
- Medical Devices
- Off-Loading Devices Market
Off-Loading Devices Market Size, Share, and Growth Forecast, 2026 - 2033
Off-Loading Devices Market by Product Type (Off-loading Footwear, Total Contact Casts), End-user (Hospitals, Specialized Clinics, Healthcare Stores, Online Stores), Application (Diabetic Foot Ulcer Management, Pressure Ulcer Management, Post-Surgical Rehabilitation), and Regional Analysis for 2026-2033
Off-Loading Devices Market Share and Trends Analysis
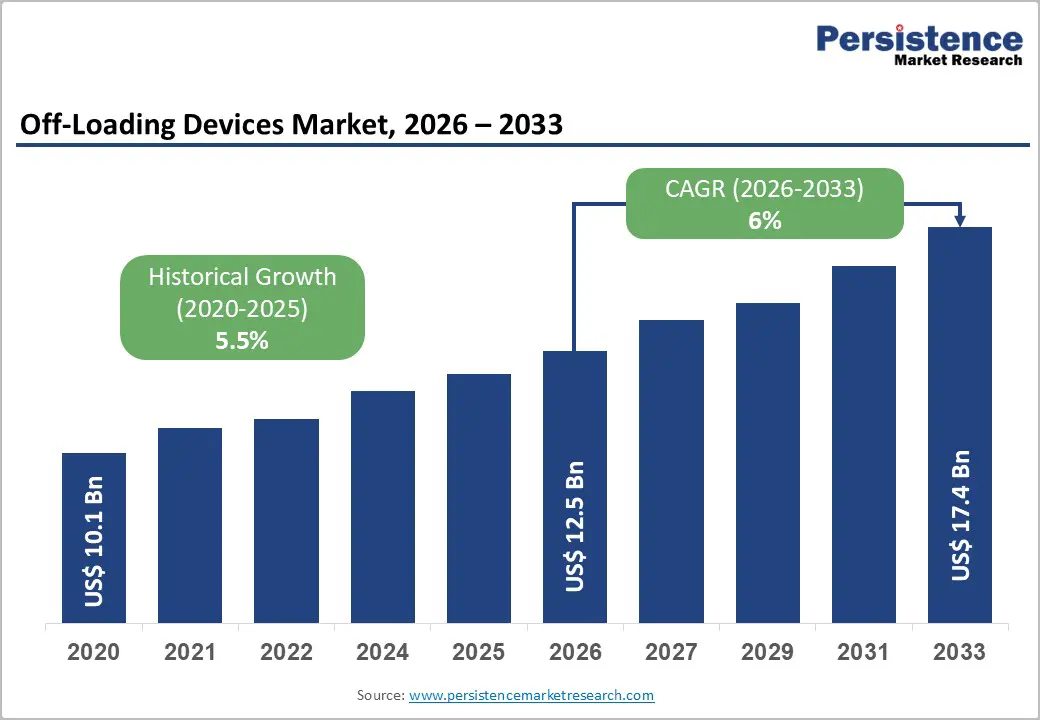
The global off-loading devices market size is likely to be valued at US$ 12.5 billion in 2026, and is projected to reach US$ 17.4 billion by 2033, growing at a CAGR of 6% during the forecast period 2026−2033. This growth is occurring as demand is increasing across healthcare, industrial, and logistics sectors where pressure redistribution and load management solutions are essential for safety and operational efficiency. In healthcare settings, rising awareness of pressure injury prevention and diabetic foot management is driving adoption of specialized devices designed to reduce tissue stress and improve patient outcomes. Aging populations are also contributing to demand because older adults are experiencing higher risk of mobility limitations and chronic conditions that require supportive equipment. These demographic and clinical factors are creating sustained demand across hospitals, long-term care facilities, and home care environments.
Market expansion is also being supported by regulatory compliance requirements, infrastructure investments, and the growing emphasis on preventive care strategies across both developed and emerging economies. Healthcare systems are prioritizing early intervention approaches to reduce treatment costs associated with complications such as pressure ulcers and mobility-related injuries. Technological advancements are improving device functionality through enhanced materials, ergonomic design, and patient monitoring capabilities, which is increasing clinical effectiveness and user acceptance. In industrial and logistics sectors, organizations are adopting off-loading solutions to reduce worker injury risk and improve productivity, which is expanding the market beyond traditional medical applications.
