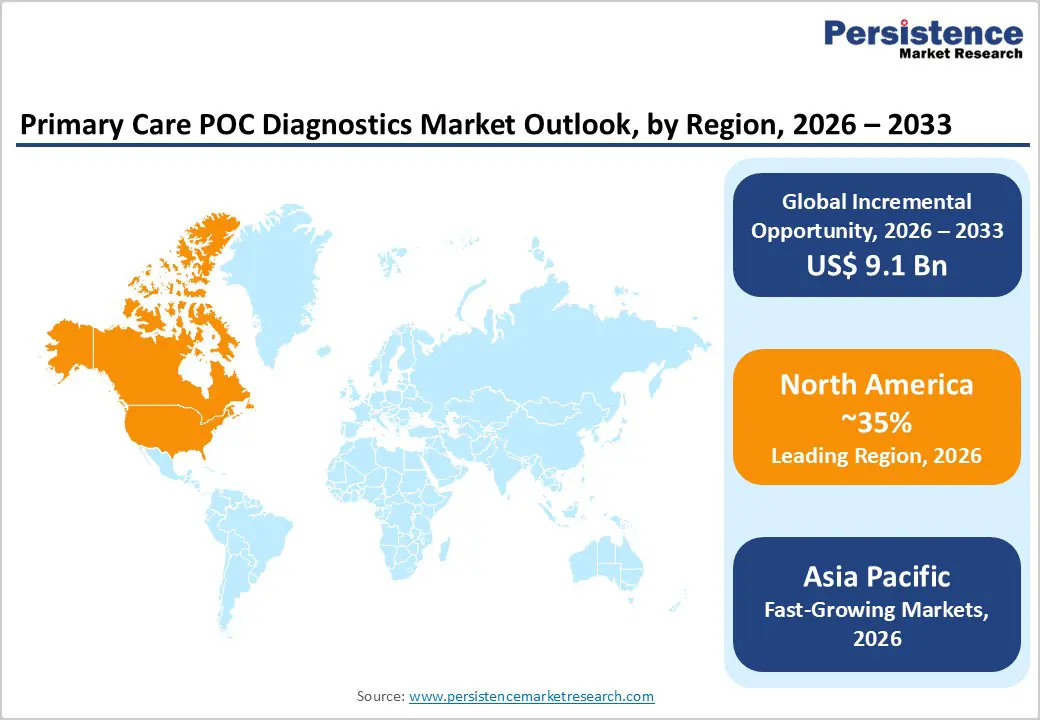
- Global Primary Care POC Diagnostics Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Bn) Analysis by Region, 2020-2025
- Current Market Size (US$ Bn) Forecast, by Region, 2026-2033
- North America
- Europe
- East Asia
- South Asia & Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
- Pricing Analysis
- North America Market Size (US$ Bn) Forecast, by Country, 2026-2033
- U.S.
- Canada
- North America Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- North America Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- Europe Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
- Europe Market Size (US$ Bn) Forecast, by Country, 2026-2033
- Germany
- Italy
- France
- U.K.
- Spain
- Russia
- Rest of Europe
- Europe Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- Europe Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- East Asia Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
- East Asia Market Size (US$ Bn) Forecast, by Country, 2026-2033
- China
- Japan
- South Korea
- East Asia Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- East Asia Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- South Asia & Oceania Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
- South Asia & Oceania Market Size (US$ Bn) Forecast, by Country, 2026-2033
- India
- Southeast Asia
- ANZ
- Rest of SAO
- South Asia & Oceania Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- South Asia & Oceania Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- Latin America Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
- Latin America Market Size (US$ Bn) Forecast, by Country, 2026-2033
- Brazil
- Mexico
- Rest of LATAM
- Latin America Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- Latin America Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- Middle East & Africa Primary Care POC Diagnostics Market Outlook: Historical (2020 - 2025) and Forecast (2026 - 2033)
- Key Highlights
- Middle East & Africa Market Size (US$ Bn) Forecast, by Country, 2026-2033
- GCC Countries
- South Africa
- Northern Africa
- Rest of MEA
- Middle East & Africa Market Size (US$ Bn) Forecast, by Product, 2026-2033
- Glucose tests
- Heart & cholesterol tests
- Infectious disease tests