Key Industry Highlights
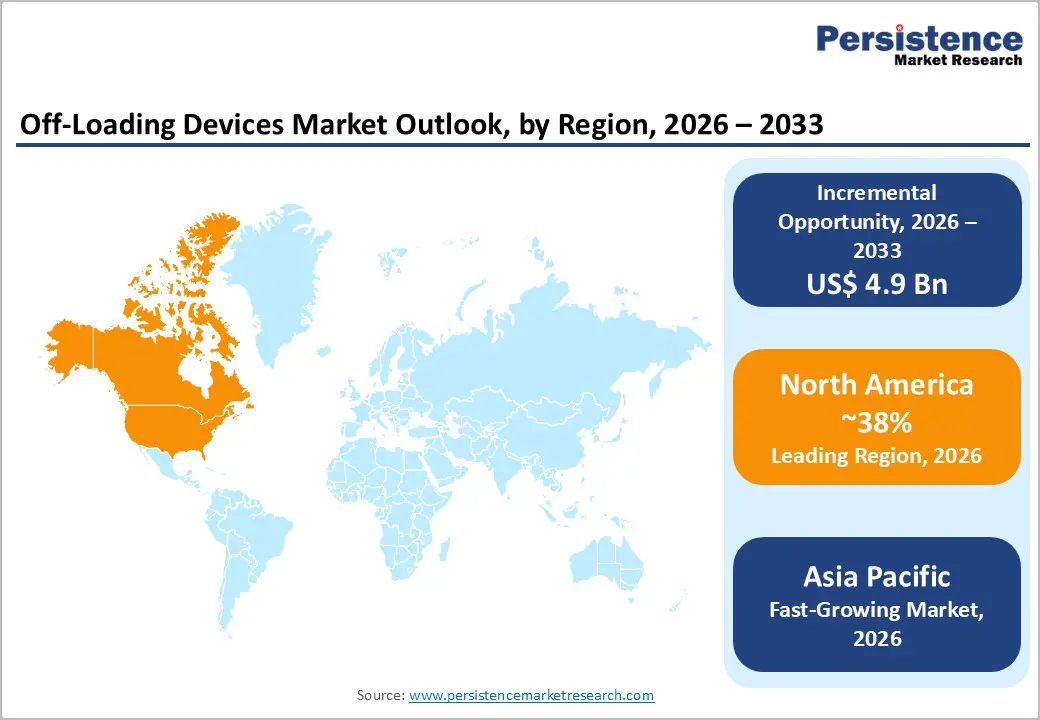
- Dominant Region: North America is expected to hold around 35% market share in 2026, owing to robust healthcare reimbursement systems.
- Fastest-growing Market: The Asia Pacific market is set to be the fastest-growing through 2033, aided by rapid healthcare infrastructure development and aging population trends.
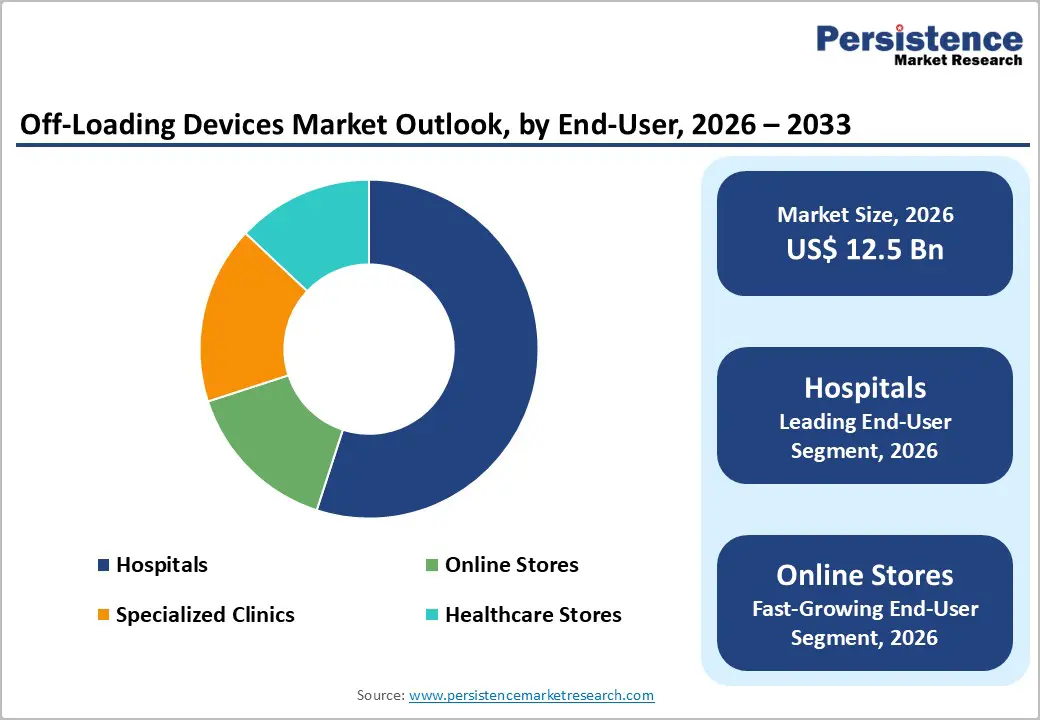
- Dominant End-User: Hospitals are slated to dominate with an approximate 55% revenue share in 2026, benefiting from integrated systems that link devices to electronic health records (EHRs).
- Fastest-growing End-User: Online stores are expected to grow the fastest over the 2026-2033 forecast period, fueled by e-commerce expansion.
- Market Drivers: Advancements in materials science, pressure distribution technologies, and digital health integration are transforming the off-loading device landscape.
| Key Insights | Details |
|---|---|
| Off-Loading Devices Market Size (2026E) | US$ 12.5 Bn |
| Market Value Forecast (2033F) | US$ 17.4 Bn |
| Projected Growth (CAGR 2026 to 2033) | 6% |
| Historical Market Growth (CAGR 2020 to 2025) | 5.5% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Rising Prevalence of Pressure Injuries and Chronic Diseases
Healthcare institutions worldwide are prioritizing preventive strategies to reduce the incidence of pressure ulcers and diabetic foot complications, which are common among patients with diabetes mellitus (DM). Offloading devices are playing a critical role by redistributing pressure away from vulnerable anatomical areas, thereby lowering the risk of tissue damage and subsequent complications. Hospitals and long-term care facilities are investing in these solutions because prevention is reducing overall treatment costs associated with wound management, surgical interventions, and prolonged hospitalization. Regulatory authorities are reinforcing this approach through accreditation requirements and clinical guidelines that emphasize preventive care protocols. As a result, the demand for effective offloading technologies is expanding across acute care, rehabilitation centers, and home healthcare environments.
Healthcare providers are increasingly incorporating offloading solutions into routine patient management plans as technological innovation improves device usability and effectiveness. Advances such as embedded pressure monitoring sensors and smart footwear systems are enhancing patient adherence by providing real-time feedback and performance tracking. Customizable designs are accommodating diverse mobility levels and anatomical needs, which is improving clinical outcomes and patient comfort. Training programs for healthcare professionals are emphasizing proper device fitting and usage techniques to maximize therapeutic benefits. Lightweight materials and ergonomic designs are enabling patients to maintain mobility confidence while undergoing treatment. Policy initiatives that support funding for accessible medical technologies are further strengthening adoption, which is helping healthcare systems reduce long-term resource burdens and improve population health outcomes.
Technological Innovation and Product Development
Advancements in materials science, pressure redistribution technologies, and digital health integration are transforming the offloading device landscape by improving clinical performance and patient usability. Manufacturers are incorporating innovations such as pressure-sensing systems, smart textiles, and lightweight composite materials to enhance therapeutic effectiveness while maintaining comfort during prolonged use. These developments are improving patient adherence as devices are becoming more ergonomic and less restrictive to mobility. Companies are also developing three-dimensional (3D) printed orthotic devices that enable precise anatomical customization, which is improving pressure relief accuracy and reducing complications associated with improper fitting. Internet of Things (IoT)-enabled monitoring systems are allowing real-time tracking of pressure distribution, which is providing clinicians with actionable insights to optimize treatment plans.