- Pregnancy & fertility tests
- Cancer marker tests
- Coagulation tests
- Others
- Middle East & Africa Market Size (US$ Bn) Forecast, by End User, 2026-2033
- Hospitals
- Clinics / Diagnostic centers
- Home care
- Pharmacy / Retail clinics
- Research labs
- Competition Landscape
- Market Share Analysis, 2025
- Market Structure
- Competition Intensity Mapping
- Competition Dashboard
- Company Profiles
- Roche Diagnostics
- Company Overview
- Product Portfolio/Offerings
- Key Financials
- SWOT Analysis
- Company Strategy and Key Developments
- BD Biosciences
- Danaher Corporation
- Abbott Laboratories
- Instrumentation Laboratory
- bioMerieux
- Siemens Healthcare
- Quidel Corporation
- Roche Diagnostics
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations
Primary Care POC Diagnostics Market Share and Trends Analysis
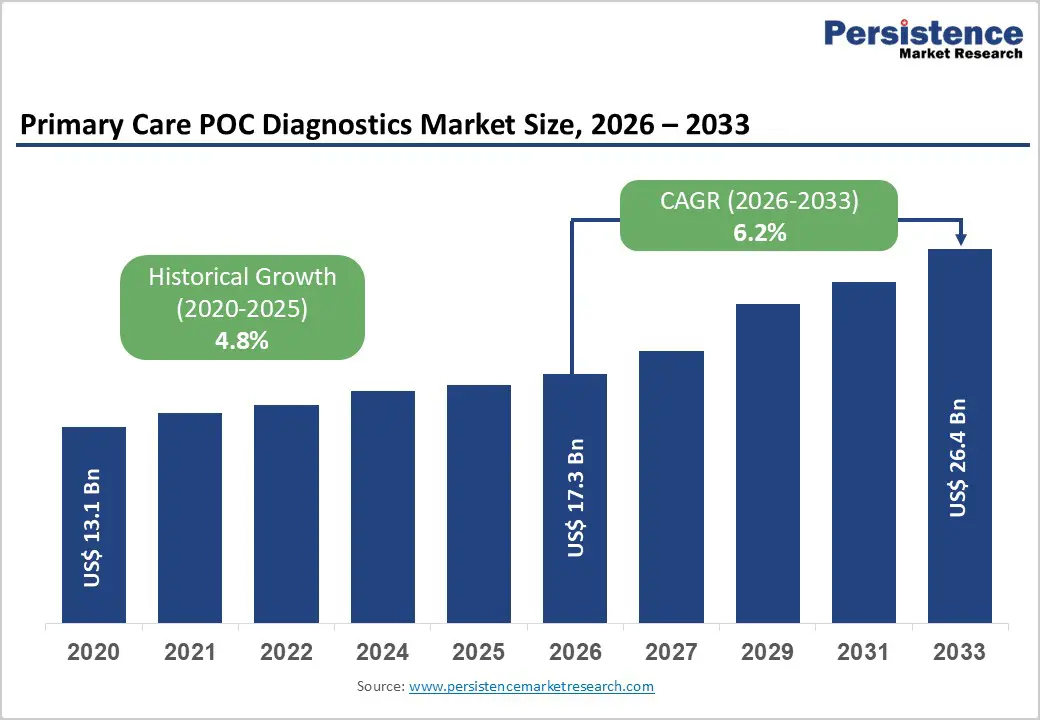
The global primary care POC diagnostics market is expected to be valued at US$ 17.3 billion in 2026 and projected to reach US$ 26.4 billion by 2033, growing at a CAGR of 6.2% between 2026 and 2033.
Market expansion is fundamentally driven by three distinct structural mechanisms rather than incremental demand shifts. First, technological inflection points in high-sensitivity cardiac troponin assays achieving near-perfect correlation with laboratory-based platforms (r = 0.999) enable 1-hour ACS rule-out decision pathways, establishing POC testing as a clinical prerequisite rather than a convenience alternative in emergency medicine. Second, regulatory expansion of CLIA-waived testing, combined with 2025 regulatory updates strengthening Proficiency Testing and personnel qualifications, enables pharmacy and clinic deployment of molecular diagnostics multiplexing (enabling simultaneous influenza/RSV/COVID differentiation and targeted antiviral selection), creating structural demand independent of individual disease prevalence. Third, geographic market asymmetries position Asia Pacific as the fastest-growing region through manufacturing cost convergence, enabling affordable POC deployment across rural primary health centers in China, India, and ASEAN, addressing diagnostic infrastructure gaps affecting over 20 countries facing 20%+ chronic disease premature death risk.