Clinical research is demonstrating improved outcomes from advanced offloading technologies compared with traditional solutions, which is encouraging healthcare systems to adopt innovative devices. Manufacturers are investing significantly in research & development (R&D) to meet evolving clinical requirements and competitive pressures while complying with regulatory standards. Engineers are designing breathable and skin-friendly materials that minimize irritation and improve patient comfort, while embedded sensors are alerting caregivers to pressure changes that may increase injury risk. Future device designs are expected to integrate artificial intelligence (AI) capabilities that enable predictive adjustments based on patient movement and usage patterns. Portable devices that connect with mobile applications are supporting remote monitoring and personalized therapy adjustments, which is enhancing clinical decision-making.
High Initial Capital Investment and Cost Barriers
Advanced offloading devices are requiring substantial capital investment, particularly for pressure redistribution beds and complex orthotic solutions that involve specialized materials and integrated technologies. Healthcare facilities operating under budget constraints, especially in developing regions, are facing adoption barriers despite clear clinical benefits. Smaller institutions are often hesitant to invest because reimbursement policies are not fully aligning with rising manufacturing and operational costs, which is compressing margins for device producers. Supply chain disruptions related to specialty components are further increasing production expenses and delivery timelines. Institutional procurement processes are frequently prolonged due to administrative approvals and budget reviews, which is delaying payments and creating cash flow challenges for manufacturers.
Healthcare professionals are increasingly prioritizing cost containment over innovation in financially constrained environments, which is influencing purchasing behavior. Providers are exploring alternative financing models such as leasing arrangements and installment-based procurement to reduce upfront expenditure burdens. Governments are also considering subsidy programs and funding initiatives to improve access to essential medical equipment within public healthcare systems. Manufacturers are responding by partnering with financial institutions to offer flexible payment solutions and by redesigning products to reduce material usage without compromising performance. Facilities are adopting modular systems that allow incremental scaling based on demand, which is improving affordability and operational flexibility. These strategies are expected to stabilize revenue streams, expand market access, and balance affordability with quality healthcare delivery over time.
Regulatory Complexity and Compliance Burdens
Fragmented regulatory frameworks across global markets are creating significant barriers to entry and expansion for offloading device manufacturers. Companies are facing varying approval requirements across regions, which is increasing the need for extensive clinical testing, quality assurance processes, and regulatory documentation. In the U.S., the Food and Drug Administration (FDA) classifies certain advanced offloading systems as Class III medical devices, which requires rigorous clinical trials and comprehensive safety validation before commercialization. Manufacturers are therefore modifying product designs to meet country-specific standards, which is increasing production complexity and reducing economies of scale associated with mass manufacturing. Smaller companies are experiencing disproportionate compliance burdens due to limited regulatory resources, which is slowing market entry and restricting global expansion opportunities.
Regulatory misalignment is also forcing producers to develop separate product versions for regions such as Europe, Asia, and North America, which is fragmenting research and development investments and delaying international product launches. Governments and trade organizations are increasingly advocating for mutual recognition agreements to simplify approval pathways and facilitate cross-border device distribution. Manufacturers are participating in industry associations to influence policy development while simultaneously standardizing core components across product variants to reduce engineering redundancy. Regulatory authorities are gradually adopting digital submission platforms that are expected to accelerate review timelines in the future. Strategic prioritization of markets with clearer regulatory pathways is enabling faster commercialization, while ongoing harmonization efforts are expected to improve device accessibility and stimulate innovation worldwide.
Aggressive Healthcare Infrastructure Development in Emerging Economies
Rapid healthcare infrastructure expansion across Asia Pacific, Latin America, and the Middle East is opening unprecedented opportunities for offloading devices as governments continue investing in hospitals, clinics, and specialized care facilities to serve growing populations. India, China, and several Southeast Asian nations are increasing healthcare expenditure to improve access to medical services, which is generating steady demand from public healthcare institutions. Rising middle-income populations are also supporting the growth of private healthcare providers, where families are seeking preventive care and early intervention solutions for chronic conditions. Healthcare providers are adopting offloading technologies to manage complications associated with diabetes and mobility disorders at earlier stages, which is helping reduce long-term hospitalization costs and improving patient outcomes. This infrastructure development is strengthening healthcare delivery capacity while expanding market potential for medical device manufacturers.
Companies are responding by forming regional manufacturing and distribution partnerships to reduce costs and improve market access through localized production strategies. Training programs for local healthcare professionals and sales teams are improving service quality and customer engagement, while customized product designs are addressing diverse patient needs across different demographic groups. Governments are encouraging foreign investment through incentives and policy support, which is facilitating technology transfer and supply chain development. Private clinics and home healthcare networks are integrating portable offloading solutions into preventive care and rehabilitation programs, expanding adoption beyond hospital settings. Manufacturers are also building strong after-sales support systems and leveraging digital platforms for caregiver training and patient education, creating long-term growth opportunities.
Digital Health Integration and Remote Monitoring Capabilities
Integration of offloading devices with digital health platforms is enabling continuous pressure monitoring, predictive analytics, and remote clinical decision support across healthcare settings. Connected device ecosystems are allowing providers to track patient adherence, monitor pressure distribution patterns, and identify early indicators of tissue damage before complications develop. Engineers are designing these systems to support seamless data transmission between devices and clinical interfaces, while clinicians are accessing real-time insights through mobile dashboards and centralized monitoring platforms. Patients are receiving immediate feedback on device usage, which is improving adherence and encouraging proactive self-management. Telehealth adoption is further accelerating the use of home-based offloading solutions, particularly in rural and underserved areas where remote oversight is improving access to specialized care.
Healthcare teams are increasingly adjusting treatment strategies based on live data streams that provide objective insights into patient behavior and clinical progress. Families are using mobile applications to record daily activity, monitor outcomes, and communicate concerns promptly with care providers. Developers are creating AI algorithms that predict pressure injury risk and recommend preventive interventions, while hospitals are integrating these platforms with EHRs to maintain comprehensive patient information. Nurses are conducting virtual consultations to ensure proper device fitting and functionality, which is improving treatment effectiveness outside traditional clinical environments. These innovations are reducing complications, enhancing patient independence, and enabling scalable digital healthcare solutions that strengthen long-term outcomes and commercial opportunities for industry participants.
Category-wise Analysis
Product Type Insights
Footwear is projected to hold an estimated 65% of the off-loading devices market revenue share in 2026. These devices provide effective pressure redistribution while allowing patients to maintain mobility during treatment. Products such as adjustable walking boots and therapeutic shoes are widely used across hospitals and outpatient clinics due to their ease of application and adaptability to different clinical needs. Clinicians are valuing features such as breathable materials, ergonomic construction, and customizable fitting systems that accommodate diverse foot anatomies. Manufacturers are integrating cushioning layers and rigid sole structures to reduce shear forces and localized pressure points, which is improving therapeutic outcomes. The segment is also leading the market due to strong inclusion within routine diabetic foot management protocols and high reported success rates in ulcer prevention.
Total contact casts are expected to be the fastest-growing during the 2026 to 2033 forecast period, since they provide superior immobilization and pressure offloading for complex wound cases. These casts, typically made from plaster or fiberglass materials, encase the entire foot and lower leg to minimize movement and promote consistent pressure redistribution. Surgeons are applying them in acute ulcer management scenarios where precise offloading is critical for healing. Innovations such as lightweight composites and rapid-setting materials are improving patient comfort and clinical efficiency, which is increasing acceptance among healthcare providers. Home healthcare teams are training patients on maintenance and monitoring to reduce complications associated with prolonged casting. Clinical evidence demonstrating faster healing outcomes compared with removable devices is driving demand, while manufacturers are expanding production capacity to meet rising requirements from specialized wound care centers.
End-User Insights
Hospitals are expected to remain the prime end-user with an approximate 55% off-loading devices market share in 2026, owing to the capacity of acute care facilities to manage high patient volumes requiring wound prevention and mobility support interventions. These institutions are deploying offloading devices across specialized wound care units where multidisciplinary teams, including podiatrists and vascular specialists, are integrating them into structured treatment protocols. Hospitals are also benefiting from integrated systems that connect medical devices with EHRs, which is improving clinical documentation and treatment tracking. Regulatory requirements emphasizing pressure injury prevention and patient safety are reinforcing hospital adoption, while staff training programs can sustain long-term utilization across clinical settings.
Online stores are projected to record the highest 2026-2033 CAGR, fueled by the proliferation of e-commerce platforms and increasing adoption of home-based care solutions. Patients and caregivers are gaining access to a broad selection of offloading devices through digital platforms that provide detailed product information, comparison tools, and user reviews to support purchasing decisions. Retailers are collaborating with manufacturers to enable direct-to-consumer delivery models, which are improving accessibility and reducing procurement delays. Telehealth consultations are frequently recommending specific products, which is driving online purchasing behavior. Subscription-based services are also emerging to supply replacement components and ongoing guidance, which is enhancing customer retention.
Regional Insights
North America Off-Loading Devices Market Trends
North America is projected to account for approximately 38% of the off-loading devices market value in 2026 on the back of its advanced healthcare infrastructure and strong emphasis on preventive care. The U.S. drives regional leadership through well-established medical facilities, structured reimbursement systems, and regulatory frameworks that encourage adoption of technologies aimed at reducing hospital-acquired pressure injuries. Healthcare providers are increasingly implementing preventive solutions to avoid financial penalties associated with complications and readmissions, which is strengthening demand for offloading devices. The FDA has defined approval pathways for medical devices, which is supporting innovation and commercialization. Technology clusters in states such as California, Massachusetts, and Texas are advancing smart materials and connected medical solutions, enabling hospitals to integrate these devices into routine wound care management.
Demographic and lifestyle factors are further supporting market growth across the region, as aging populations and increasing obesity prevalence are raising the incidence of mobility-related conditions and chronic wounds. Canada and Mexico are expanding healthcare investments to address similar trends, while private healthcare providers are growing alongside public systems, which is accelerating device adoption. Integrated care networks are linking hospitals with home healthcare services to support continuous patient monitoring and treatment continuity. Policymakers are enforcing quality standards that promote preventive interventions, encouraging healthcare institutions to adopt advanced offloading technologies. Data analytics tools are supporting clinicians in refining treatment strategies, which is improving outcomes and strengthening North America’s market position.
Europe Off-Loading Devices Market Trends
Europe is likely to occupy a prominent position in the global market for off-loading devices, boosted by its well-established universal healthcare systems and strong regulatory frameworks that emphasize preventive care. Germany, the U.K., France, and Spain spearhead regional performance through advanced medical infrastructure, aging population management programs, and established manufacturing capabilities. The European Union (EU) Medical Device Regulation (MDR) is standardizing approval requirements across member states, which is facilitating product distribution while ensuring rigorous clinical validation. Germany is maintaining leadership in healthcare technology adoption and infrastructure modernization, while the UK National Health Service (NHS) is prioritizing evidence-based procurement strategies focused on prevention and long-term cost efficiency. France and Spain are increasing investments in healthcare services to support elderly populations, and Nordic countries are allocating substantial funding toward proactive wound management programs.
Regulatory alignment across Europe is reducing market entry barriers while improving operational efficiency for manufacturers and healthcare providers. Demographic aging trends across major economies are creating consistent demand for advanced medical technologies that support mobility and wound prevention. Quality standards and clinical guidelines are encouraging healthcare institutions to adopt technologically advanced devices that improve patient outcomes. Investment activity is increasing in digital health solutions and telehealth-enabled monitoring systems, which is enhancing integration between devices and clinical workflows. Companies are also targeting Central and Eastern Europe as infrastructure development accelerates, creating new growth opportunities through regional partnerships. Hospitals are integrating connected systems with electronic patient records to improve monitoring and treatment coordination, while clinicians are adopting standardized protocols to ensure consistent device usage. Future market development is expected to emphasize rural healthcare delivery and AI-enabled monitoring capabilities, reinforcing Europe’s influence on global preventive care standards.
Asia Pacific Off-Loading Devices Market Trends
The market in Asia Pacific is expected to register the fastest growth due to rapid healthcare infrastructure development and significant demographic shifts across the region. China and India are driving demand through large-scale hospital construction programs and increasing emphasis on preventive healthcare strategies to manage chronic diseases. Governments are upgrading healthcare systems to address rising rates of diabetes and mobility-related conditions, while aging populations are creating urgent demand for effective pressure redistribution solutions. Private healthcare providers in India are expanding services to meet the needs of growing urban populations, offering customized products and specialized care programs. Manufacturers are leveraging regional production capabilities to deliver cost-effective devices, and multinational companies are establishing local manufacturing facilities to reduce logistics expenses and improve market responsiveness.
Demographic changes and increasing prevalence of chronic diseases are sustaining long-term market expansion across Asia Pacific as healthcare spending continues to rise. Governments are prioritizing quality standards and medical technology adoption, while regional initiatives led by the ASEAN are promoting regulatory harmonization to facilitate trade and market access. Local manufacturers are competing with global companies through efficient distribution networks and regionally tailored product offerings. Hospitals are incorporating digital tools to improve patient monitoring and treatment coordination, and clinicians are adapting devices to accommodate diverse anatomical and mobility needs within community healthcare settings. Investors are funding joint ventures that combine innovation with affordability, and emerging wearable integrations with mobile applications are expected to enhance remote monitoring capabilities.
Competitive Landscape
The global off-loading devices market structure is moderately consolidated, led by Stryker, Hill-Rom Holdings, Invacare Corporation, DJO Global, and Medline Industries. Market structure reflects significant barriers to entry, including strict regulatory compliance requirements, capital-intensive production processes, and long-standing procurement relationships with hospitals and healthcare systems. These factors are favoring established manufacturers that possess strong distribution networks and clinical credibility. However, fragmentation remains evident within segments such as orthotic footwear and customized devices, where regional producers and specialized firms are maintaining competitive advantages through tailored product offerings and localized market knowledge.
Industry consolidation is gradually accelerating as major manufacturers pursue acquisitions to expand technological capabilities and product portfolios. Larger companies are acquiring specialized offloading device manufacturers and digital health startups to integrate advanced monitoring technologies and data-driven solutions into their offerings. Vertical integration strategies are also strengthening supply chain control and improving operational efficiency, which is enhancing profitability and market positioning. Companies are increasingly investing in digital health platforms that connect devices with patient monitoring systems, creating additional revenue streams through service-based models.
Key Industry Developments
- In February 2026, Andhra Pradesh MedTech Zone (AMTZ) signed a five-year memorandum of understanding (MoU) with Narayan Seva Sansthan (NSS) to co-develop and manufacture affordable, high-quality prosthetic, orthotic, and assistive technologies in India, combining AMTZ’s manufacturing ecosystem with NSS’s clinical expertise.
- In March 2025, RF Health acquired the HeelP.O.D. pressure offloading device from NelDerm to strengthen its pressure injury prevention portfolio, expanding solutions that address heel ulcers and sacral injuries, which are among the most common hospital-acquired pressure injuries.
Companies Covered in Off-Loading Devices Market
- Stryker Corporation
- Hill-Rom Holdings
- Invacare Corporation
- DJO Global
- Össur Corporation
- Medline Industries
- GE Healthcare
- Span-America Medical Systems
- Kinetic Concepts International
- Theralogix LLC
- Hollister Incorporated
- Smith & Nephew plc
- Jaco Electronics
- umotech
Frequently Asked Questions
The global off-loading devices is projected to reach US$ 12.5 billion in 2026.
The market is primarily driven by the rising prevalence of diabetes and pressure ulcers, aging populations, regulatory mandates for prevention, and healthcare investments.
The market is poised to witness a CAGR of 6% from 2026 to 2033.
Key market opportunities lie in digital health integration with IoT monitoring and expansion into home-based telehealth solutions, which offer strong growth potential.
Stryker Corporation, Hill-Rom Holdings, Invacare Corporation, DJO Global, and Medline Industries are some of the key players in the market.







